L4 - Dissolution and Precipitation
Solution and Precipitation Reactions
Introduction: Salt in Water Experiment
Simple experiment: Adding salt (NaCl) to water.
Observation: Salt disappears, forming a solution.
Saturation: Point where no more salt dissolves.
Representation
Plotting concentration of Na^+ or Cl^- ions vs. time.
Two phases:
Dissolution: Salt dissociates into ions.
Saturation: Equilibrium between solid salt and ions.
Dissolution Reaction
NaCl(s) \rightarrow Na^+(aq) + Cl^-(aq)
aq indicates aqueous species.
Saturation
Equilibrium between solid NaCl and Na^+ and Cl^- ions in solution.
NaCl(s) \rightleftharpoons Na^+(aq) + Cl^-(aq)
Solubility
Concentration of substance in solution at saturation.
Function of pressure and temperature.
Formal Representation of Solid Dissolution
Solid composed of p atoms of cation B and n atoms of anion A:
B_pA_n
Dissolution (non-equilibrium):
B_pA_n(s) \rightarrow pB^{n+}(aq) + nA^{p-}(aq)
At equilibrium:
B_pA_n(s) \rightleftharpoons pB^{n+}(aq) + nA^{p-}(aq)
Solubility Definition
Concentration of cation or anion at saturation, normalized by stoichiometric coefficient.
Units: mol/L or mol/kg.
Solubility Product (Ksp)
Equilibrium constant for dissolution equilibrium.
Law of Mass Action:
K_{sp} = \frac{a_{B^{n+}}^p a_{A^{p-}}^n}{a_{B_pA_n}}=a^p_{B^{n+}(eq)} \cdot a^n_{a^{p-}(eq)} because the activity of a pure solid is 1
K_{sp} is a product of ion activities at saturation and is called the solubility product.
Temperature and pressure dependent.
Ion Activity Product (IAP)
Reaction quotient (Q) for dissolution reaction.
Same expression as K_{sp}, but not necessarily at equilibrium.
Example: Calcium Carbonate Dissolution
CaCO_3(s) \rightleftharpoons Ca^{2+}(aq) + CO_3^{2-}(aq)
IAP expression:
IAP = a_{Ca^{2+}} \cdot a_{CO_3^{2-}}
Saturation Index (SI)
Measure of saturation state.
SI = log(\frac{IAP}{K_{sp}})
SI = 0: Solution is saturated (equilibrium).
SI > 0: Solution is supersaturated (precipitation).
SI < 0: Solution is undersaturated (dissolution).
Example Problem: Calcium Sulfate (Anhydrite)
Given: Concentrations of Ca^{2+} and SO_4^{2-}, K_{sp} of anhydrite.
[Ca^{2+}] = 5 \cdot 10^{-2} \text M
[SO_4^{2-}] = 7 \cdot 10^{-3} \text{M}
K_{sp(anhydrite)}=10^{-4.59}
Question 1: Is the solution supersaturated?
Question 2: How much mineral precipitates to reach equilibrium?
Assumptions
Activity coefficients = 1 (dilute solutions).
Solution


Kinetics vs. Thermodynamics
Thermodynamics predicts stable state (minimum Gibbs free energy), but the actual dissolution or precipitation in the appropriate saturation will depend on kinetic factors.
Seawater: Supersaturated with CaCO_3, but slow precipitation. The formation of CaCO_3 is kinetically inhibited.
Calcite in pure water: Undersaturated, but slow dissolution.
Metastability
System in non-equilibrium state due to slow kinetics.
Several supersaturated mineral phases may exist.
Unstable precursor phase: Precipitates faster, but not thermodynamically stable.
Example: calcite often precipitates via an amorphous calcium carbonate (ACC) precursor that is metastable.
Biomineralization: Organisms use metastable precursors for skeleton formation.
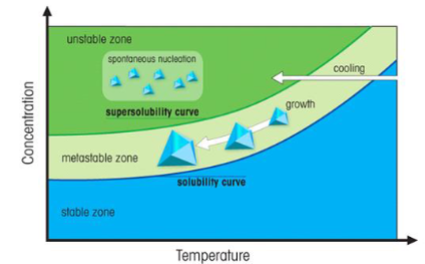
Metastable Zone
Solution supersaturated, but no precipitation due to slow reaction.
Homogeneous (spontaneous) nucleation: Direct precipitation in solution.
Heterogeneous nucleation: Precipitation on a seed crystal (lower activation energy).
Stability Diagrams
Predict mineral precipitation based on solution composition.
Two-dimensional representation of stability fields.
Example: Magnesium-containing Solution
Minerals: Brucite (Mg(OH)_2) and Magnesite (MgCO_3).
Axes: pH and log[Mg^{2+}].
Steps to Build Diagram
Write dissolution reactions.
Express [Mg^{2+}] as a function of pH.
Brucite Stability
Mg(OH)_2(s) \rightleftharpoons Mg^{2+}(aq) + 2OH^-(aq)
K_{sp,Bru} = [Mg^{2+}][OH^-]^2=10^{-11.6}
[Mg^{2+}] = \frac{K_{sp,Bru}}{[OH^-]^2}
Using K_w = [H^+][OH^-], [OH^-] = \frac{K_w}{[H^+]}:
[Mg^{2+}] = \frac{K_{sp,Bru}[H^+]^2}{Kw^2}
log[Mg^{2+}] = log(K_{sp,Bru}) + 2log[H^+] - 2log(K_w)
log[Mg^{2+}] = 16.4 - 2pH
Magnesite Stability
MgCO_3(s) \rightleftharpoons Mg^{2+}(aq) + CO_3^{2-}(aq)
K_{sp,Mag}=[Mg^{2+}][CO_3^{2-}]=10^{-7.5}
Carbonate system considerations (DIC, pH).
Three pH domains:
High pH (pH > pK_{a_2}): [CO_3^{2-}] \approx DIC
Intermediate pH (pK_{a_1} < pH < pK_{a_2}): [HCO_3^-] \approx DIC
Low pH (pH < pK_{a_1}): [H_2CO_3] \approx DIC
Using appropriate carbonate equilibrium constants to relate [CO_3^{2-}] to DIC and pH.
High pH
K_{sp,Mag}=\left\lbrack Mg^{2+}\right\rbrack\left\lbrack CO_{3^{}}^{2-}\right\rbrack=\left\lbrack Mg^{2+}\right\rbrack\sum CO_2
\log\left\lbrack Mg^{2+}\right\rbrack=\log K_{sp,Mag}-\log\sum CO_2=-pK_{sp,Mag}+p\sum CO_2
log[Mg^{2+}] = log(\frac{K_{sp}}{DIC})
log[Mg^{2+}] = -5
Horizontal line.
Intermediate pH
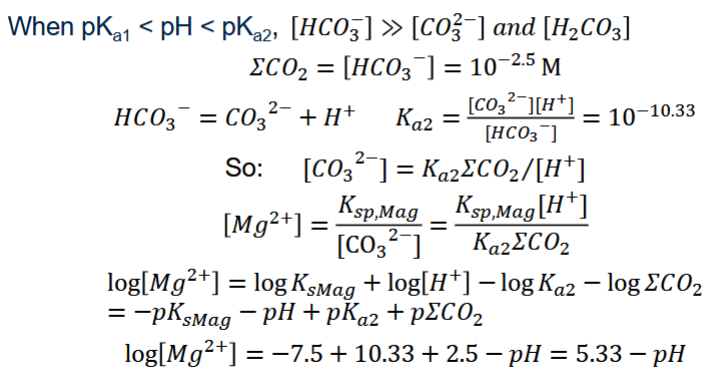
Low pH
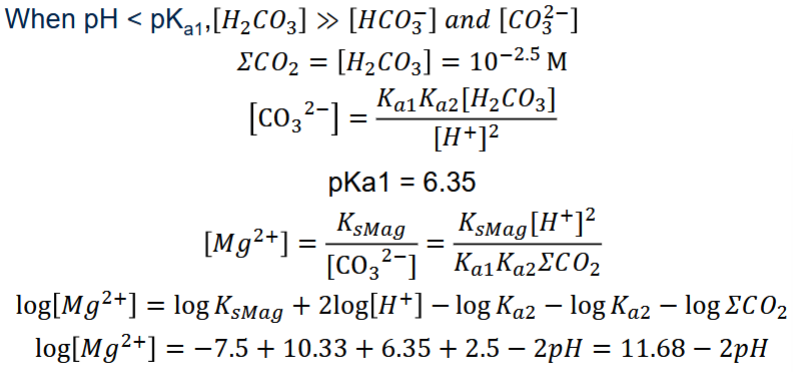
Overlapping Stability Fields
Stable mineral is the one with lowest solubility (lowest line on diagram).
Applications of Stability Diagrams
Predicting mineral dissolution/precipitation in natural waters.
Weathering processes.
Impact of CO_2$$ on Ocean Acidification and the effect on production of calcium carbonate skeletal material by some marine organisms. Increasing acidity reduces the saturation index of calcium carbonate.