VSEPR Theory
Valence Shell Electron Repulsion (VSEPR) Theory: a theory that the shape of a molecule is determined by the repulsion between electrons in the bonds and unshared pairs of the atoms.
Electrons are all negatively charged, so they repel each other. Valence electrons exist in two places:
unshared electrons (in pairs), attached to only one atom
as part of a covalent bond (shared pair of electrons) between two atoms
The VSEPR shape of the molecule is the shape that occurs when all of these “clouds” of electrons are as far apart as possible
The Lewis structure of a molecule represents the structure in 2 dimensions. The VSEPR shape is the 3-dimensional equivalent.
Table of VSEPR Shapes
Electron Clouds | Bond Atoms | Lone Pairs | Hybridization | Bond Angle | Picture | VSEPR Shape |
4 | 4 | 0 | sp3 | 109.5° | 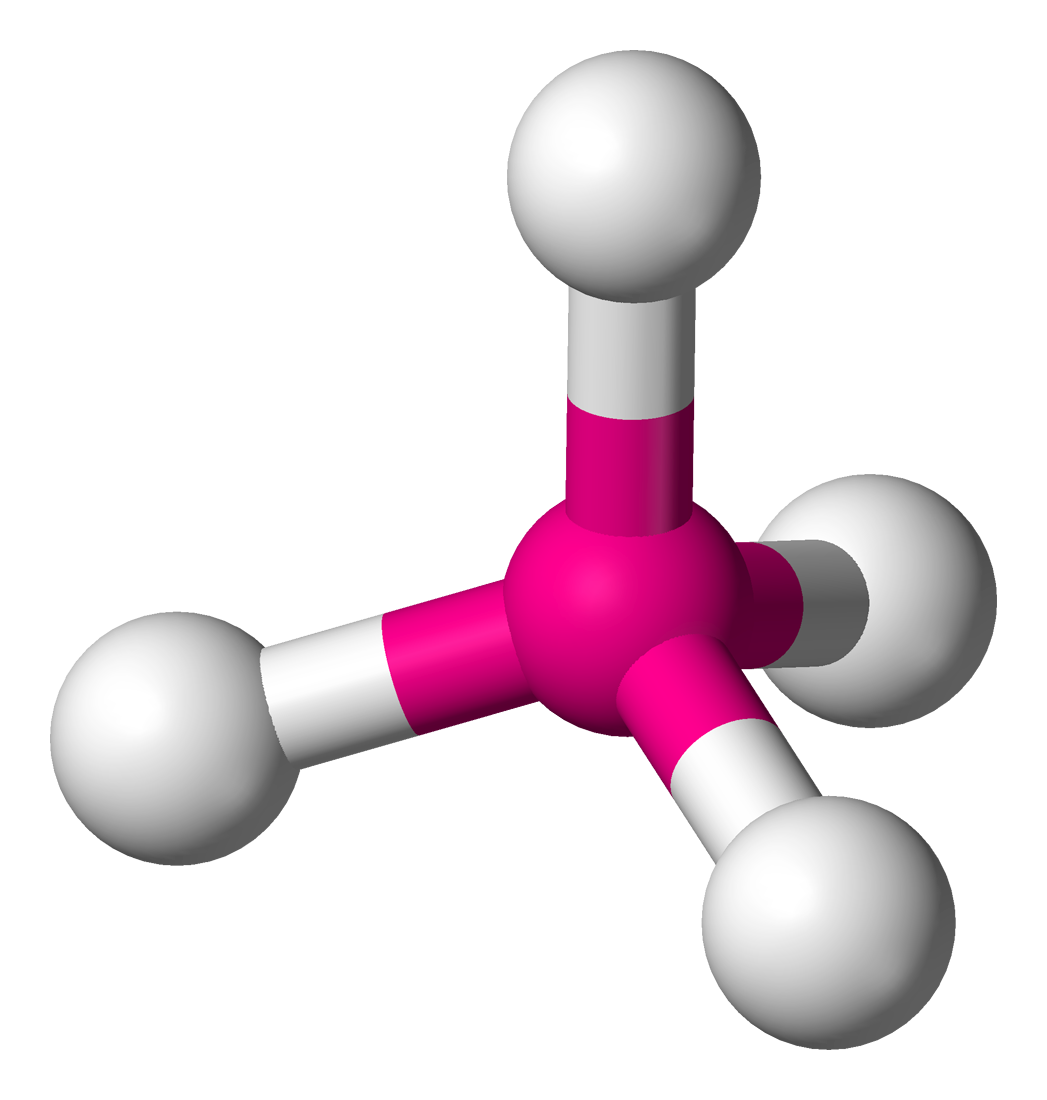 | tetrahedral |
4 | 3 | 1 | sp3 | 107.5° | 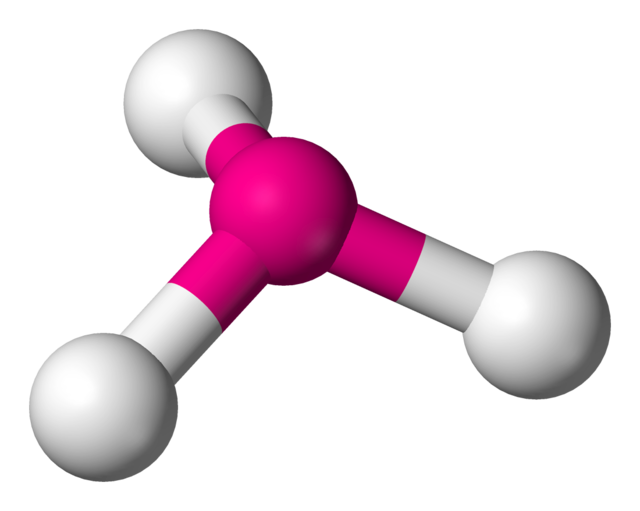 | trigonal pyramidal |
4 | 2 | 2 | sp3 | 104.5° | 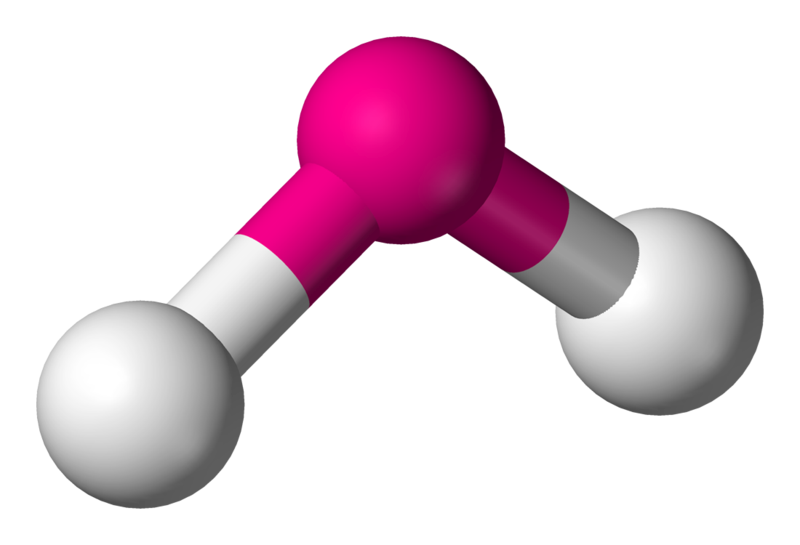 | bent |
3 | 3 | 0 | sp2 | 120° | 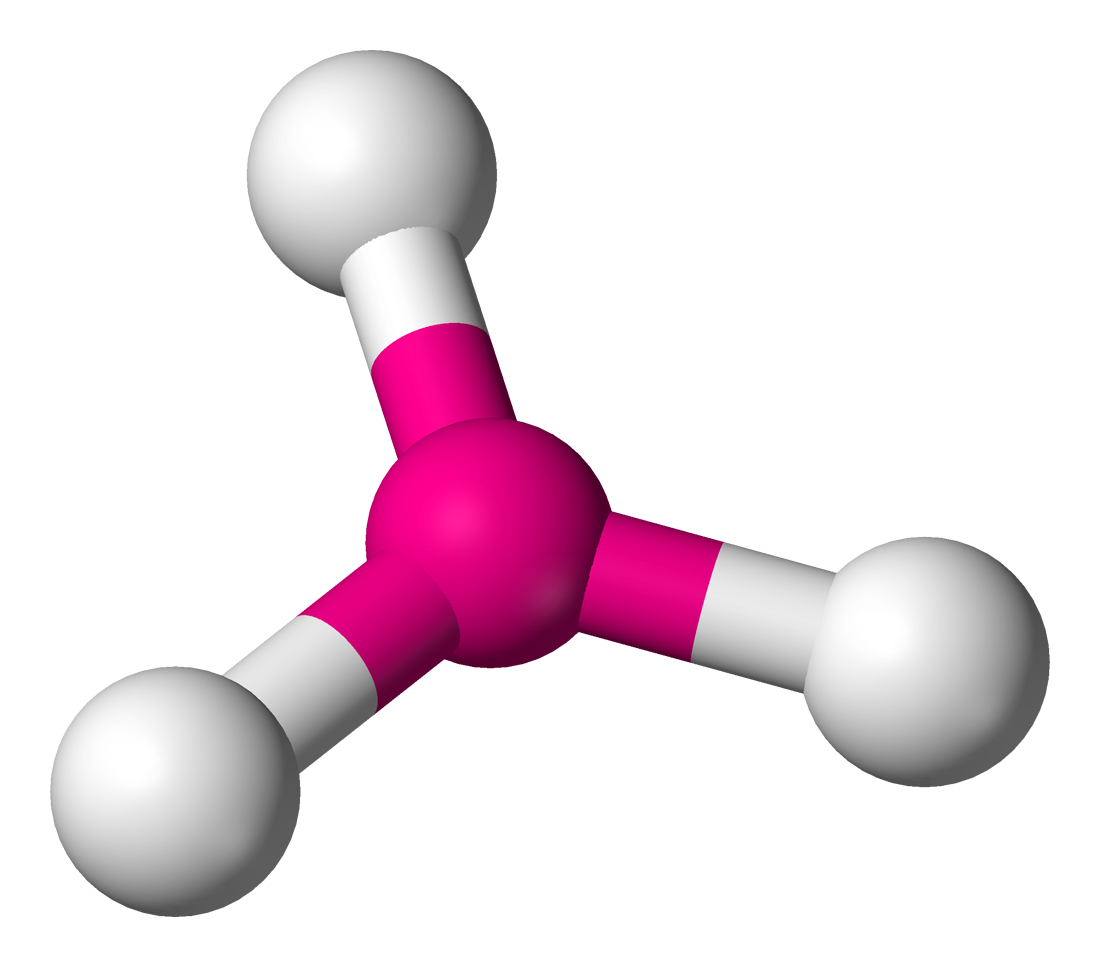 | trigonal planar |
3 | 2 | 1 | sp2 | 118° | 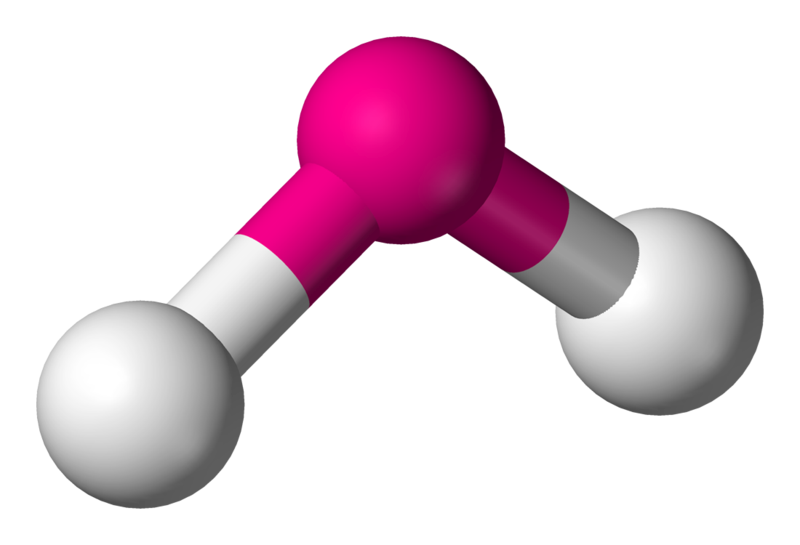 | bent |
2 | 2 | 0 | sp | 180° | 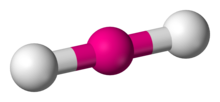 | linear |
/
*Note: Even though tetrahedral, trigonal pyramidal and bent can have four electron groups (tetrahedral EG), they experience different bond angles because unbonded pairs repel more strongly than bonded electrons.
Table of Expanded VSEPR Shapes
Electron Clouds | Bond Atoms | Lone Pairs | Hybridization | Bond Angle | Picture | VSEPR Shape |
6 | 6 | 0 | d2sp3 | 90° | 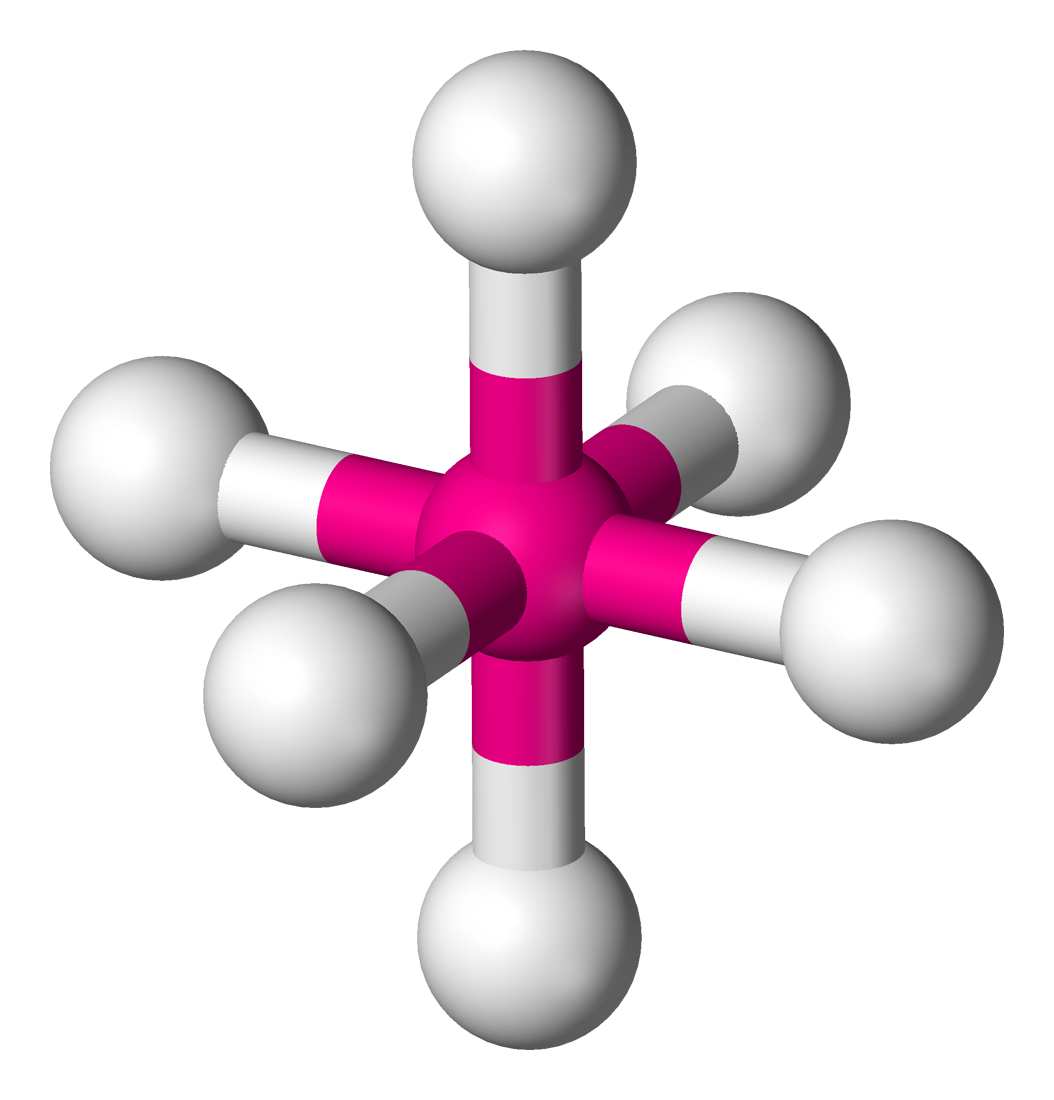 | octahedral |
6 | 5 | 1 | d2sp3 | 90° & 180° | 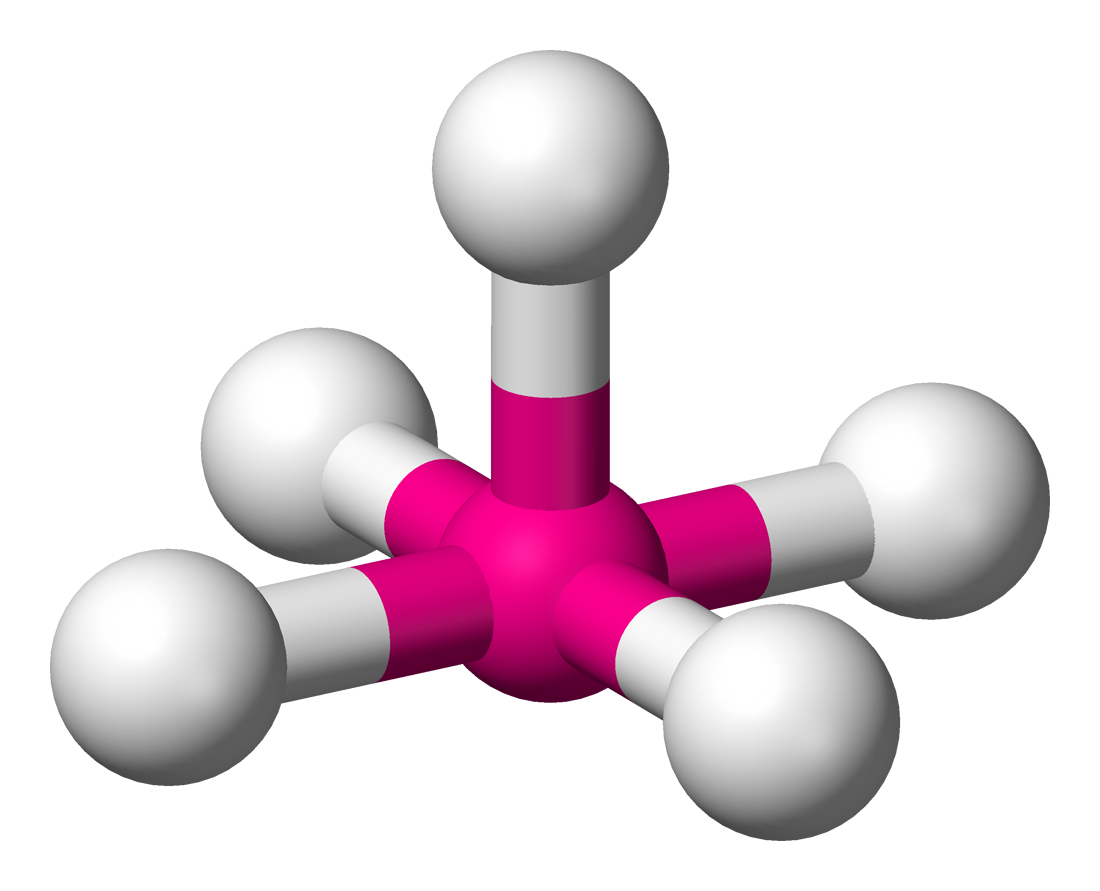 | square pyramidal |
6 | 4 | 1 | d2sp3 | 90° & 180° | 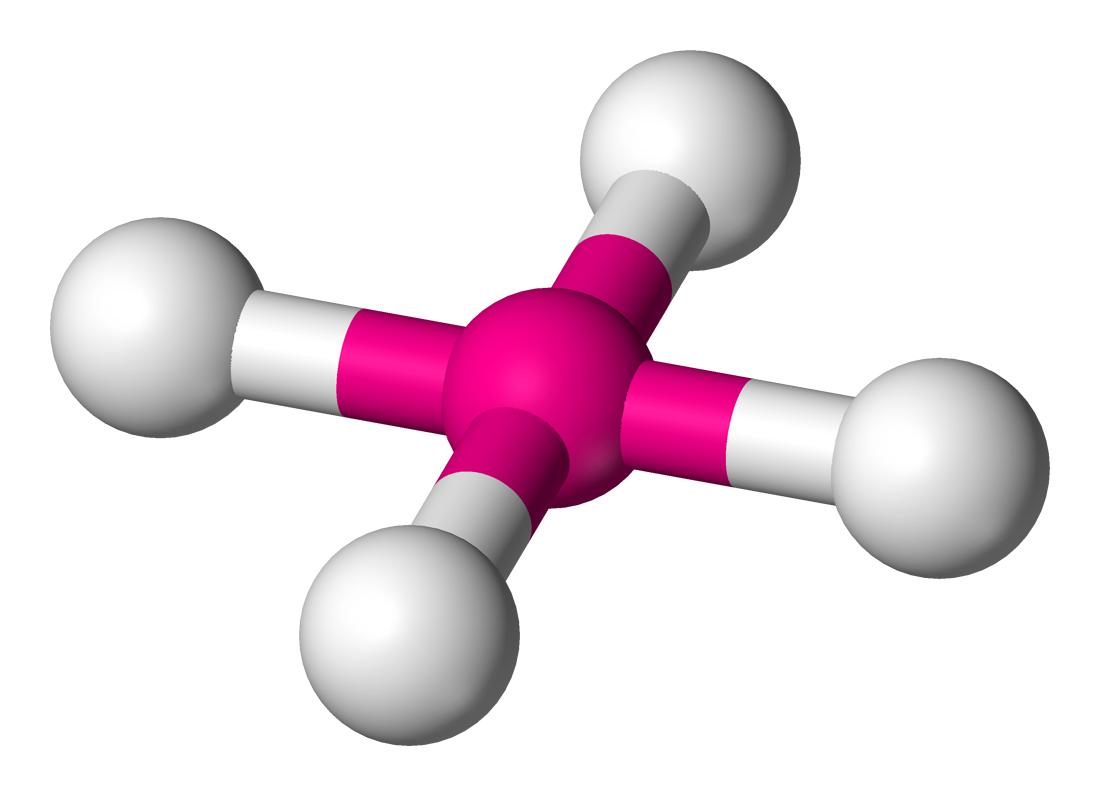 | square planar |
6 | 3 | 3 | d2sp3 | 90° & 180° | 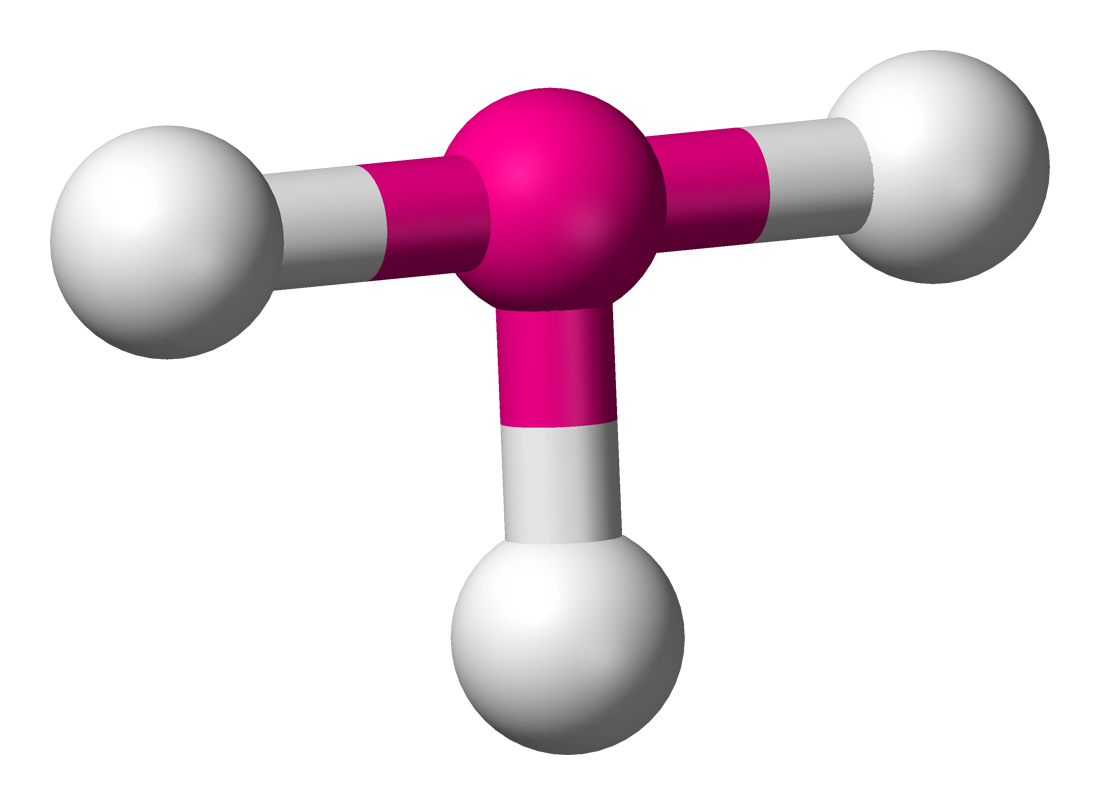 | T-shaped |
6 | 2 | 4 | d2sp3 | 180° | 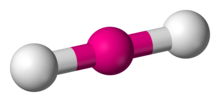 | linear |
5 | 5 | 0 | dsp3 | 90° & 120° | 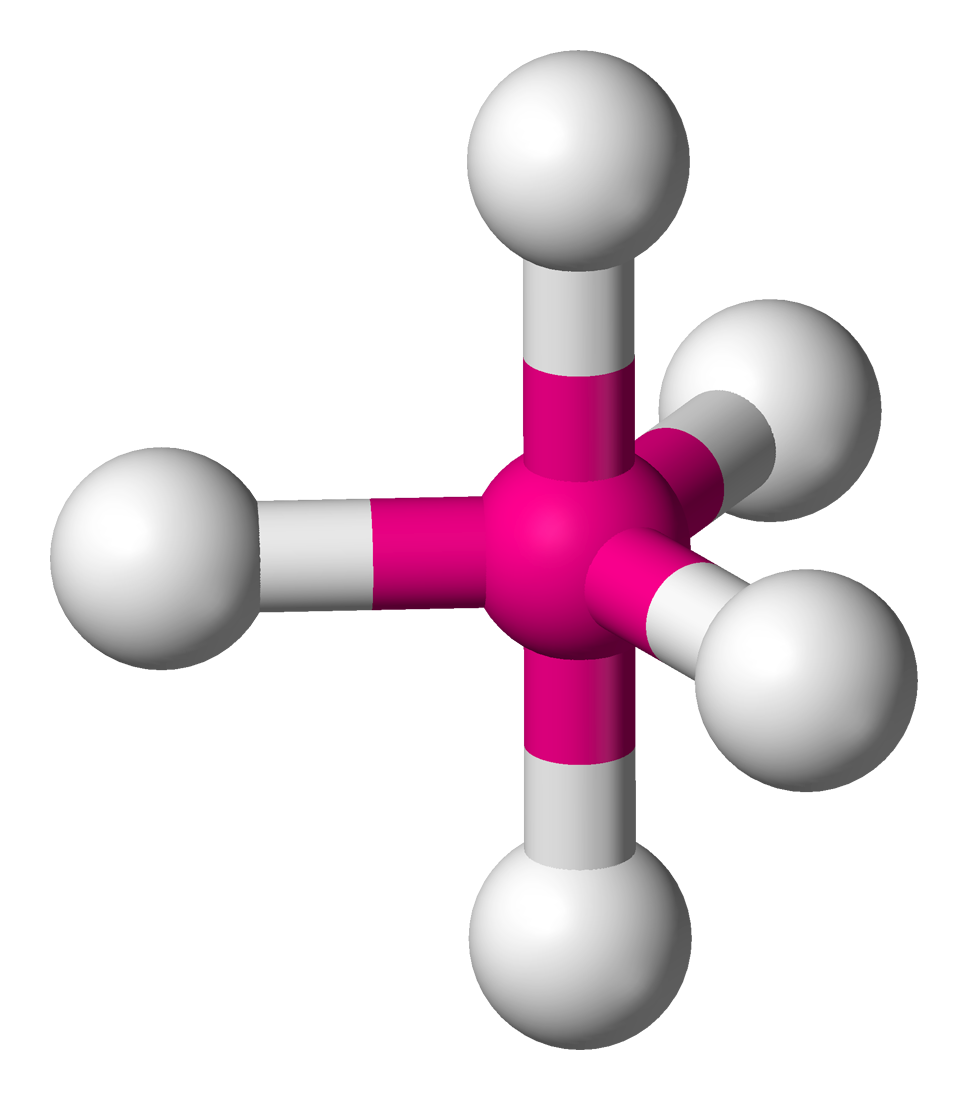 | trigonal bipyramidal |
5 | 4 | 1 | dsp3 | 90° & 120° | 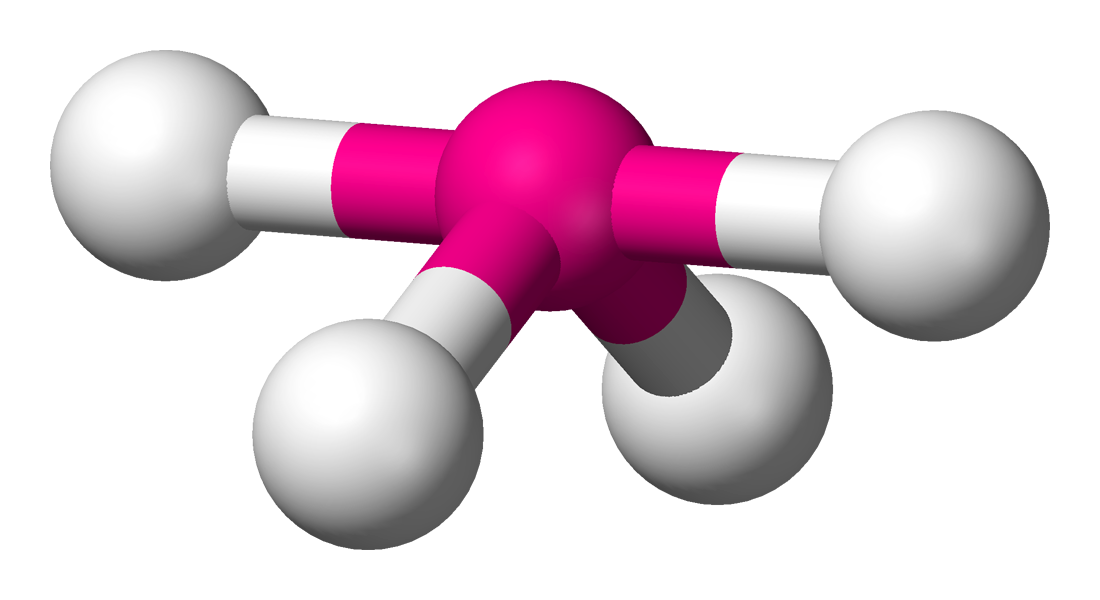 | seesaw |
5 | 3 | 2 | dsp3 | 90° & 180° |  | T-shaped |
5 | 2 | 3 | dsp3 | 180° |  | linear |