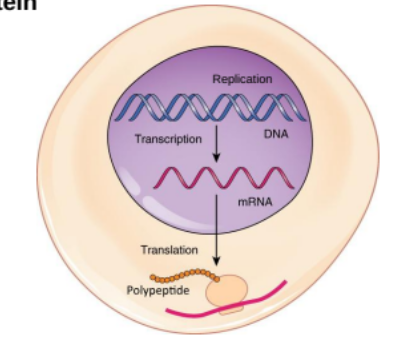
Protein synthesis
From Nucleic Acids to Proteins
Lecture Information
Lecture presented by Dr. Susanna Cogo, University of Reading.
Email: s.cogo@reading.ac.uk
Intended Learning Outcomes (ILOs)
At the end of this lecture you will be able to:
Discuss the structure and functions of proteins.
Explain the genetic code.
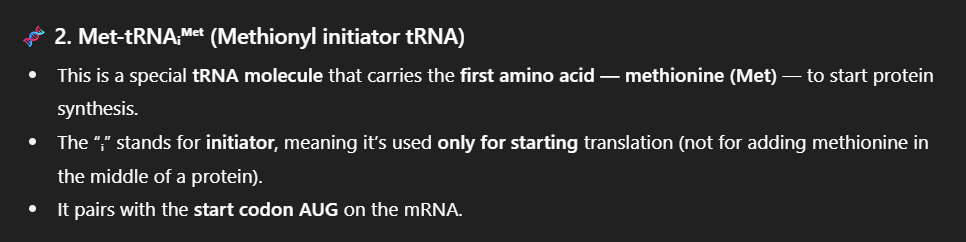
Describe the structure and function of transfer RNA (tRNA) and ribosomal RNA (rRNA).
Outline the main steps of translation.
Overview of Translation
Process of Translation
Translation is the process by which a protein is synthesized from the information contained in a molecule of messenger RNA (mRNA).
Functions of Proteins
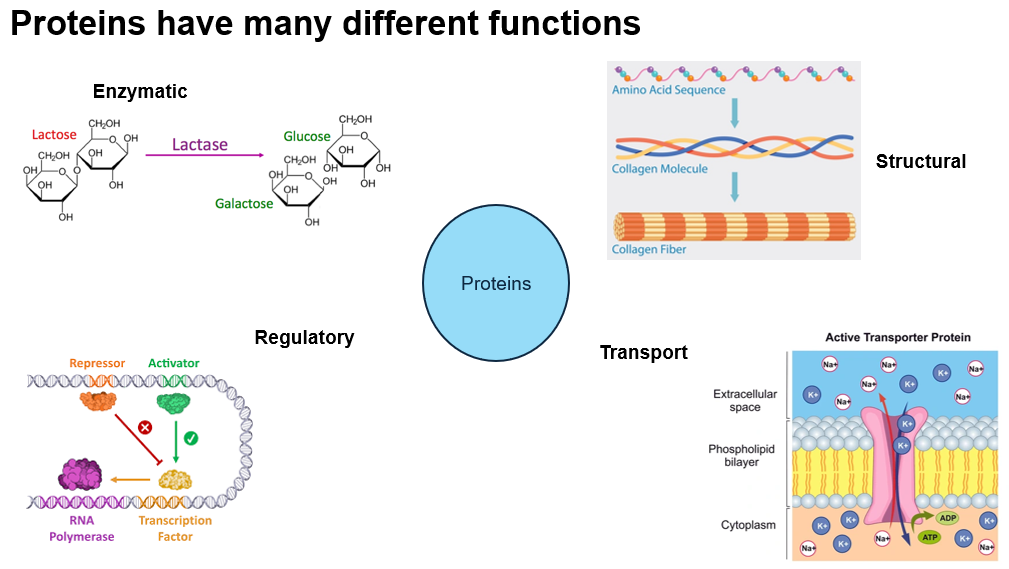
Proteins serve various functions, illustrated as follows:
Enzymatic: Catalyze biochemical reactions.
Example: Lactase enzyme breaking down lactose into glucose and galactose.
Regulatory: Involved in gene expression, such as proteins that act as repressor or activator in transcription.
Structural: Provide structural support, such as collagen molecules forming collagen fibers.
Transport: Facilitate the movement of ions and molecules across cell membranes, for example through active transport proteins.
Amino Acids: The Building Blocks of Proteins
Basics of Amino Acids
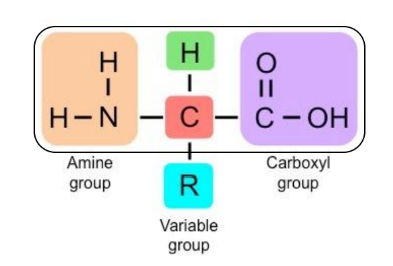
Amino acids (aa) are the monomers of proteins. The general structure consists of:
Carboxyl group: COOH.
Amine group: NH2.
Variable group (R): Determines the properties of the amino acid.
Classification of Amino Acids
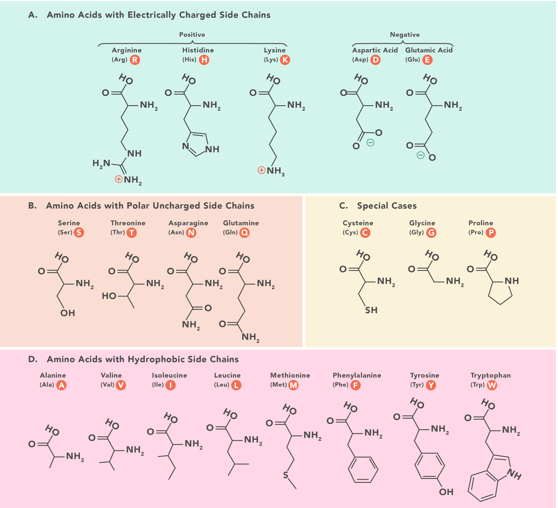
1. Amino Acids with Electrically Charged Side Chains:
Negative: Aspartic acid (Asp), Glutamic acid (Glu).
Positive: Arginine (Arg), Histidine (His), Lysine (Lys).
2. Amino Acids with Polar Uncharged Side Chains:
Asparagine (Asn), Serine (Ser), Threonine (Thr), Glutamine (Gln), Cysteine (Cys), Proline (Pro).
3. Amino Acids with Hydrophobic Side Chains:
Alanine (Ala), Valine (Val), Leucine (Leu), Isoleucine (Ile), Methionine (Met), Phenylalanine (Phe), Tyrosine (Tyr), Tryptophan (Trp).
Essential vs Non-Essential Amino Acids
Non-essential amino acids: can be synthesized by the body (e.g., Alanine, Serine, Proline).
Essential amino acids: must be obtained from the diet (e.g., Isoleucine, Methionine, Phenylalanine).

Structure of Proteins
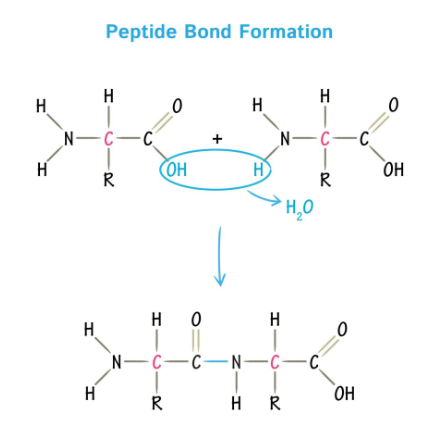
Peptide Bonds
Polypeptide chains are formed via peptide bonds, linking amino acids together.
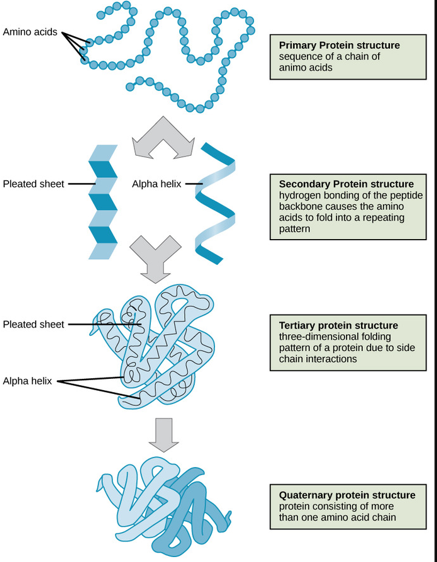
The sequence of amino acids in a protein represents its primary structure.
Protein Folding and Structure
Secondary Structure: Interaction with neighboring amino acids leads to folded structures such as alpha-helices and beta-pleated sheets.
Tertiary Structure: Interaction of secondary structures leads to the overall 3D conformation.
Quaternary Structure: Association of multiple polypeptide chains, potentially forming functional units called domains.
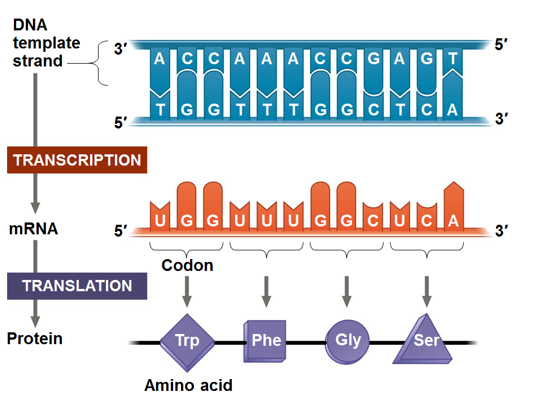
The Genetic Code
Codons
The basic unit of the genetic code is called a codon, with each codon comprised of three nucleotides (Triplets)
Established by Crick and colleagues in 1961, this triplet code outlines the relationship between nucleotides and amino acids.
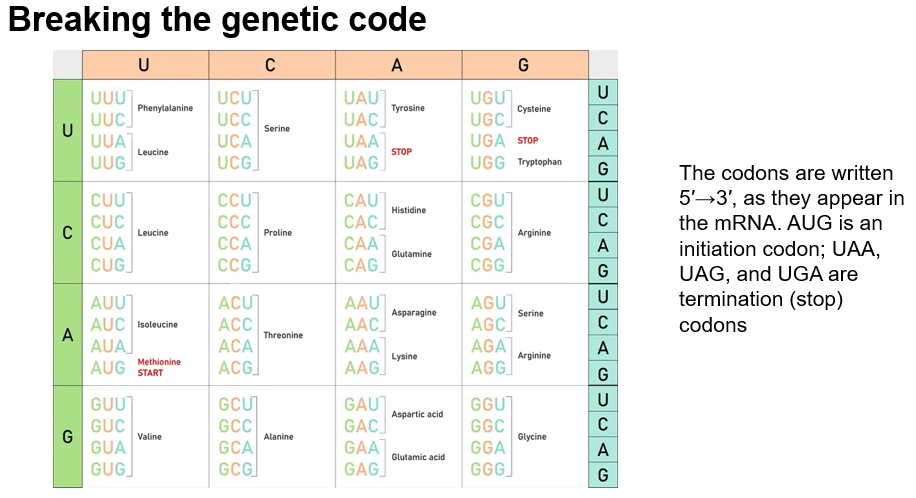
Breaking the Genetic Code
Total Codons: 64 possible codons are identified:
3 Stop Codons: UAA, UAG, UGA.
61 Sense(normal) Codons: Each coding for specific amino acids.
The genetic code is described as:
Degenerate: More than one codon may specify a particular amino acid.
Not Ambiguous: No codon specifies more than one amino acid.
Reading Frame: Codons must be read sequentially without overlap, starting from a designated initiation codon (AUG).
Universality of the Genetic Code
The genetic code is nearly universal across organisms
Genes cna be transcribed and translated after being transferred from one species to another
Classes of RNA in Protein Synthesis
Types of RNA
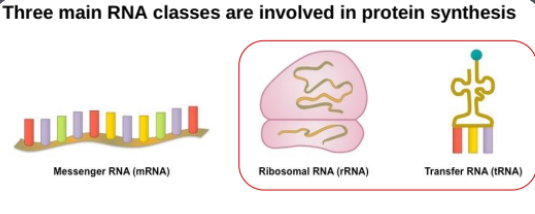
Three main classes involved in translating genetic information:
Messenger RNA (mRNA): Carries genetic information from the nucleus to the ribosome.
Ribosomal RNA (rRNA): Component of ribosomes, facilitating protein synthesis.
Transfer RNA (tRNA): Links amino acids to codons on the mRNA during translation.
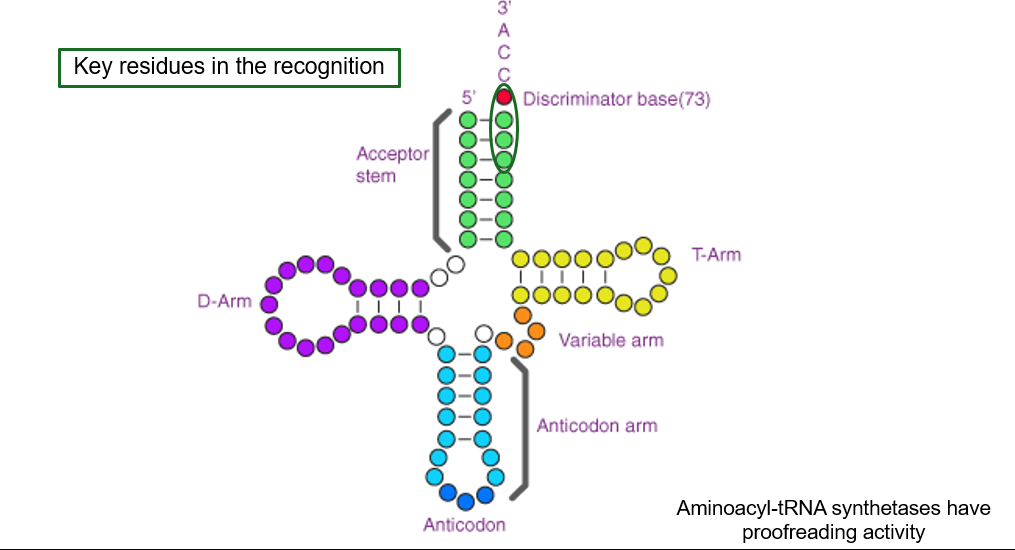
Transfer RNAs (tRNA)
Function and Structure
tRNA functions as the bridge between the genetic code and the amino acids in proteins.
Each tRNA carries a specific amino acid and binds to the correct codon on the mRNA through its anticodon.
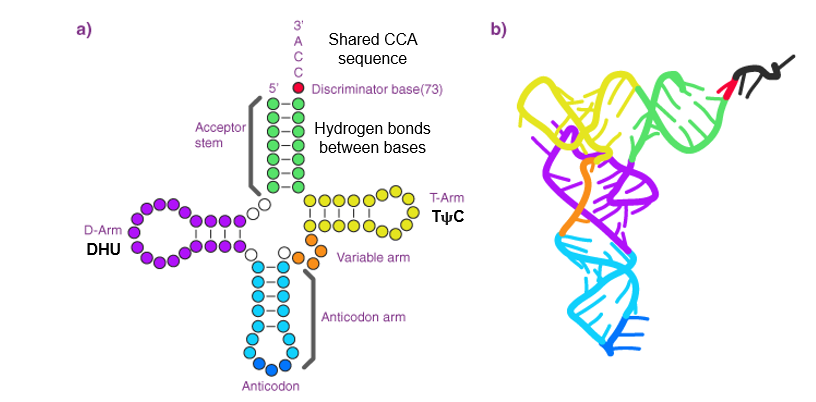
Structural Features:
74-95 nucleotides long, with a shared CCA sequence at 3' end.
Anticodon loop contains a three-base sequence complementary to the mRNA codon.
D-loop and T-arm assist enzyme recognition and ribosome binding.
Ribosomal RNA (rRNA)
Composition and Function
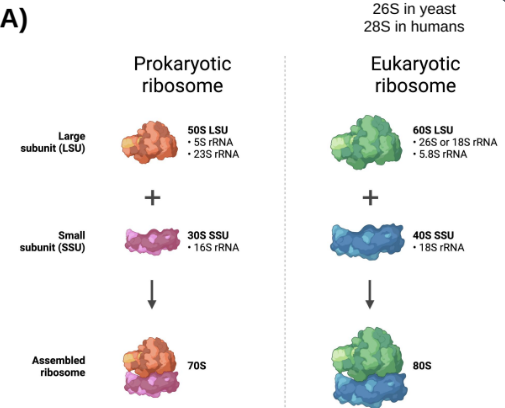
Ribosomes consist of over 50 RNA molecules and proteins, acting as the site for translation.
Abundant in cells (>20,000 ribosomes/cell), comprising 80% of the total RNA.
rRNA is categorized based on size (measured in Svedberg units).
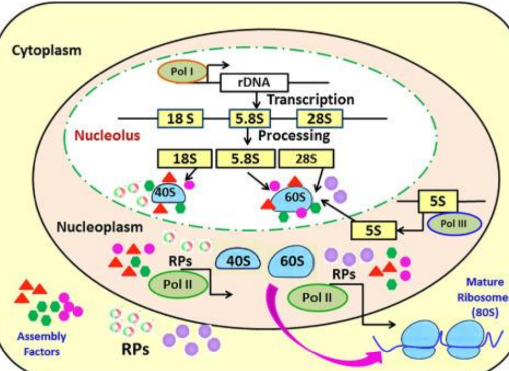
Eukaryotic Ribosomal RNA Genes
Multiple copies of rRNA genes are clustered, coding for:
Large rRNA (28S, 18S, 5.8S)
Small rRNA (5S)
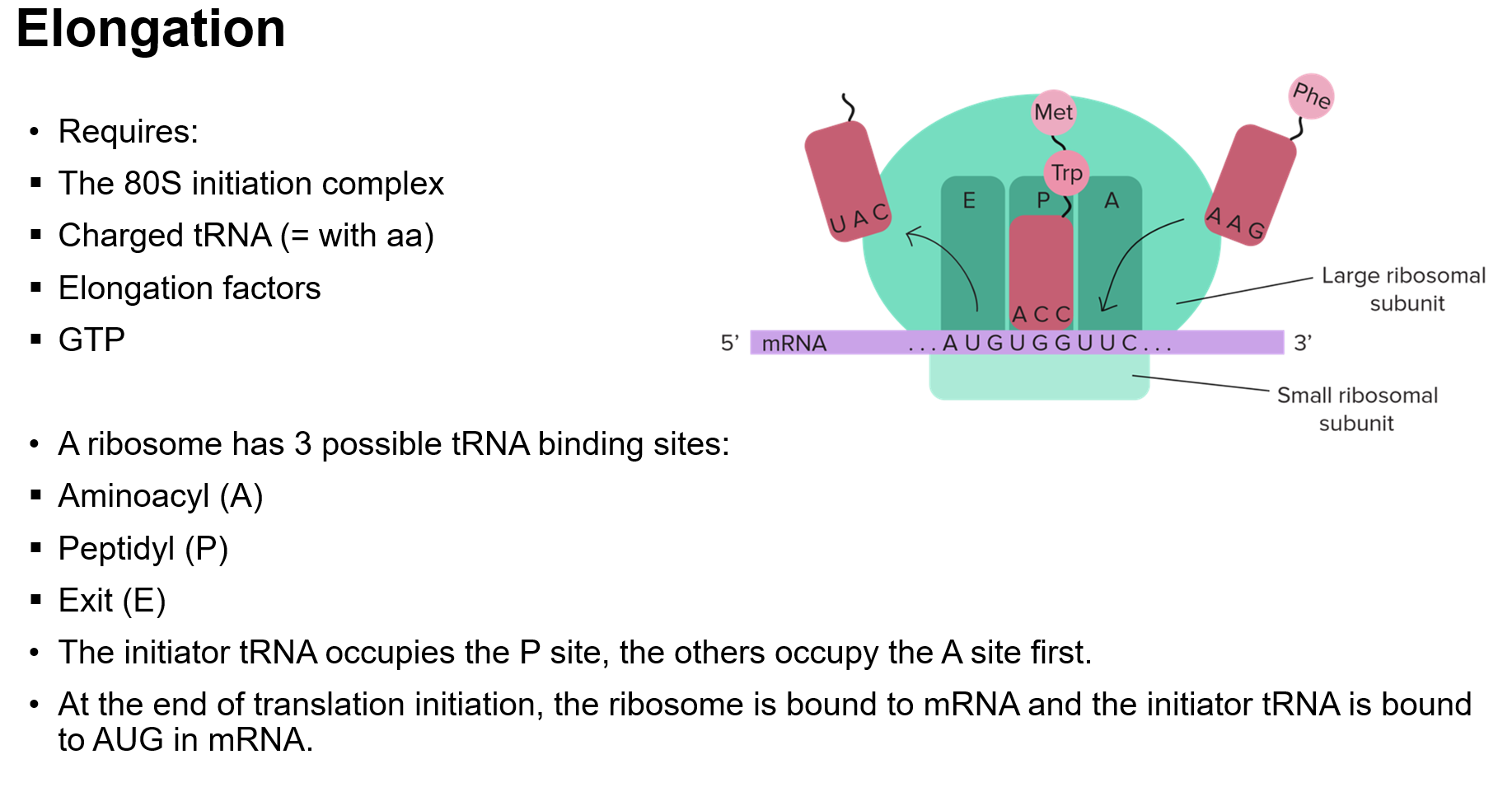
The Stages of Translation
When and Where Translation Occurs
What: mRNA is translated into proteins.
When: Translation occurs after mRNA is transported from the nucleus to the cytoplasm.
Where: Ribosomes, starting near the 5' end of mRNA.
Stages of Translation
tRNA Charging: Binding of tRNAs to their corresponding amino acids.
Initiation: Assembly of all necessary components at the ribosome.
Elongation: Addition of amino acids to the growing polypeptide chain.
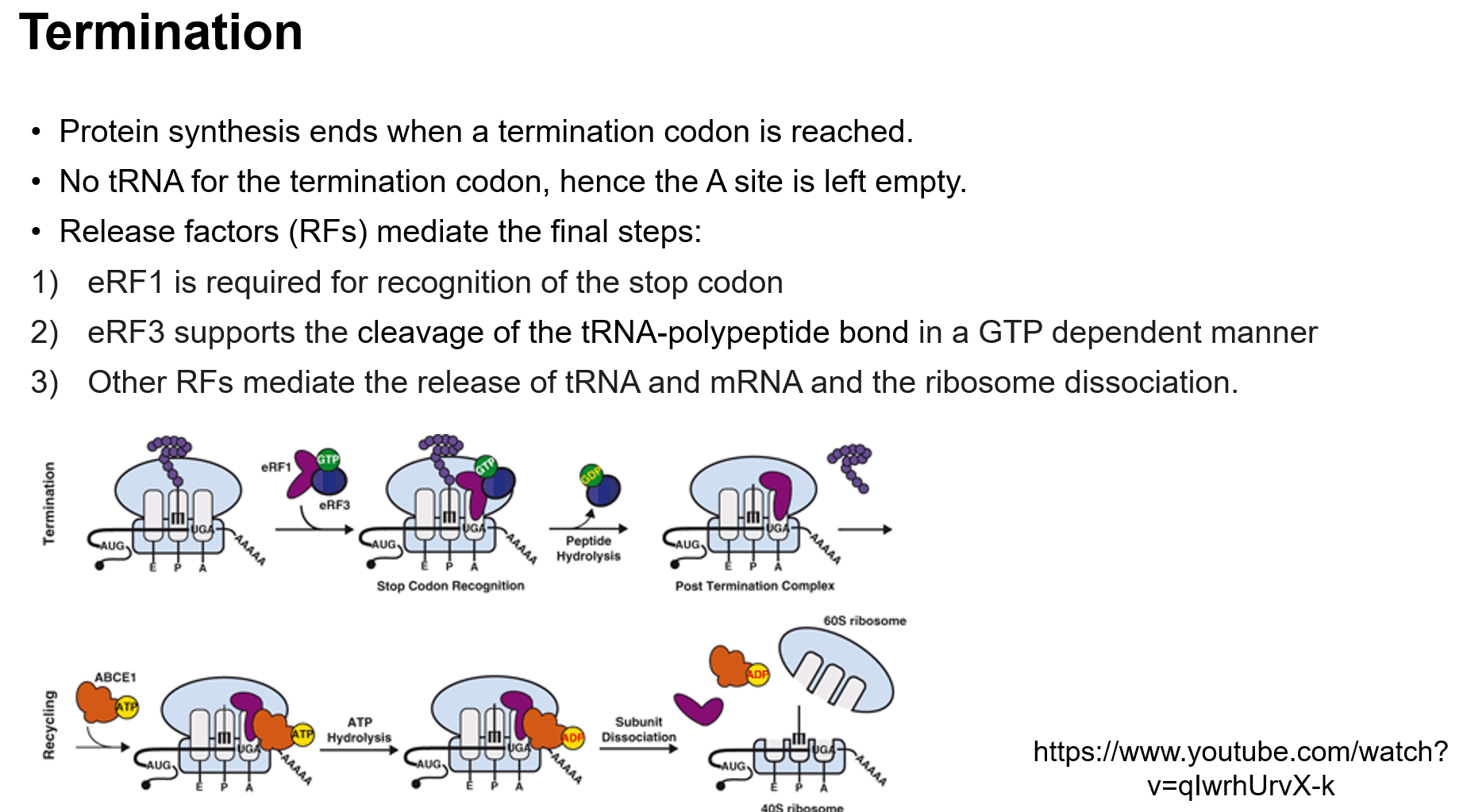
Termination: Completion of protein synthesis at stop codons.
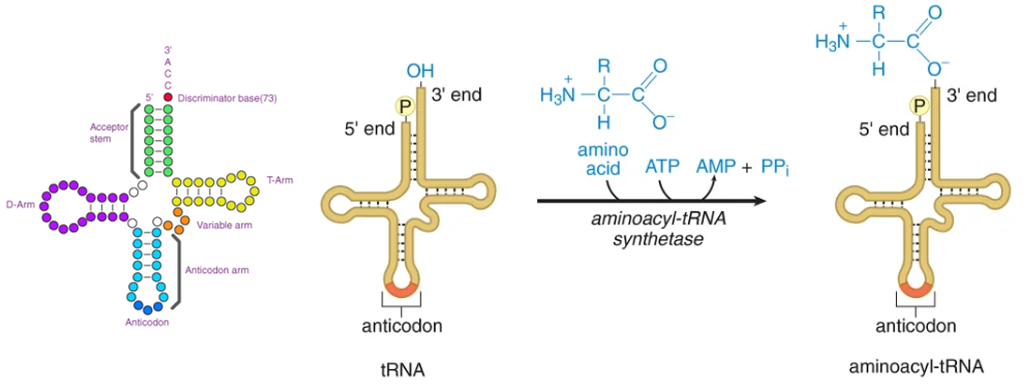
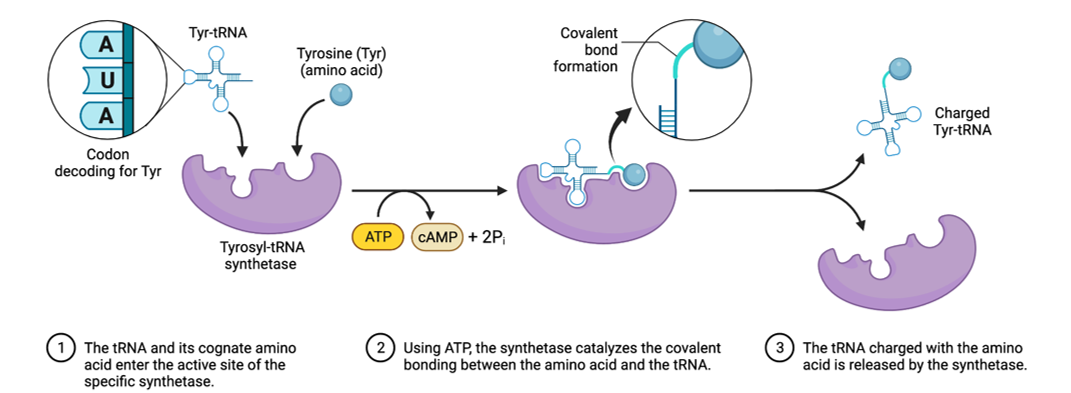
tRNA Charging
The CCA sequence is present in all tRNAs, with amino acids attached to the 3' end.
Specificity determined by aminoacyl-tRNA synthetases based on nucleotide sequences and properties of amino acids.
recognition of tRNA by the aminoacyl-tRNA synthesis is mediated by the nucleotide sequence
Recognition of amino acid by the aminoacyl-tRNAsynthesis is mediated by size, charge and R groups
Aminoacyl-tRNA synthesis (or aminoacylation) is the process of attaching an amino acid to its correct tRNA.
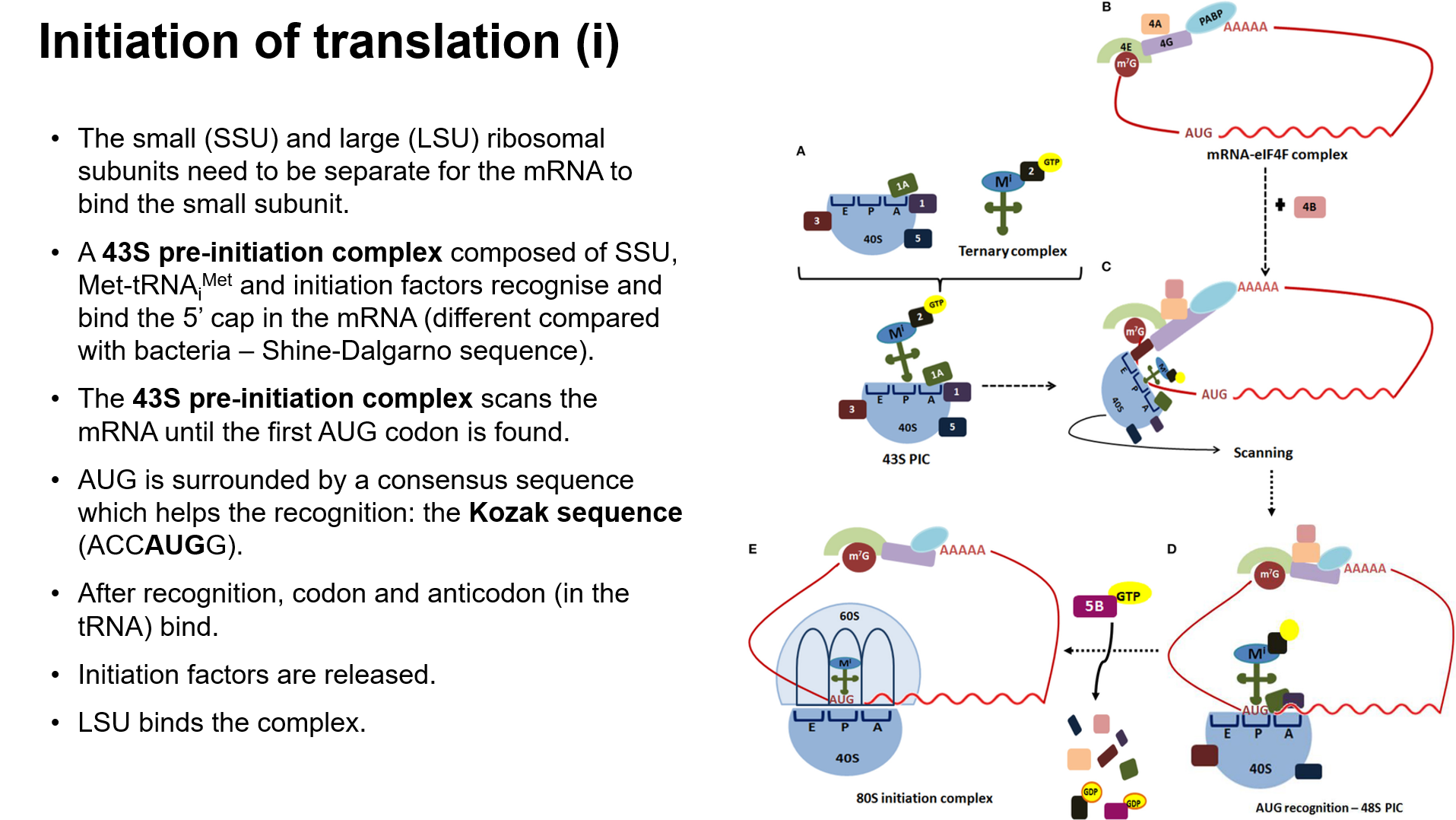
Initiation of Translation

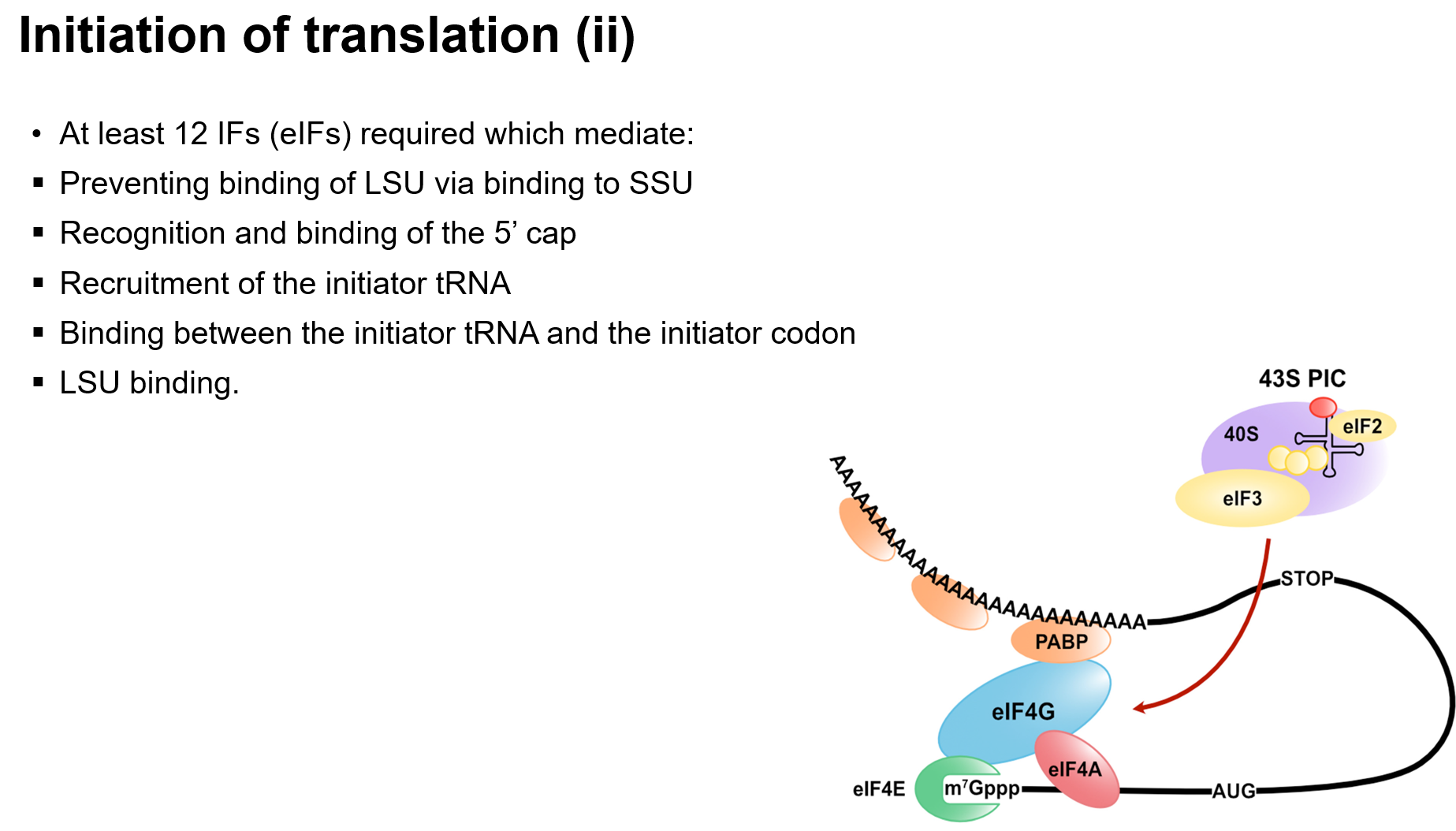
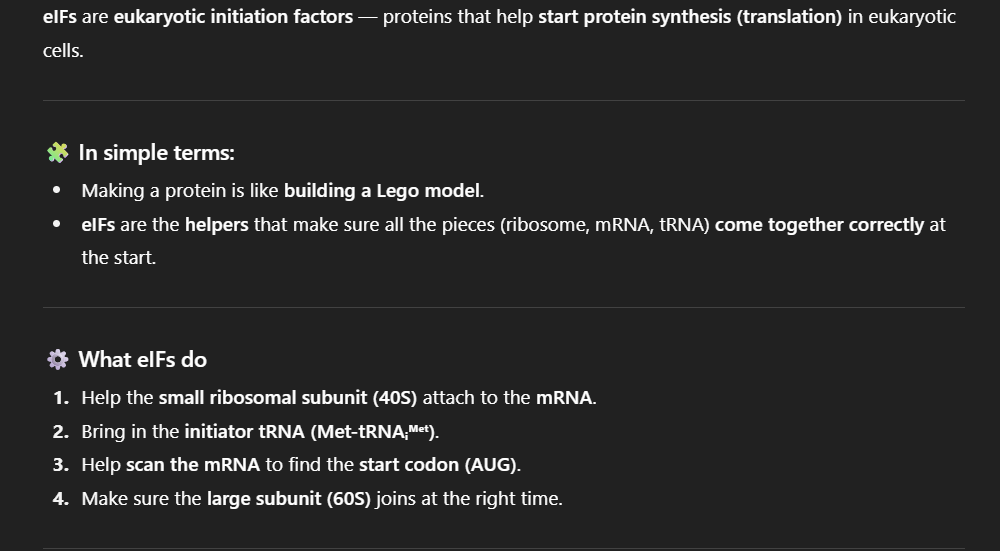
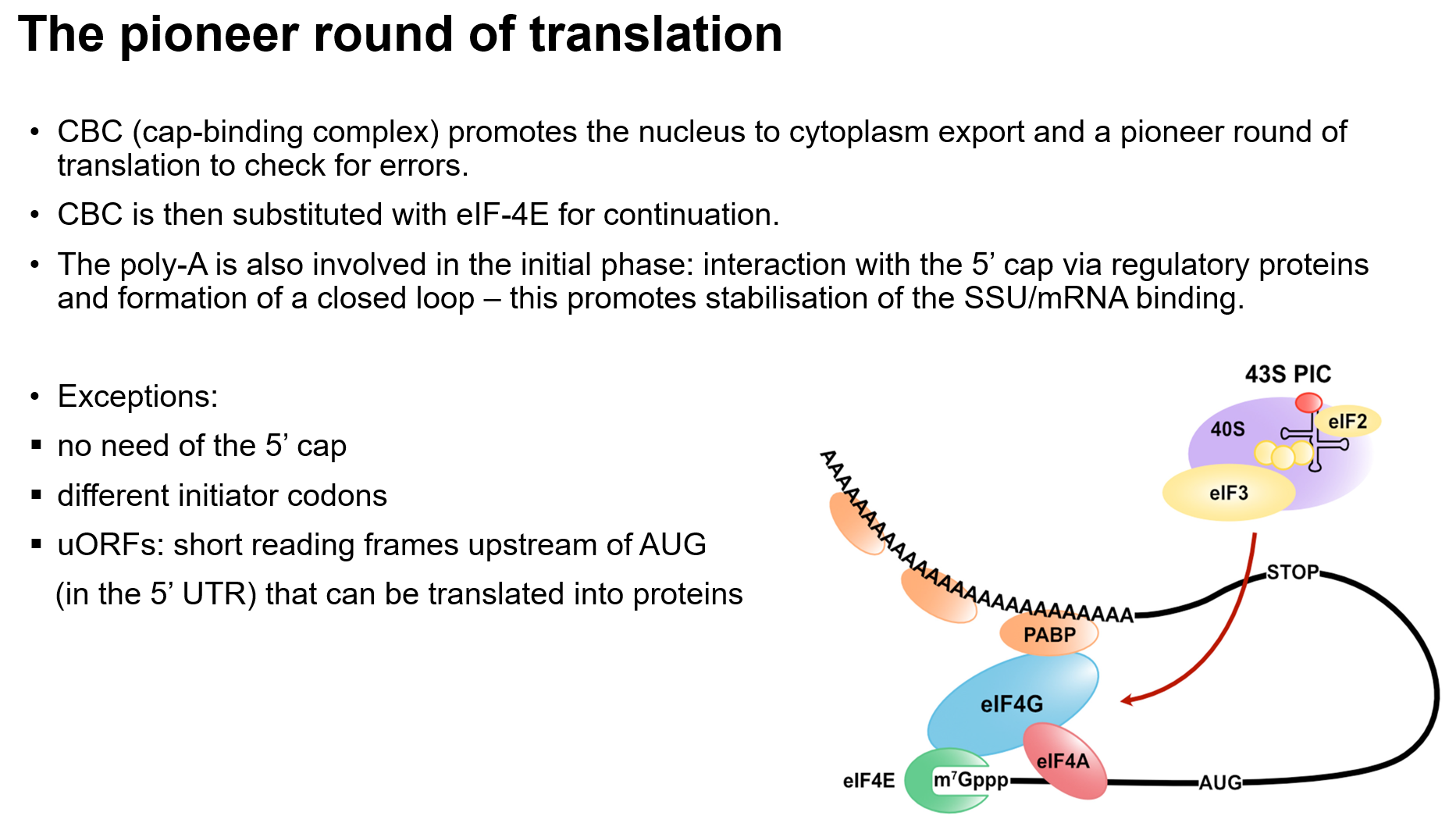
Binding of Components: mRNA, ribosomal subunits, initiation factors, and initiator tRNA.
Pre-initiation Complex (43S): Recognizes the 5' cap on mRNA and scans for AUG codon.
Kozak Sequence: Surrounding sequence that aids in recognizing the initiation codon.



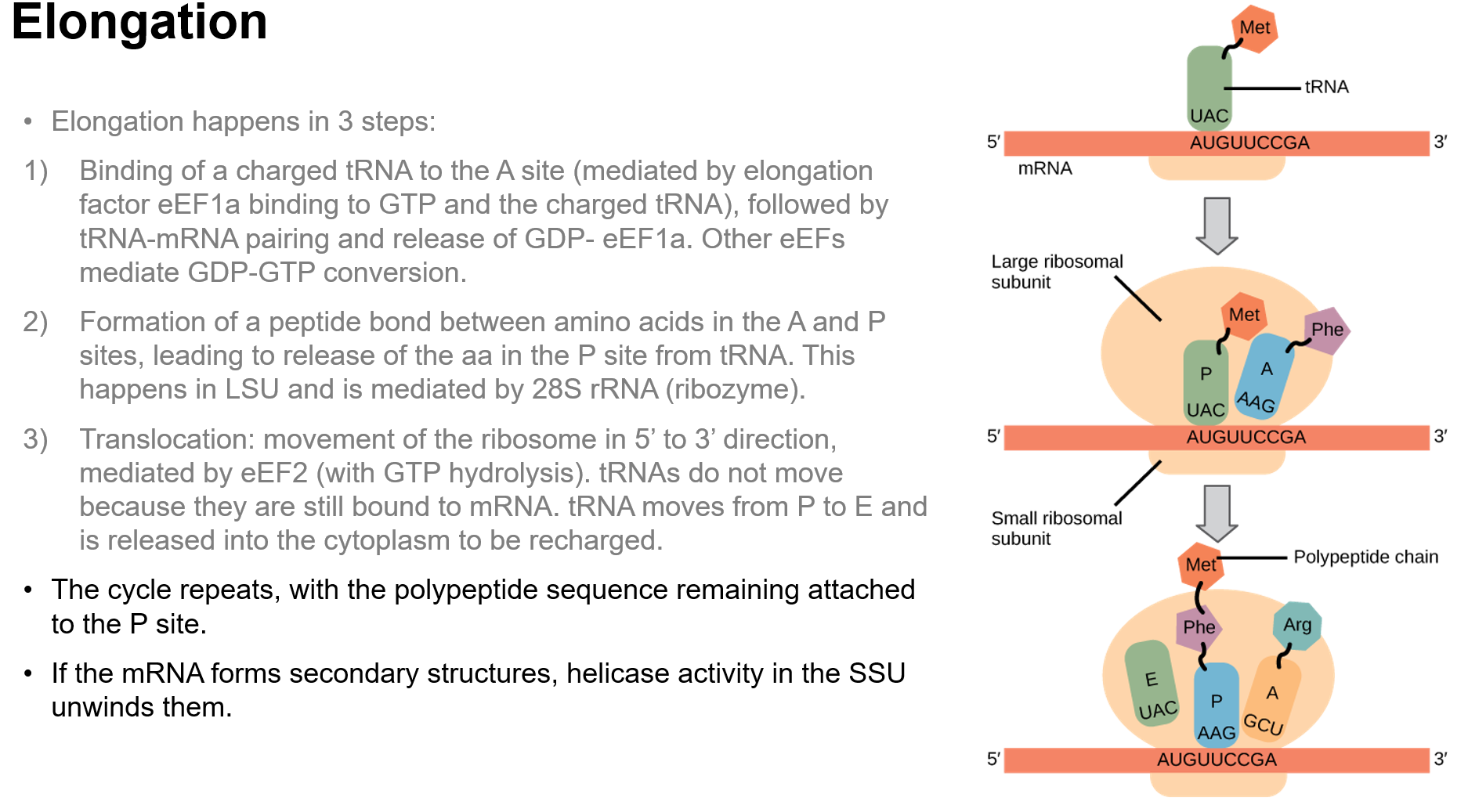
Elongation of the Polypeptide Chain
Involves binding of charged tRNA to A site, formation of peptide bonds mediated by rRNA, and movement of ribosome (translocation).
Termination of Translation
Occurs upon reaching a stop codon, with no corresponding tRNA, and facilitated by release factors.
Implications of Translation Defects
Protein synthesis errors can lead to diseases, including Diphtheria, caused by toxins that disrupt translation processes.
Research References on Diseases Related to Translation
Scheper et al., "Translation matters: protein synthesis defects in inherited disease", Nature Reviews Genetics, 2007.
Jia et al., "Protein translation: biological processes and therapeutic strategies for human diseases", Signal Transduction and Targeted Therapy, 2024.
Exceptions to the Central Dogma
Examples include prion diseases and mechanisms that subvert typical genetic information flow, highlighting unique biological phenomena.
Suggested Further Reading
Chapters 4-7 from course materials provide context on DNA, replication, gene expression, and protein translation.