Oct 13th - Lipid Bilayer
lipid bilayer separate intracellular spaces from the outside environment
a cell consists of isolated space(s) that are capable of regulated exchange with their surroundings
an ideal barrier is naturally impermeable to the majority of molecules present in both the environment & the cell
most biological molecular are hydrophilic but a purely hydrophobic barrier would not be stable in aqueous solution
cell membranes consist of lipid bilayers and associated proteins
the basic structure of cell membranes is a bilayer of mostly phospholipids and associated proteins
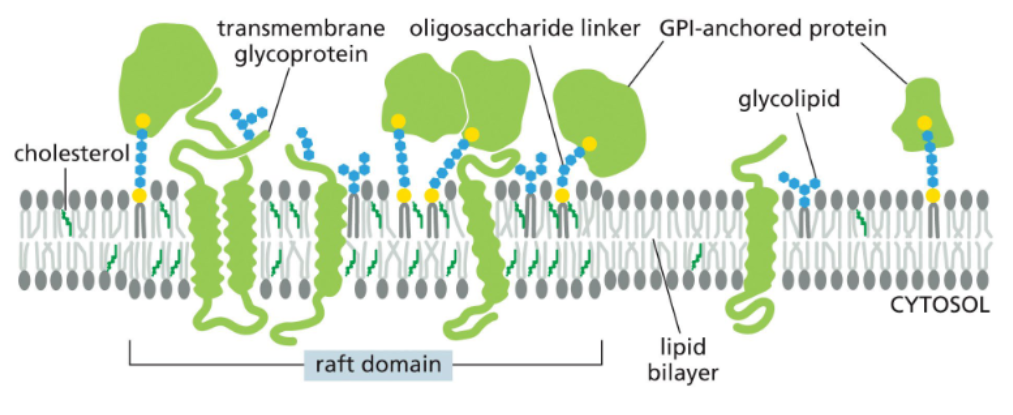
some proteins and lipid compositions can isolate particular plasma membrane regions into functionally distinct “rafts”
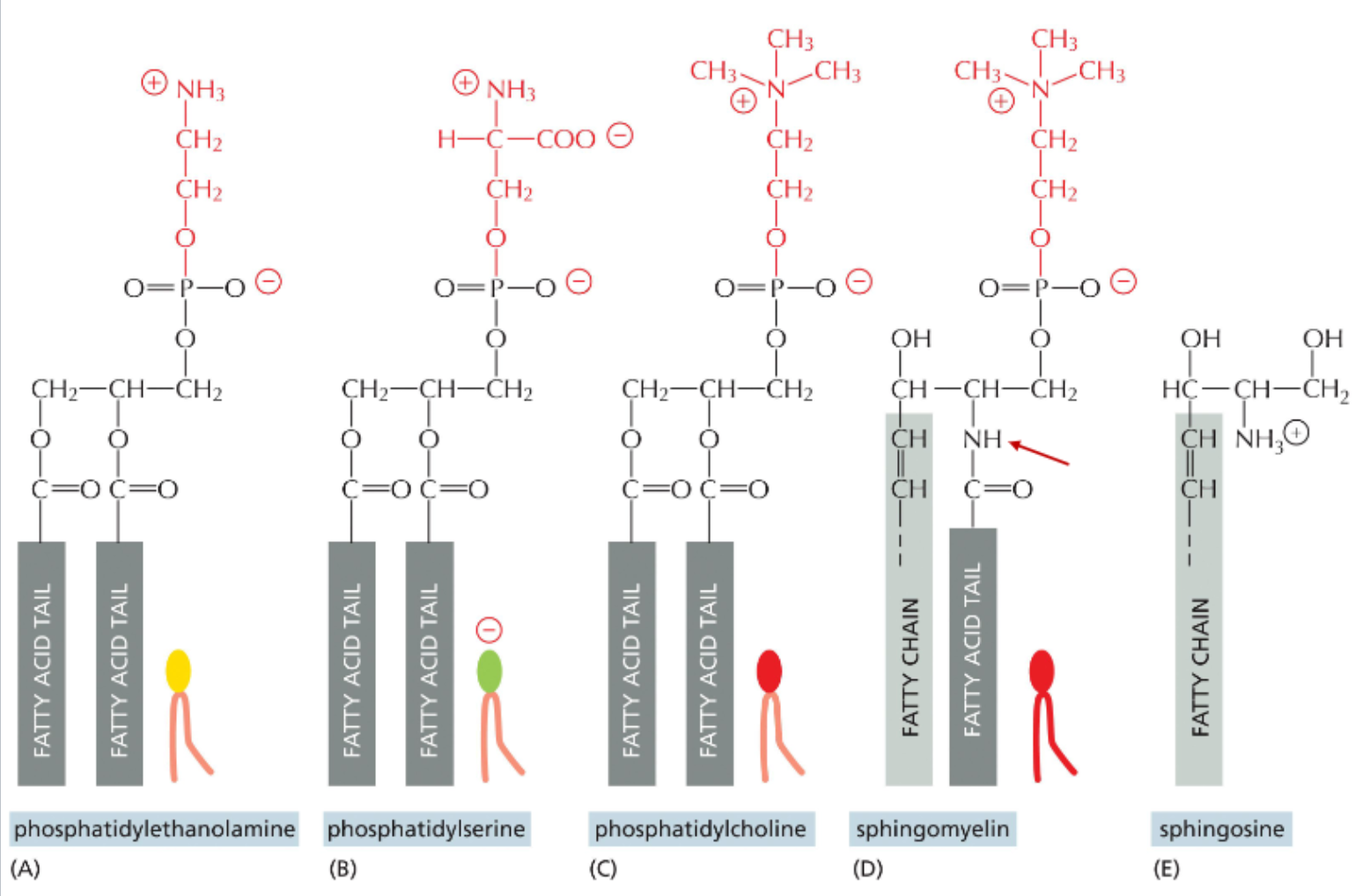
phospholipids are the main component of lipid bilayers
the most abundant lipids in the membrane are phospholipids
phospholipids consist of glycerol linked to 1 or 2 fatty acids and phosphate
the phosphate group is linked to many different types of head group
one fatty acid chain is often unsaturated
saturation & length of the fatty acid chains vary and impact membrane fluidity
the four main lipids of the mammalian plasma membrane
phosphatidylserine has a net negative charge
sphingolipids are derivatives of sphingosine & contain an amino group in place of a hydroxyl on their backbone
cholesterol
a slightly amphiphilic molecule; short tail
steroids have a hydroxyl group — slightly hydrophobic
typically orients itself in the bilayer with their hydroxyl group close to the polar head groups of adjacent phospholipid molecules
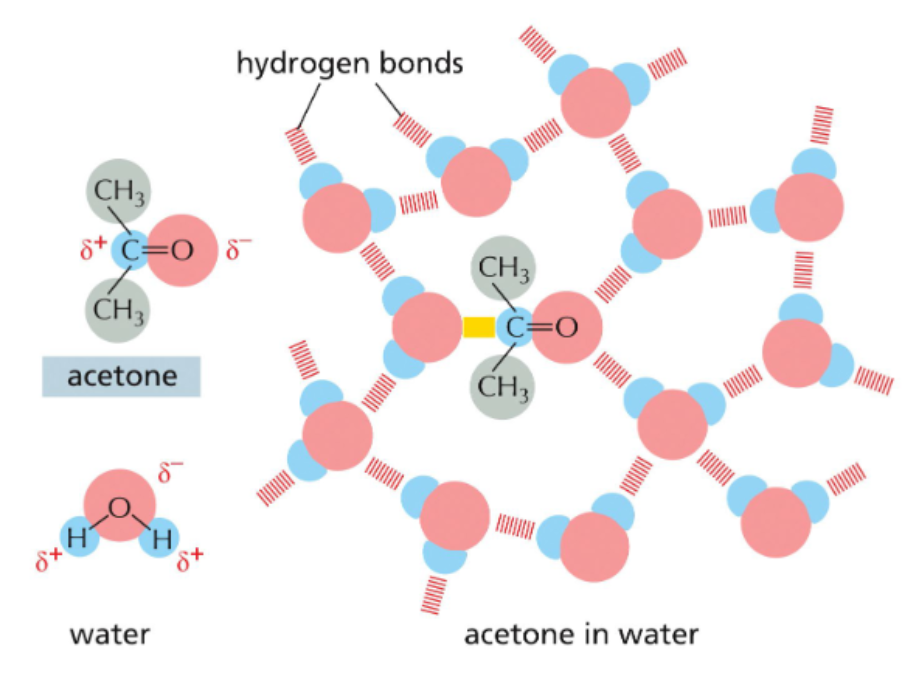
hydrophilic molecules hydrogen bond with water
hydrophilic molecules dissolve readily in water because they contain charged groups or uncharged polar groups that can form either favorable electrostatic interactions or hydrogen bonds with water molecules
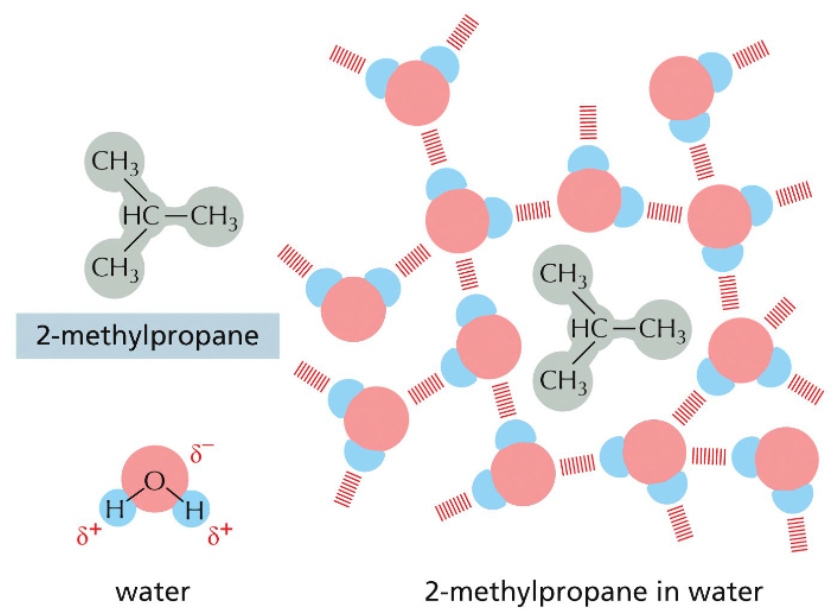
hydrophobic molecules are excluded from hydrogen bond networks
nonpolar molecules lack charge separation & cannot electrostatically interact with water
hydrophobic molecules disrupt the hydrogen bonding network of aqueous solutions
more hydrogen bonds are possible if hydrophobic molecules are aggregated into one space
more hydrogen bonds = more disorder = more entropy
hydrophobic molecules aren’t attracted to each other; they are pushed together
amphiphilic molecules form structures that minimize hydrophobic contact with water
they spontaneously pack together to minimize exposure of their hydrophobic tails to water and maximize exposure of their hydrophilic heads to water
depending on their shape, the optimal packing arrangement is achieved in either of two ways: micelle or bilayer
sphere formation limits contact between hydrophobic layer with water
in water, a lipid bilayer is inherently unstable because lipid tails are exposed to water at the edges
random folding eventually results in bilayer sphere in which no hydrophobic residues are exposed to water
energetically favorable; pure phospholipids in water will form liposomes spontaneously
phospholipid within a bilayer is dynamic
once a liposome is established, individuals’ lipids in the bilayer are capable of movement
lateral diffusion — movement of a phospholipid on the same leaflet of the bilayer
flexion — repositioning of a lipids hydrophobic tails relative to its polar head group
rotation — spinning of a phospholipid
flip-flop — movement of a phospholipid from one leaflet to other
energetically unfavorable; must be facilitated by flippases
membrane compositions influences its fluidity
fluidity is the degree of freedom of movement among membrane lipids
membrane fluidity is a function of both temperature and composition
a membrane with a higher concentration of unsaturated lipids will be more fluid than one with more saturated lipids (double bonds in tail region limit interaction & stacking)
cholesterol is a fluidity buffer
it prevents increases in fluidity w/temp by obstructing lipid movement
it prevents decreases in fluidity at lower temperatures by interfering with stacking
it also decreases the membrane permeability
lipids in synthetic liposomes spontaneously partition into phase separation rafts
liposomes formed from different mixtures of lipids can spontaneously form phase separated regions in which certain lipids are enriched
raft-like domains may form in living cells via interactions between lipids & proteins
organize and concentrate membrane proteins for transport in membrane vesicles or working together in protein assemblies, such as when they convert extracellular signals into intracellular ones
membrane lipids are distributed asymmetrically in the plasma membrane
the asymmetrical distribution of membrane lipids is functionally significant
on the plasma membrane:
glycolipids are restricted to the extracellular leaflet where they form a nearly continuous sugar coat (glycocalyx) around the cell — some cells are recognized based on sugars on their membrane lipids & proteins
glycolipids are formed through modification of membrane lipids by enzymes in the golgi
restriction of phosphatidylserine to the inner leaflet contributes to the net negative charge of the inner leaflet
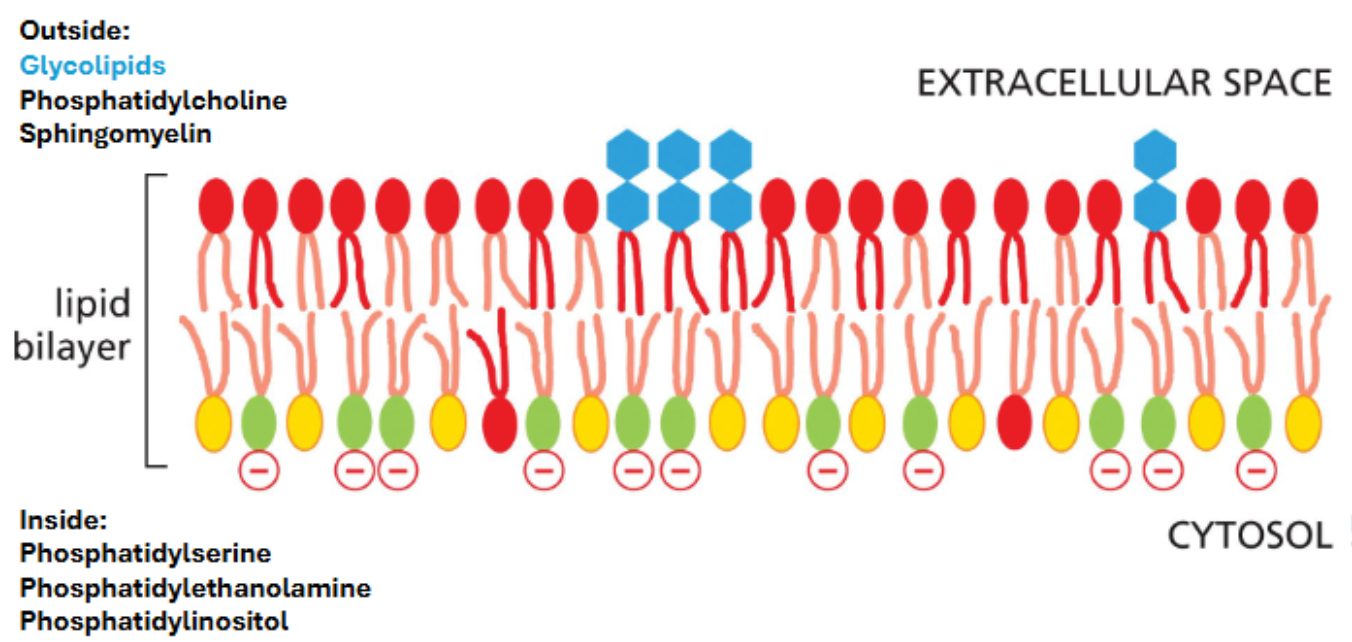
plasma membrane lipid asymmetry contributes to several processes
glycolipids on the outer leaflet facilitate interactions between cells
phosphatidylinositol of varying levels of phosphorylation reside on the inner leaflet & mediate signaling events
protein kinase C gets activated from extracellular signals — localizes to the membrane only at phosphatidylserine aggregates
phosphoinositide 3 kinase (PI3K) converts PIP2 → PIP3 & generates a docking site for the signaling protein Akt
phosphatidylserine on the outer leaflet signals for cell apoptosis
glycolipids are sugar-modified lipids
sugar-containing lipids — glycolipids
exclusively in the outer leaflet
addition of sugars to lipids occurs in the golgi lumen
some pathogens exploit glycolipids
cholera toxin binds to GM1 ganglioside
polyoma viruses also bind to gangliosides
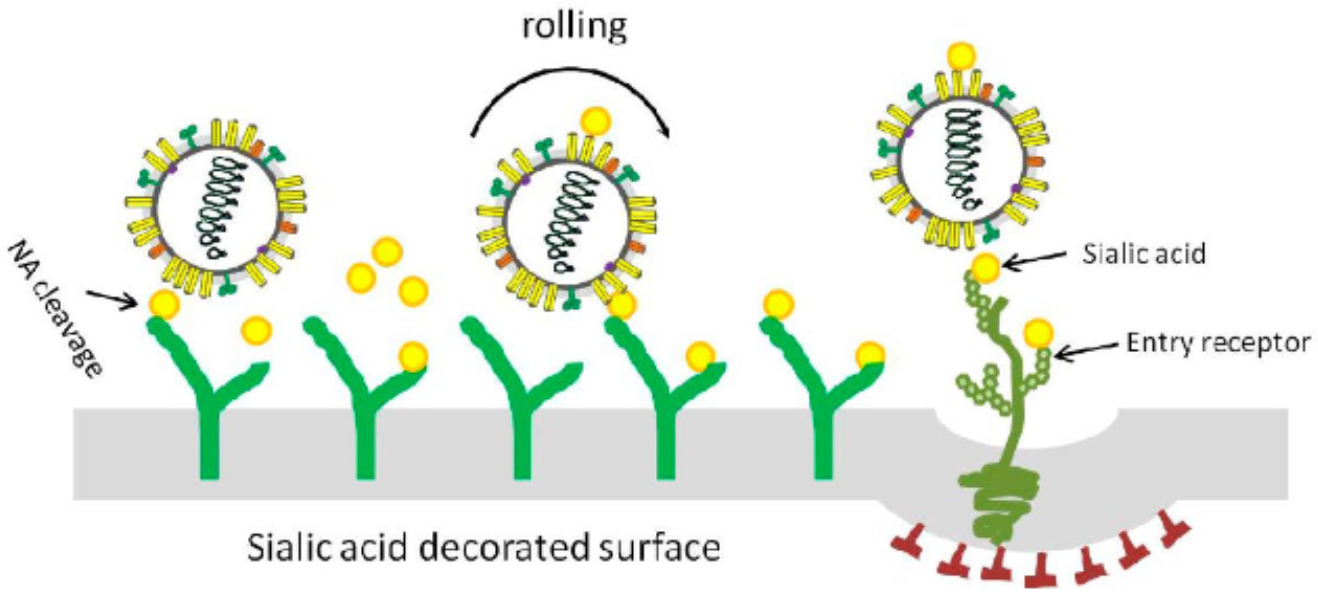
influenza binds to sialic acid residues
when the virus encounters its entry receptor it is internalized
new flu viruses stick to surface sialic acids of its host cell; its neuraminidase cleaves the sugar to release the virus
different influenza subtypes are classified by their specific hemagglutinin and neuraminidase proteins
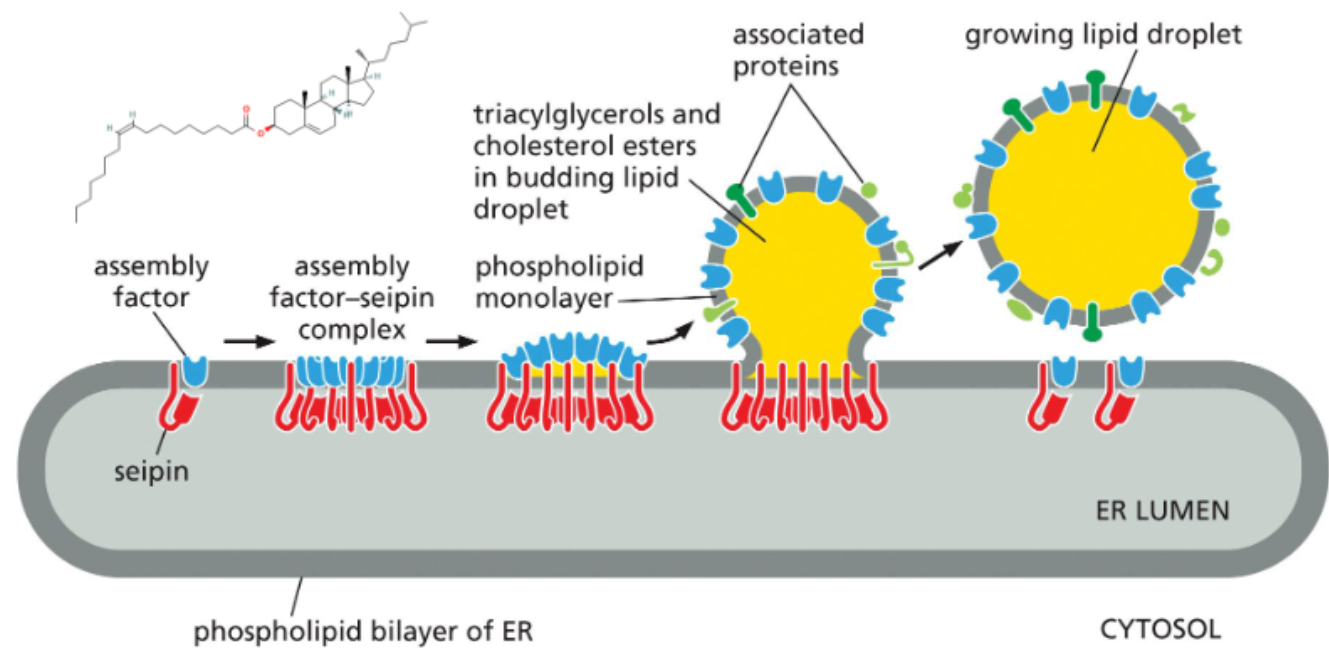
triglycerides and cholesterol esters are stored in single-layered vesicles
both components are not amphiphilic so they will not form micelles or liposomes
these fats are stored in lipid droplets that form within the lipid bilayer of the ER
as the droplet expands, a single leaflet surrounds it bringing ER membrane proteins with it
some of these proteins are involved in lipid metabolism
in cells of the intestines, the droplets form inside the ER lumen and are trafficked out of cell
KEY CONCEPTS
Membranes separate cells’ inner spaces from their surrounding environment and (among eukaryotes) allow reactions the partitioned to different parts of the cell
Membrane formation relies on the amphipathic nature of its comprising lipids – these molecules arrange into bilayer spheres (liposomes) to maximize contact between polar groups and water and minimize contact between hydrophobic groups and water.
Multiple lipid types contribute to membranes, and they are distributed unequally between the two leaflets.
Phosphatidylcholine, glycolipids, and sphingolipids are most common on the outer leaflet; phosphatidylethanolamine,
phosphatidylserine, and phosphatidylinositol are more common on the inner leaflet.
Membrane fluidity refers to the movement of individual lipids in the membrane. Lipids can diffuse, flex, rotate, and flip, but flipping is the least energetically favorable and very slow.
Different lipids partition into phase separated lipid rafts in synthetic liposomes. Similar partitioning of lipids and membrane proteins can be seen in living cells, but the process is more dynamic.
Unequal distributions of lipids on membranes contributes to cellular functions such as cell-cell recognition and intracellular signaling.
Glycolipids and glycoproteins on the outer leaflet form a glycocalyx around cells. Pathogens can bind to components of the glycocalyx.