History of atom
ight lets get it:
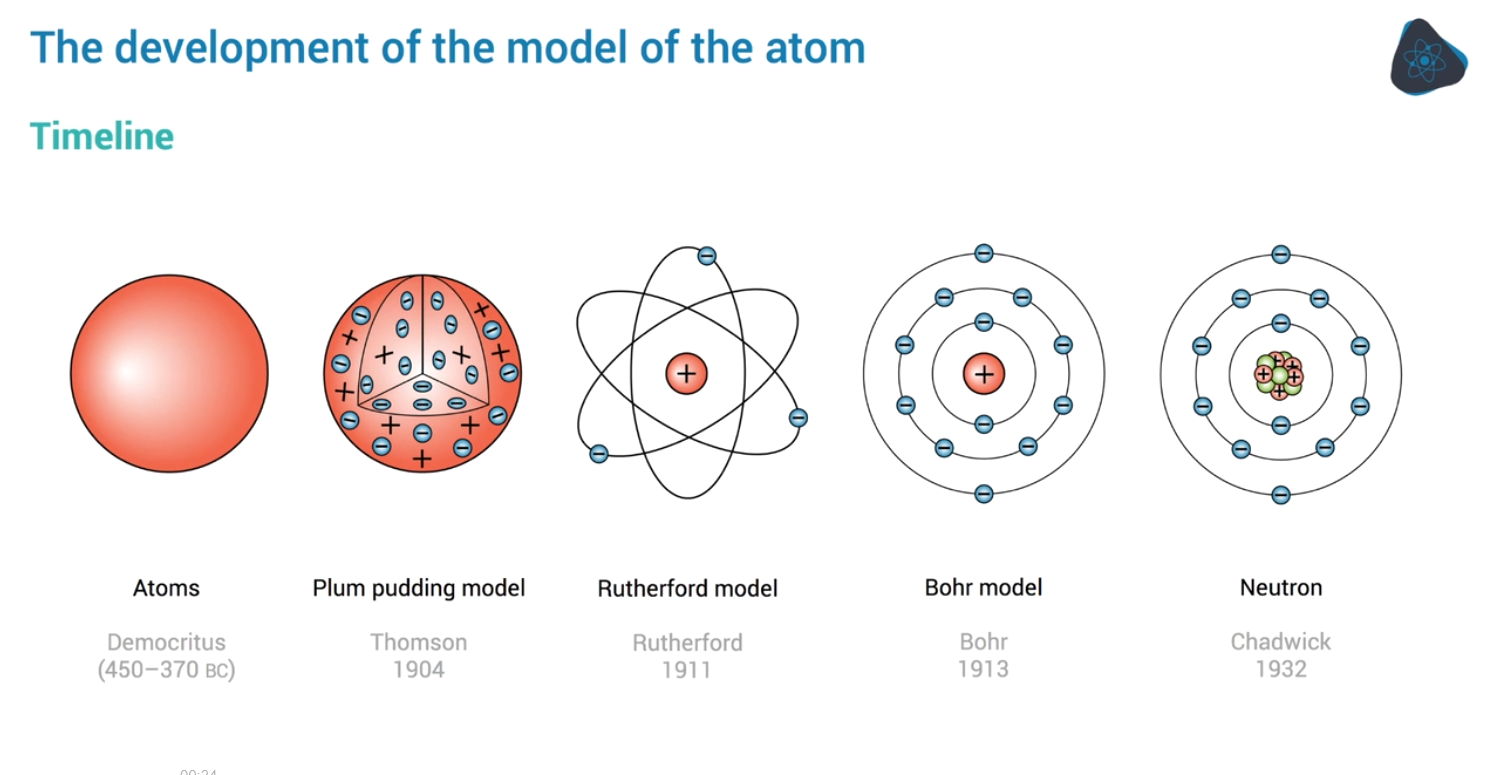
In di beningin we got Democritus (450-370) with tiny unbreakable spheres called atoms
John Dalton (1804) just agreed with this as well affirming damocritus’s guess. The belief that: atoms are tiny spheres that couldn’t be broken down any further with each element being made up of a different type of atom.
JJ Thompson (1897) discovered elections since they could be removed form atoms came up with the plum pudding model: A positive sphere with negative electrons dotted around and inside it
However this failed to answer many questions such as:
Why doesn’t the atom collapse in on itself due to electrostatic attraction
what holds the atom together- NO EXPLANATION
Lack of experimental evidence- he never tested his theory
The Rutherford experiment later disproved this
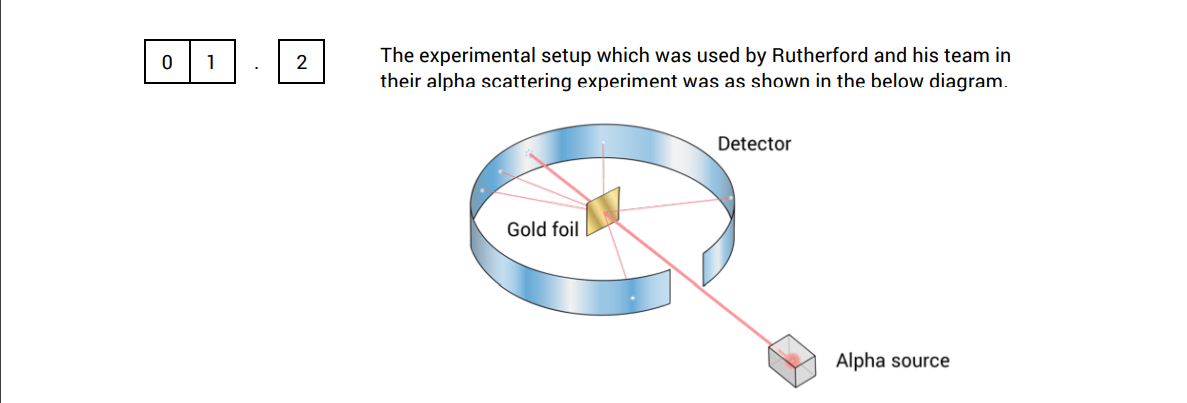
In 1909: Geiger and Marsden tested the plum pudding model under Ernest Ruderthford’s instructions: Fire alpha particles at a thin gold sheet.
If the plum pudding model was correct, the positive and negative charges would cancel each other out and the alpha particles would pass straight through. However a small number were deflected and some were even bounced right back.
This led Ernest to conclude: (1911)
that the atom was mostly empty space - because the majority of alpha particles went straight through the foil
The nucleus is positively charged- because of the few bounced off
The nucleus is at least 10000 times smaller than the atom- because a large amount was unaffected by this positive nucleus
Most mass in concentrated at the nucleus - Because the majority of alpha particles went straight through the foil
electrons orbit the positive nucleus- So that they don’t collapse into the nucleus via electrostatic attraction
This happened because alpha particles that flew close to the positive nucleus got deflected and changed course. particles that got too close got completely redirected and the rest just went straight through.
The reason why they used gold foil was because it was very thin at only a few atoms of thickness
Later on Ernest also discovered the proton in 1919
and James Chadwick discovered the neutron in 1932
the last person we care about (screw you Scrodinger) is Niels Bohr:
Bohr said that electrons only move around the nucleus in fixed shells:
The further away from the shell they are the more energy it has
Electrons could gain / lose energy by jumping between orbits
emitting electro-magnetic radiation (EMR) when it moves down and loses energy
absorbing EMR when it moves up and gains energy
Expected Questions