BIOC*2580 - 3
Overview of Carbohydrates and Isomerism
Discussed the types of sugars known as carbohydrates, focusing mainly on monosaccharides.
I. Monosaccharides and Isomers
A. Types of Isomers in Monosaccharides
Enantiomers:
Defined as mirror images of one another.
Diastereomers:
Defined as two structures (in this case, sugars) that have the same chirality at some carbons but different chirality at others.
B. Epimers
Definition: A type of diastereomer where two sugars differ from each other in configuration at exactly one chiral carbon atom.
Chirality Explanation:
If two structures have identical configurations except for one chiral carbon, they are referred to as epimers of one another.
Example:
In the case of sugars, we can see how the configurations around chiral carbon atoms differ, leading to the conclusion that they are epimers.
C. Relationship Between Isomers
All epimers are diastereomers.
If the difference in configuration occurs at more than one chiral carbon, then they belong to the larger category of diastereomers.
Practical Importance: Understanding these terms is crucial for studying enzyme action, specifically epimerases, which convert one epimer to another.
II. Identifying Monosaccharide Relationships
A. Aldoses and Ketoses
Discussed two sugar structures and distinguished them as either aldoses (aldehyde sugars) or ketoses (ketone sugars).
Process of Identification:
Evaluation of all chiral carbons to determine if they are mirror images to classify as enantiomers.
B. Example Sugar Structures
Identified two structures as D and L sugars based on configurations of chiral carbons relative to the carbonyl.
Described how the position of hydroxyl groups is crucial in determining the classification as D or L sugars:
D sugars: Hydroxyl on the right side
L sugars: Hydroxyl on the left side
III. Total Number of Stereoisomers
A. Formula for Stereoisomers
The total number of stereoisomers for any sugar structure can be calculated with the formula:
2^n where (n) is the number of chiral centers.Example Cases:
Glyceraldehyde (1 chiral center): (2^1 = 2) stereoisomers (D-glyceraldehyde and L-glyceraldehyde).
A five-carbon aldotriose (3 chiral centers): (2^3 = 8) stereoisomers—4 D sugars and 4 L sugars.
B. Calculation Example
For a generic structure with 4 chiral centers, the total number of possible stereoisomers would be:
X = 2^4 = 16
Number of D sugars Y = 8
IV. Common D Ketoses to Aldoses
A. Relationship of Carbon Chain Length to Stereoisomer Number
The number of possible isomers generally doubles with the addition of carbon atoms:
3 Carbons (Triose): 1 D sugar
4 Carbons (Tetrose): 2 D sugars
5 Carbons (Pentose): 4 D sugars
6 Carbons (Hexose): 8 D sugars
B. Pentoses and Hexoses Differentiation
Explained that aldoses may have an additional chiral center compared to ketoses, thereby increasing the total number of stereoisomers in aldoses with equal chain length.
V. Intramolecular Reactions of Sugars
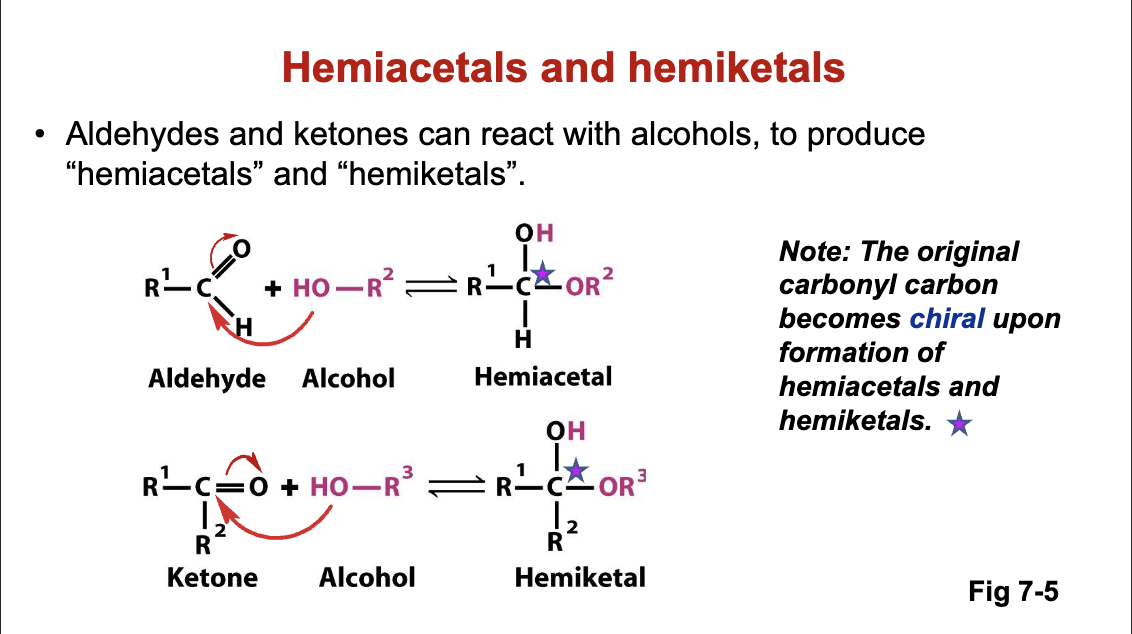
A. Hemiacetal and Hemiketal Formation
Describes how linear forms of aldoses and ketoses can react via condensation with alcohols to form hemiacetals and hemiketals.
Linear Formation Chemistry:
Aldehyde + Alcohol ——> Hemiacetal
Ketone + Alcohol ——> Hemiketal
B. Hemiacetal and Hemiketal Characteristics
The carbonyl carbon in the hemiacetal/hemiketal formation becomes a chiral center post-reaction.
All sugars (considering linear forms) possess carbonyl groups which react to form new cyclic structures.
D. Haworth Projections
Definition: A common way to represent the cyclic structure of monosaccharides, particularly furanoses (5-membered rings) and pyranoses (6-membered rings).
In a Haworth projection:
The ring is drawn as a flat hexagon or pentagon, with the oxygen atom usually in the back right.
Substituents (hydroxyl groups, hydrogen atoms) on carbon atoms above the plane of the ring are typically shown above the ring, and those below the plane are shown below the ring.
The anomeric carbon's orientation determines the alpha or beta anomer:
Alpha Anomer: The hydroxyl group on the anomeric carbon is trans to the CH2OHCH2OH group at the highest numbered chiral center (typically pointing down in D-sugars).
Beta Anomer: The hydroxyl group on the anomeric carbon is cis to the CH2OHCH2OH group at the highest numbered chiral center (typically pointing up in D-sugars).
C. Example of Glucose Cyclization
Describes how glucose (an aldose) reacts with its hydroxyl groups to form a ring structure and produce two forms: alpha-glucose and beta-glucose (anomers).
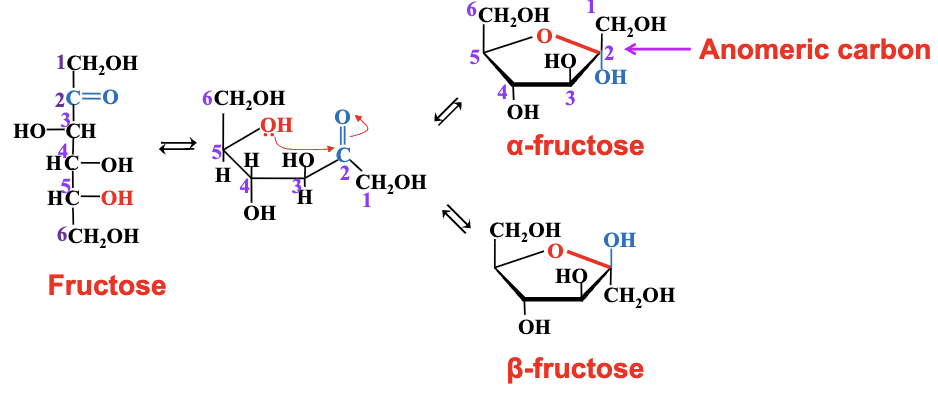
the hemiacetal or hemiketal formation is intramolecular: the alcohol and the aldehyde (or ketone) are present in the same molecule.
Hemiacetal and hemiketal formation turns sugars into ring
structures.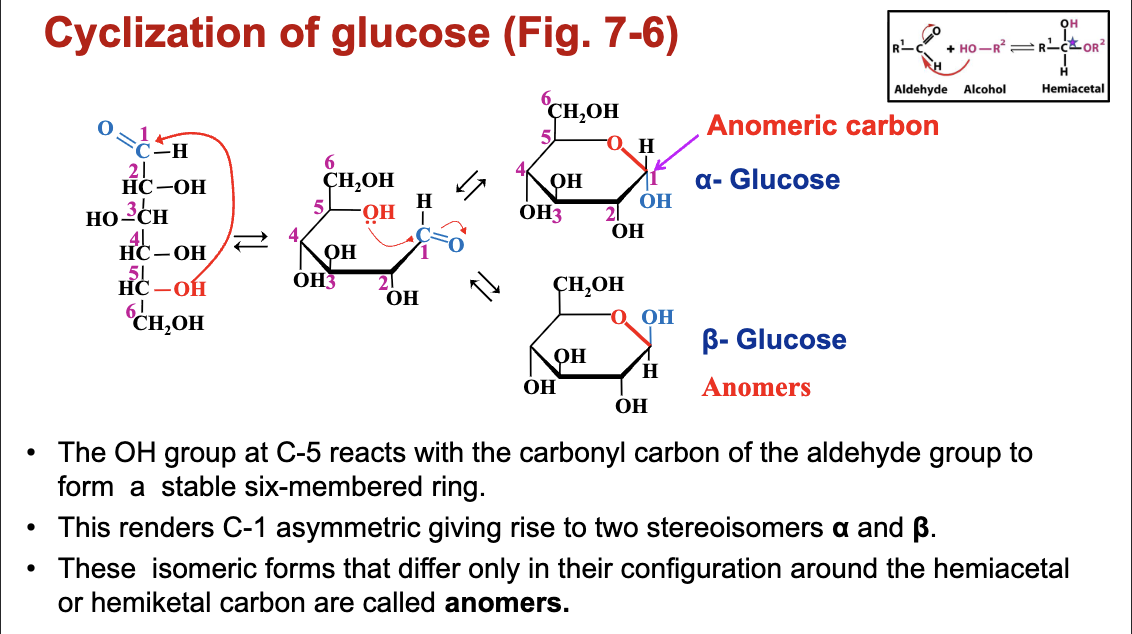
Anomeric Carbon: Referring to the newly formed chiral carbon in cyclization.
VI. Mutarotation
A. Definition and Reaction Process
When sugars are dissolved, they reach equilibrium between their linear and cyclic forms—demonstrating a process called mutarotation.
Mutarotation leads to a change in optical rotation as the sugar molecules interconvert.
B. Equilibrium of Glucose Forms
At equilibrium, glucose exists as:
1/3 alpha, 2/3 beta, and trace amounts of linear form.
Affects intensity of polarized light rotation.
A. Definition and Reaction Process
When sugars are dissolved in solution, they do not remain in a fixed cyclic form; instead, they undergo a dynamic process called mutarotation, reaching an equilibrium between their linear and cyclic forms.
This interconversion between anomeric forms (alpha and beta) proceeds through the transient open-chain aldehydic or ketonic form.
Mutarotation leads to a change in the observed optical rotation of a sugar solution over time until equilibrium is reached, due to the different specific rotations of the alpha and beta anomers.
B. Equilibrium of Glucose Forms
At equilibrium, glucose primarily exists as:
Approximately 1/3 alpha-D-glucopyranose and 2/3 beta-D-glucopyranose.
Trace amounts of the linear (open-chain) form are also present, which facilitates the interconversion but is not abundant.
The predominance of the beta anomer in equilibrium is often attributed to its greater thermodynamic stability (e.g., the equatorial position of the anomeric hydroxyl in beta-D-glucose).
The change in the ratio of alpha and beta forms affects the intensity and direction of polarized light rotation.
VII. Glycosidic Bond Formation
A. Glycosidic Bonds in Sugar Reactions
When sugars cyclize, the anomeric carbon can react with alcohols or amines to form glycosides (sugar derivatives).
Glycosides are formed through condensation reactions, which result in the release of water.
This reaction can be generalized as:
Alcohol/nucleophile + Anomeric carbon → Glycosidic bond (with water release).
VIII. Reducing Sugars
Definition: Carbohydrates that can act as a reducing agent due to the presence of a free aldehyde or ketone group (specifically, a hemiacetal or hemiketal group in their cyclic form) that can open to form the reactive aldehyde or ketone.
Mechanism: The open-chain form of these sugars contains a carbonyl group that can be oxidized, while simultaneously reducing another compound.
Examples of Reducing Sugars:
Monosaccharides: All monosaccharides, such as glucose, fructose, and galactose, are reducing sugars because they possess a free anomeric carbon.
Disaccharides: Some disaccharides like lactose and maltose are reducing sugars as they retain a free anomeric carbon capable of opening to the linear form.
Non-reducing Sugars:
Sucrose: This disaccharide is a non-reducing sugar because its anomeric carbons are linked together, preventing the formation of a free aldehyde or ketone group for oxidation.
Biological and Medical Importance:
The reducing property of sugars is exploited in various biochemical tests. For example, the detection and measurement of glucose levels in blood or urine rely on its reducing capability, which is crucial for diagnosing and managing conditions like diabetes.
VIII. Identifying Anomeric Carbon in Ring Structures
The anomeric carbon is the most reactive one in ring structures, identified by its bonding to two oxygen atoms.
IX. Summary
All discussed reactions and characteristics—cyclization, hemiacetal/hemiketal formation, and glycosidic bonds—are essential in understanding carbohydrate chemistry and the formation of polysaccharides.
Recognized the usefulness of determining reducing sugars in medical contexts, such as glucose measurement, ensuring a detailed understanding of their properties and implications in physiological processes.