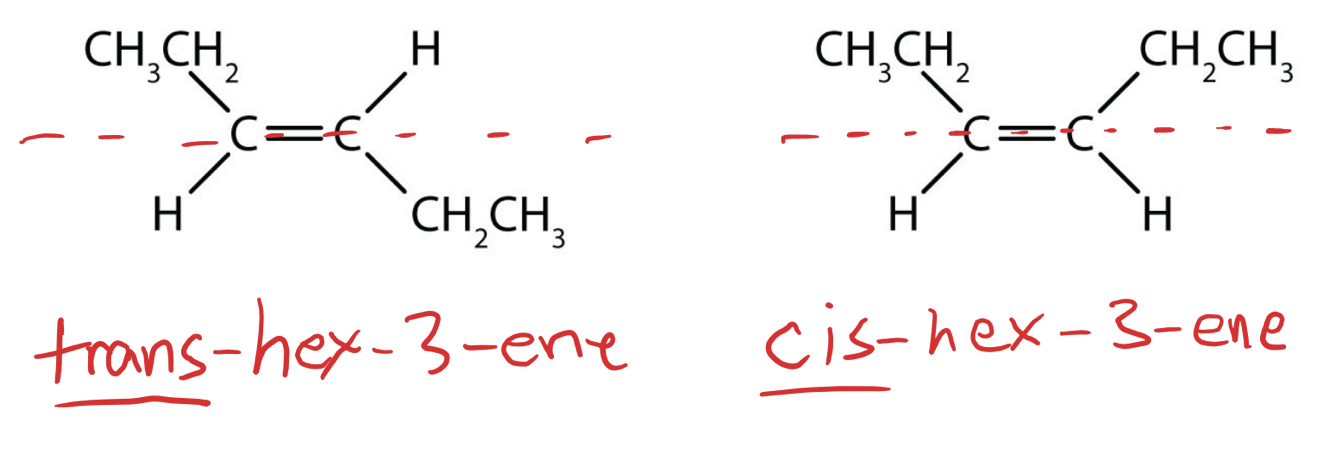1.2 Alkenes and Alkynes
- Unsaturated hydrocarbons: molecules in which at least one Carbon – Carbon bond is a double or triple bond.
- Hydrogen = syrup
- Carbon = pancake
- Saturated pancakes = alkanes
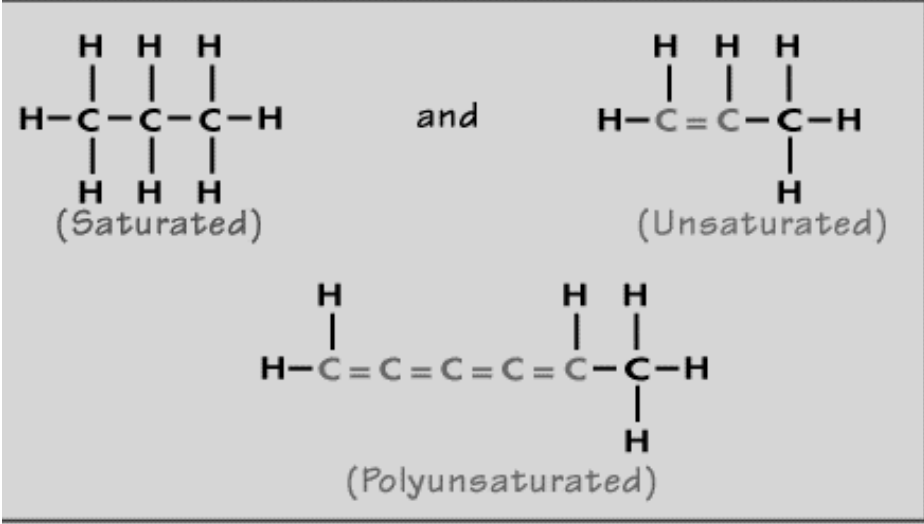
- Alkenes: hydrocarbons with at least one double bond between carbons and end with “one”
- CnH2n
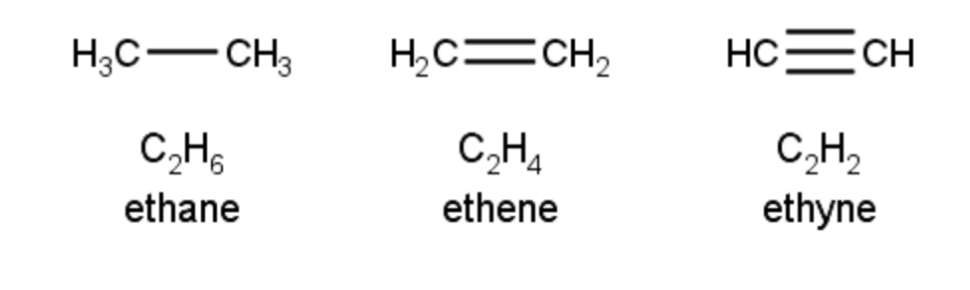
- Alkynes: hydrocarbons with at least one triple bond between carbons and end with “yne”
- CnH2n-2
Naming Alkenes and Alkynes
- Parent chain must include double / triple bond. Trumps the longest chain rule.
- A chain with multiple double/triple bond would go: octa-2,4,6-triene.
- Number chain with lowest number for substituent groups
- Name substituents as usual but indicate bond type and place of bond
- Note: you ignore the number of carbon only if the name will not change no matter where you draw the double/triple bond.
Stereoisomers
- same chemical formula and structural backbone but different arrangement of atoms in space
- Cis/Trans isomers are stereoisomers which occur only with alkenes
- Cis: the group of interest are located on the same plane of the double bond.
- Trans: the groups of interest are located on opposing planes of the double bond.