3.8 nucleophilic addition mechanisms
reduction of aldehydes and ketones by nucleophilic addition using warm aqueous NaBH4:
overall
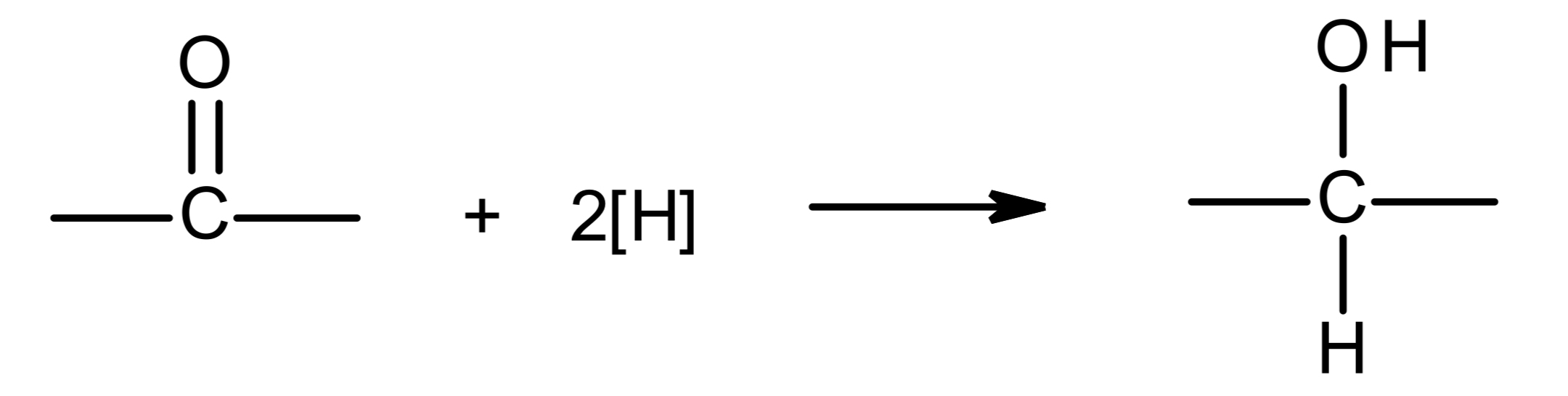
2 H atoms are added onto the C=O bond, one onto the C and one onto the O
mechanism
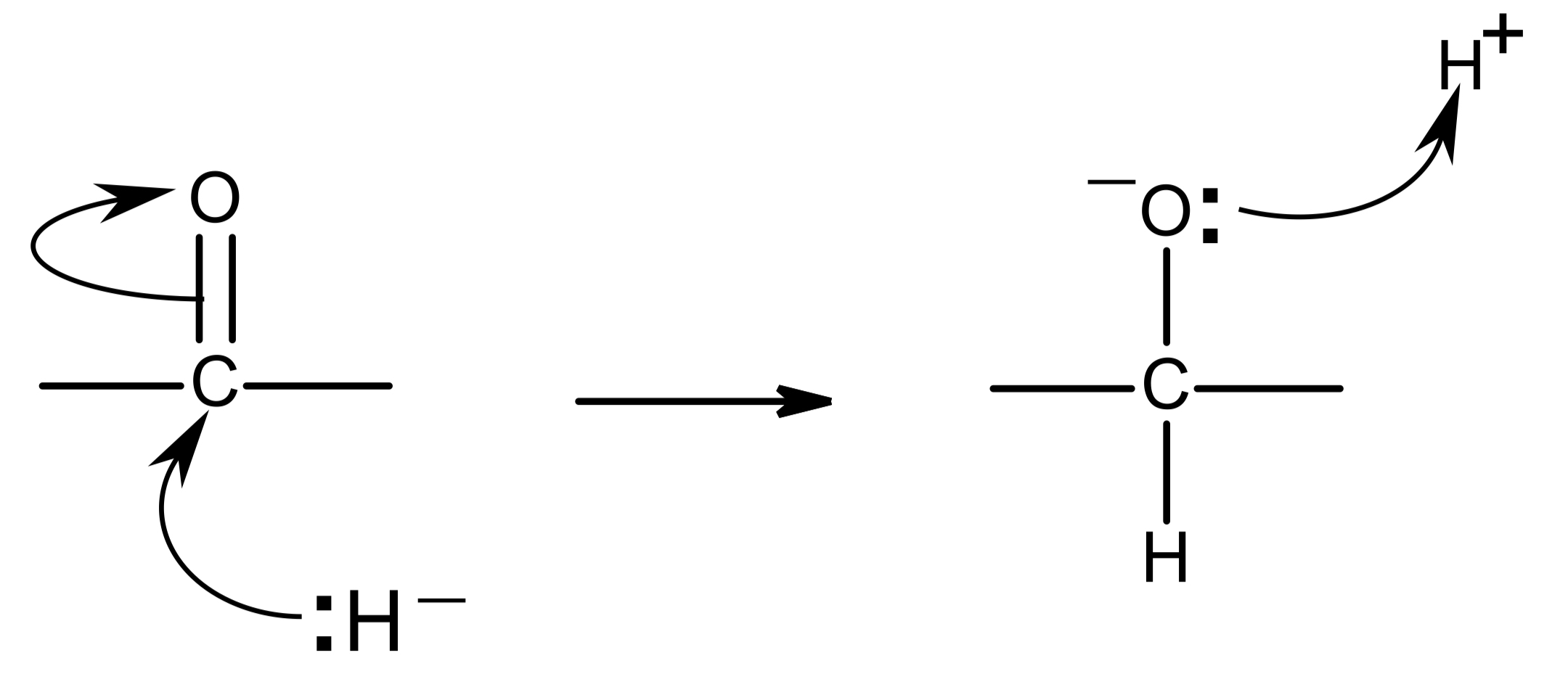
NaBH4 generates hydride ion, H-
lone pair on H- is donated to δ+ C, forming a dative covalent bond
at the same time, the pi bond in the C=O bond breaks by heterolytic fission, and both electrons in the pi bond move onto the O atom
lone pair on O- in intermediate is donated to H+ from a water molecule
nucleophilic addition reaction between aldehydes or ketones and KCN with dilute acid:
overall
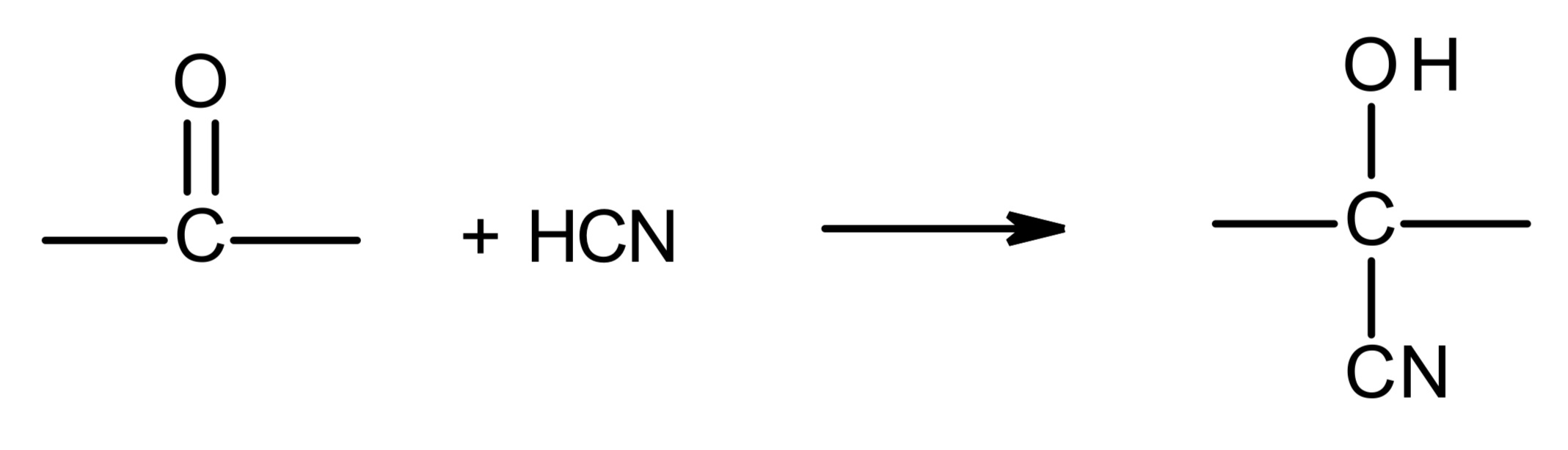
CN adds onto the C of the C=O, H adds onto the O of the C=O
mechanism
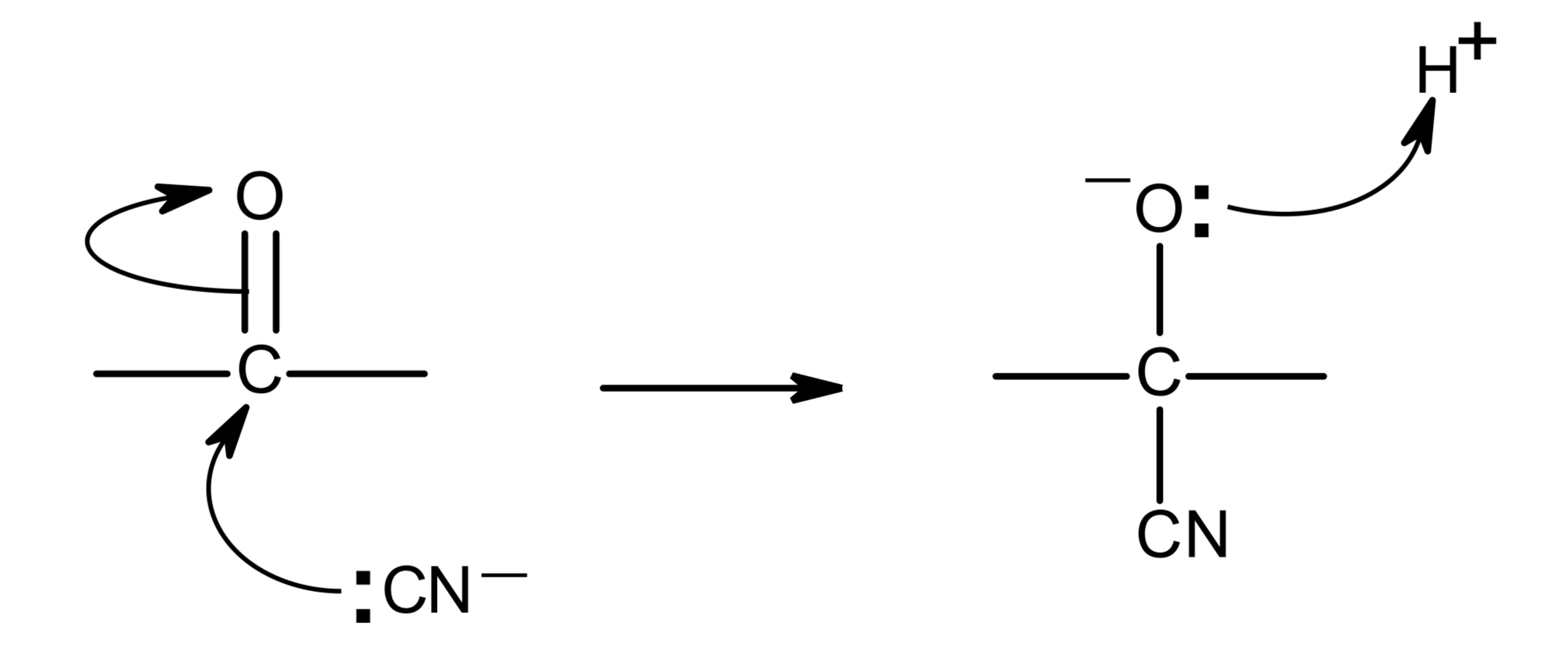
KCN generates cyanide ion, CN-
lone pair on CN- is donated to δ+ C, forming a dative covalent bond
at the same time, the pi bond in the C=O bond breaks by heterolytic fission, and both electrons in the pi bond move onto the O atom
lone pair on O- in intermediate is donated to H+ from dilute acid, e.g. H2SO4