Ionic Bonding
Ion- a charged particle, (when particles lose/gain electrons)
cation- positive ion (lost electrons, metals)
anion- negative ion (gained electrons, nonmetals)
Group 14- don't generally gain or lose 4 electrons, don't usually form ionic compounds
Transition elements- form different ions
Why do transition elements form different ions- electron configuration allows them to lose different number of elements from d subshell
ionization - where electrons are transferred between atoms oppositely charged ions result
what are oppositely charged ions held together by- electrostatic forces (ionic bonds)
charge of ionic compounds- electrically neutral

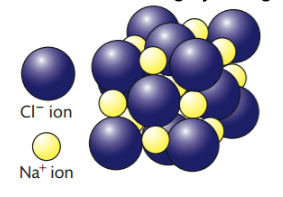
Ionic Compounds have a Lattice Structure
ionic lattice - predictable 3D crystalline structure, fixed arrangement of ions based on a repeating unit
coordination number - number of ions that surround a given ion in the lattice (NaCl lattice, the coordination number is 6 because each Na + ion is surrounded by 6 Cl ions and each Cl ion is surrounded by 6 Na + ions)
lattice energy - measure of the strength of attraction between the ions within the lattice
lattice energy for smaller ions- (greater for ions that are small and highly charged, as they have a larger charge density)
The Physical Properties of Ionic Compounds Reflect Their Lattice Structure
tend to have high melting and boiling points as the forces of electrostatic attraction between the ions in the lattice are strong and so require lots of energy to break
solids at room temperature
volatility
tendency of a substance to vaporize
low volatility
low odour
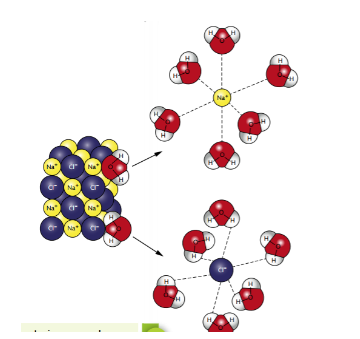
easily hydrated (ions are surrounded by water molecules)
ionic compounds are generally soluble in ionic polar solvents but not soluble in nonpolar solvents
dissolved in something other than water, they become solvated
don't conduct electricity in solid state,
but will when molten or in aqueous solution brittle (crystals shatter under force)
Different Ionic Compounds have a different extent of Ionic Character
binary compound - contains 2 elements
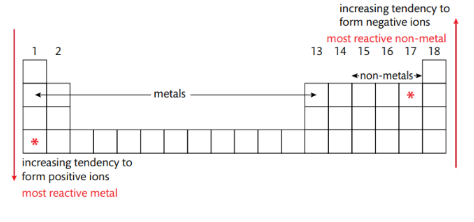
Ionic Compound Electronegativity Difference- 1.8 or higher on the Pauling Scale
Pauling Scale Trend on PT- Increase along period , up a group
Bonding Continuum

Bonding Triangle
Covalent Bonding
2+ non metals
form by atoms sharing electrons
shared electrons concentrated in region between 2 nuclei and is attracted to both
held together- electrostatic attraction (covalent bond)
formation of covalent bond - stabiizes atoms so energy is released as bond forms
forces of attraction between nuclei and shared electrons balanced by - forces of repusion between 2 nuclei (holds atoms apart at fixed distance)
octect rule- atoms react to acheive full valence shell with 8 outer electrons
diatomic molecules- 2 atoms
3 atoms- triatomic
lone pair/non bonding pair- electrons not involved in forming bond, important in shape of molecule
Atoms can share more than 1 pair of electrons to form multiple bonds
doube bond-
Short Bonds are Strong Bonds
bond length- measure between 2 bonded nucei
bond enthalpy- amount of energy needed to break bonds
multipe bonds- greater number of shared electrons
stronger electrostatic attraction between bonded nuclei