Unit 3: Cell energy
Slogan: Energy Efficiency
Critical Info
Thermodynamics
First law: Energy can be transferred and transformed, but energy CANNOT be created nor destroyed
Second law: Every energy transfer of transformation increases entropy (disorder) of the universe
Free Energy
Exergonic reactions (-ΔG): Release energy
“Export your ex cause of his negative energy“
Endergonic reactions (ΔG): Store energy
“Enter the store with a positive attitude”
Energy coupling: using an exergonic process to drive an endergonic one
Heterotrophs: need to find food/energy to consume
Enzymes
Acid-base catalysis: when covalent bonds in substrate may be destabilized when H ion transfer occurs between the active site of the enzyme and the substrate
Covalent Catalysis: when R groups interact and temporarily bond with the substrate
Metal ion Catalysis: when metal ion electrons are gained or lost without detaching from protein
Cofactors: chemicals which assist enzymic action
nonprotein enzyme helpers such as minerals (Zn, Fe, Cu)
Coenzymes: Organic cofactors
Vitamins
Regulation of enzyme activity ↴
Competitive: binds to the active site
competes for the bond with the enzyme against the substrate
Non-competitive: Binds to another part of the enzyme
alters the shape of the active site via another location
Cooperativity: A form of allosteric regulation which can amplify enzyme activity
Feedback inhibition: occurs when the end product of a metabolic pathway shuts down pathway by binding to the allosteric of an enzyme
this prevents wasting chemical resources, increase efficiency of the cell
Photosynthesis
Equation:
6CO2 + 6H2O + Light energy → C6H12O6 + 6O2
Chloroplasts are the site of photosynthesis
Light dependent reaction occur in the Thylakoids
Light-Independent reaction occur in the Stroma
Chemiosmosis: the movement of ions across a semipermeable membrane bound structure, down their electrochemical gradient
Osmosis for ions
Found in both Cellular Respiration and Photosynthesis
LEO (Loss of Electrons is Oxidation) the lion says GER (Gaining Electrons is Reduction)
Phosphorylation: the process of adding a phosphate group to a molecule
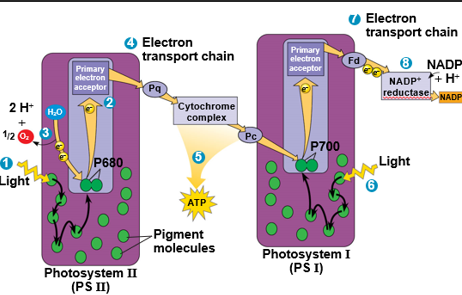
Light Reaction
Linear Electron Flow
A photon hits a light-harvesting complex of PS II
An excited electron is transferred to the primary electron acceptor
H2O is split by enzymes
H+ is released into thylakoid space
O2 is released as a byproduct
Each electron falls down an electron transport chain
this forms a proton gradient across the thylakoid membrane
Potential energy is stored in the proton gradient drives production of ATP by Chemiosmosis
In PS I, transferred light energy excites P700, which loses an electron to the primary electron acceptor
Each electron falls down another electron transport chain
NADP+ reductase catalyzes the transfer of electrons to NADP+, reducing it to NADPH
The electrons of NADPH are ready for the Calvin cycle
This process removes an H+ ion from the stroma
Cyclic Electron Flow
Same as linear flow except the electrons in photosystem I cycle back from Fd to the PS I reaction center (cytochrome complex) via a plastocyanin molecule
never go past Fd
Produces ATP but NOT NADPH
due to the fact that the electrons never make it to the NADP+ reductase
No oxygen is needed during this process
Calvin Cycle
Key points:
Creates:
1 G3P
9APD
6 NADP+
6 +pi
Carbon fixation evolution
TBF
C3 plants
C4 plants
CAM plants
Cellular Respiration
3 steps:
Glycolysis
“Sugar Splitting“
Glycolysis occurs in the cytosol outside the mitochondria
Oxidizes glucose into 2 Pyruvates
The process has net gains of:
2 ATP
2 NADH
2H2O
Note that glycolysis does NOT require O2 as it is a theorized ancient process
Glycolysis also must first invest 2 ATP before it has a payoff of 4 ATP resulting in the net of 2 ATP
Pyruvate Oxidation + Citric acid cycle (Krebs)
Pyruvate is converted to Acetyl CoA to be used for the Citric Acid cycle (Krebs)
There is a Net gain of:
2 ATP
6NADH
2FADH
Oxidative Phosphorylation (ETC and chemiosmosis)
This part uses an ETC.
PRODUCES RHE BULK OF ATP (26-28)
same concept with ETC and H+ ions.
the electrons move H+ ions against the gradient, the ATP synthase then allows H+ ions to diffuse back, creating ATP
Kahoot Questions
(Will be plugged into the notes)
What BEST describes the type of metabolic pathway of photosynthesis?
Endergonic (catabolic)
What BEST describes the type of metabolic pathway of Cellular respiration?
Exergonic (anabolic)
In respiration, O2 is the final electron (e-) acceptor in the ETX. Where for these e- come from?
NADH & FADH2
In respiration, which would be evidence of cellular respiration NOT occurring?
An increase in lactic acid concentration
Both Krebs cycle and glycolysis make a net gain of 2 ATP
Which of the following is considered substrate level phosphorylation?
Both Krebs and Glycolysis
Which of the following is involved in Oxidative Phosphorylation?
Electron Transport chain
On hot, dry days, what do C3 plants do?
Partially close their stomata
How can you measure the rate of photosynthesis? Measuring the production of O2
The “extra“ pigments in plants are called an accessory
Plants imbed pigments in what structures to be used in photosynthesis?
Thylakoid membranes
Essential Questions
How do enzymes affect the rate of biological reactions?
What environmental factors can influence the rate of reaction by impacting the structure and function of enzymes?
How do environmental conditions impact enzyme function?
What processes are shared (conserved) throughout both photosynthesis and cellular respiration?
How do membranes in the chloroplast and mitochondria contribute to their function?
How do electron transport chains create hydrogen gradients?
How do hydrogen gradients create ATP?