4: Renal System's Role in Acid-Base Balance
Buffering and Compensation
Acid-base balance involves two main processes:
Buffering: Rapidly mops up excess hydrogen ions or releases them as needed.
It prevents rapid fluctuations in hydrogen ion levels.
Buffers are limited; they can only handle as much as their resources allow.
Example: Buffering excess hydrogen ions depends on available bicarbonate ions.
Buffering is a short-term solution.
Compensation: Removes excess acid or base from the body to reset and replenish buffers.
Buffering only masks the problem; the excess acid is still present and can dissociate.
Compensation is a longer-term approach.
Role of Lungs and Kidneys
Compensation involves the respiratory system (lungs) and the renal system (kidneys).
Lungs can adjust carbon dioxide levels.
Kidneys can modify bicarbonate and hydrogen ion levels.
Carbonic Acid-Bicarbonate Buffer System
The amount of carbonic acid in the blood relates to carbon dioxide, hydrogen ions, and bicarbonate ions, which are interchangeable.
The equilibrium in the system maintains a set point, which may not be equal amounts of each component but a preferred balance.
Changes in one component affect the others.
Restoring Balance
Disturbances, like excess hydrogen ions from lactic acid during anaerobic metabolism, disrupt equilibrium.
The body restores balance by adjusting carbonic acid levels or removing hydrogen ions.
Buffering involves combining hydrogen ions and bicarbonate to form carbonic acid, adjusting the seesaw effect to lower one end and raise the other.
The body constantly converts hydrogen ions to carbonic acid and carbonic acid to carbon dioxide, fluctuating to maintain balance.
Excess hydrogen ions from cellular metabolism push the system towards carbonic acid and carbon dioxide.
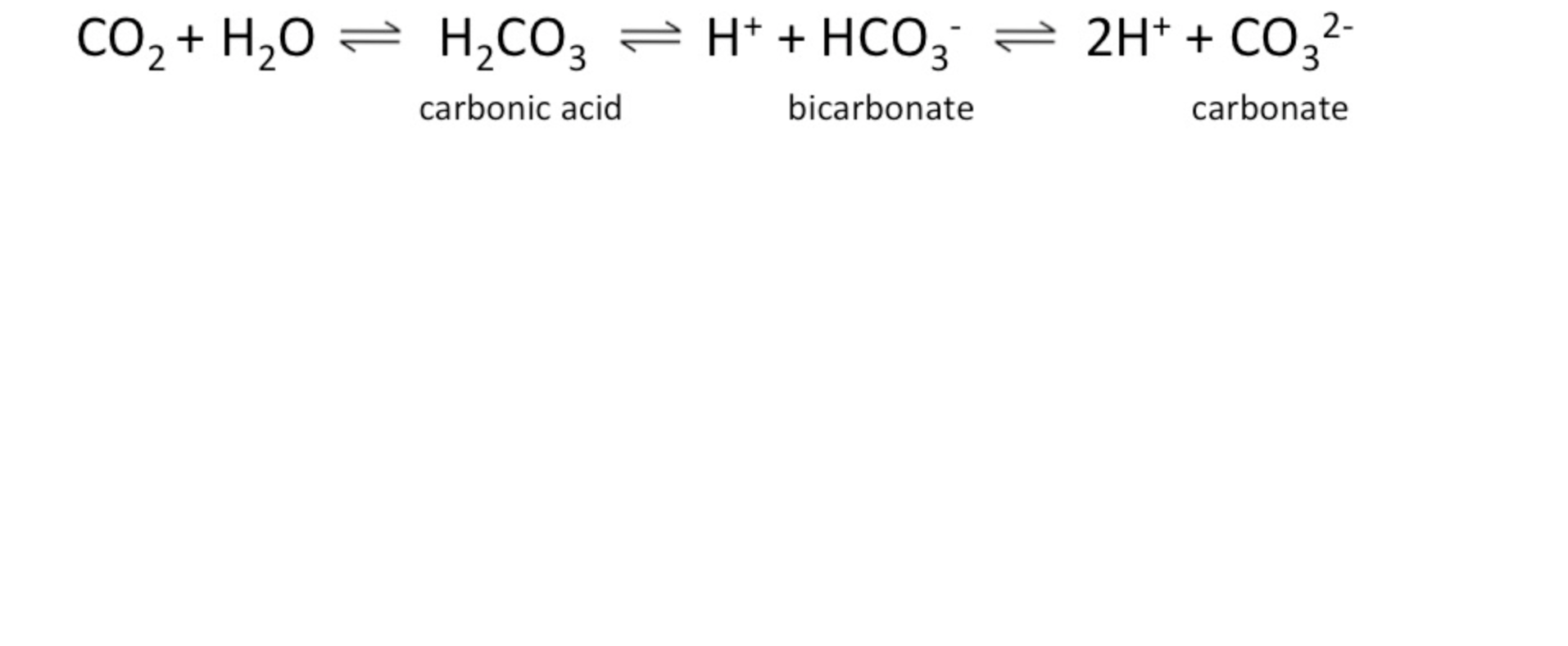
Respiratory Compensation
Increased carbon dioxide from excess hydrogen increases the partial pressure of carbon dioxide, the primary driver of respiration rate.
This increase leads to faster, deeper breathing to expel carbon dioxide, which reduces the partial pressure of carbon dioxide and restores respiratory rate to normal.
Eliminating carbon dioxide allows more carbonic acid to convert, providing scope to convert more hydrogen ions (requires bicarbonate).
Renal Compensation
Kidneys increase bicarbonate in the blood by producing and reabsorbing it.
Kidneys secrete more hydrogen ions, removing them from the body via urine.
Increased Hydrogen Ions | Decreased Hydrogen Ions |
= decreased pH = increased acidity | = increased pH = decreased acidity |
Buffering system engages immediately (however, this is limited and a short-term solution) | |
|
|
More carbonic acid forms from bicarbonate and hydrogen ions, decreasing the hydrogen ion concentration and bringing the pH back to normal. | More carbonic acid dissociates, raising hydrogen ion concentration and returning pH to normal. |
Respiratory Malfunction (Hypoventilation)
If the respiratory system malfunctions and ventilation decreases (hypoventilation), carbon dioxide elimination decreases, increasing the partial pressure of carbon dioxide.
The body attempts to increase the respiratory rate, but if the respiratory system cannot exchange carbon dioxide, it will not help.
Increased carbon dioxide increases carbonic acid and hydrogen ions, decreasing pH and increasing acidity.
Normal renal function compensates by increasing hydrogen ion secretion and increasing bicarbonate reabsorption and production, allowing greater buffering capacity and removal of hydrogen ion waste.
Renal Malfunction
If the renal system is not working well, it cannot secrete hydrogen ions, nor can it make and reabsorb bicarbonate.
This decreases the ability to buffer and impacts carbon dioxide levels, affecting pH if hydrogen ions cannot be removed.
Respiratory Compensation Details
Respiratory compensation occurs within minutes to hours, correcting blood pH changes by controlling carbon dioxide levels.
Changes in partial pressure of carbon dioxide trigger changes in breathing rate and depth.
As partial pressure of carbon dioxide increases, breathing becomes faster.
As partial pressure of carbon dioxide decreases, breathing becomes slower.
Renal Compensation Details
Renal compensation takes hours to days to correct pH and replenish buffers.
When pH is low, kidneys retain and reabsorb more bicarbonate and excrete excess hydrogen ions.
When pH is high, kidneys excrete bicarbonate and reduce hydrogen ion excretion.
Kidneys can also produce more bicarbonate when pH is low.
Plasma pH determines kidney response.
Key Consideration
When considering acid-base balance consider thinking in terms of blood pH, blood acidity or alkalinity, hydrogen ion concentration, respiratory rate, and partial pressure of carbon dioxide.