unit 6: ionic and covalent compounds
Chemical Bonds:
a mutual attraction between the valence electrons of one atom and the protons in the nucleus of another atom
Ionic Bonding:
formed between atoms of metals and nonmetals
valence electrons are completely transferred from the metal atom to the non-metal atom
between atoms in ionic bonding, there is electrostatic attraction
there is a positive sphere of charge and a negative sphere of charge
Lewis Structures:
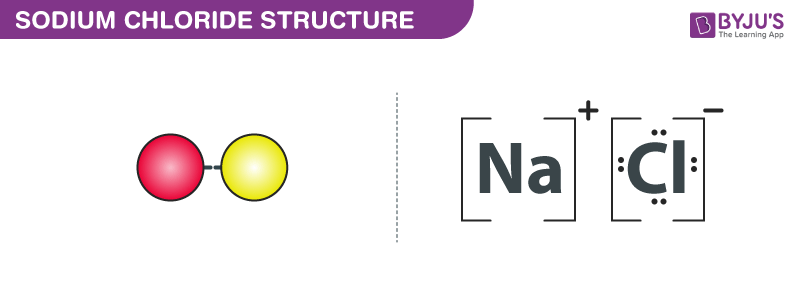
metal electron transferred to nonmetal
metal = positive
nonmetal = negative
completes octet!
Covalent Bonding:
formed between atoms of nonmetals with other nonmetals
valence electrons are shared in pairs between bonded atoms
pure covalent bonds [non polar covalent] ONLY occur between two identical atoms
BRINCLHOF!! - seven diatomic elements
even distribution of the bonding electron cloud
Polar Covalent Bonding
electrons are shared but not equally
many compounds have both characteristics of covalent and ionic bonding
electronegativity differences determine the balance of the character
the separation of charge …
the more electronegative element = a partial negative charge
the less electronegative element = a partial positive charge
dipole arrow (shows separation of charge in the molecule)
POSITIVE to NEGATIVE
Lewis Structure:
SHARES electrons

Metallic Bonding
valence electrons are shared among all the atoms of the substance
occurs when metals are bonded to themselves or mixed with other metals (ALLOYS)
Alloys & Examples
mixture of a metal with other metals or nonmetals
examples:
brass (copper and zinc)
bronze (copper and tin)
carbon steel (iron and carbon)
solder (lead and antimony)
stainless steel (iron, chromium, and nickel)
Formulas
differences in electronegativities helps us classify bonds
Ionic: EN >= 1.7
Polar Covalent: 1.6 >= EN >= 0.5
Nonpolar Covalent: EN < 0.5
electronegativity:
defined as the ability of an atom in a covalent bond to attract the bonding electron cloud to itself
trend: increases across periods; decreases down groups