Particle Physics
Linear Accelerators (LINACs)
AC supply: ensures the ions are always accelerated from one tube to the next.
Tube: The ions are attracted to the midpoint of the tube.
AC supply switches so ions are repelled towards the exit and attracted to the next tube.
Tube is built successively longer so ions spend the same amount of time accelerating through each tube.
Frequency of the supply: Constant so the polarity switches at a constant rate
*Uses electric fields only
Circular Accelerator
Cyclotron:
Alternating pd: Produces an electric fields
Causes a difference between the dees.
Alternating so when the protons pass from one dee to another, they are attracted towards the other dee.
High frequency: Specific rate of change is required so protons are attracted across gaps at right time.
Electric fields: Provides a force on proton in the gap (F=Eq )
Provides energy to accelerate proton across gap.
Has no effect on proton once it enters the dee as dee is screened against the field.
Magnetic Fields: Provides force on moving proton at all times (F=Bqv )
Force is perpendicular to motion of proton which causes circular motion.
Force provides centripetal force.
How Cyclotron works
Protons are released from the centre and enter the first dee at 90 degrees to the magnetic field.
Proton follows circular path for half a revolution until they reach a gap.
Proton is accelerated across the gap and gains energy from electric field.
Proton is attracted to negatively charged dee.
Protons are travelling faster so the radius of their path will be larger. Time of flight is constant each revolution.
Protons cross the gap twice each revolution until they are released as a beam.
Synchrotron
Made up several LINACs in a circle with a magnetic field to ensure beams stay on track.
Stationary particles are accelerated to relativistic speeds.
Charged particles emit EM radiation and lose energy.
This is emitted in form of light and is know as synchroton radiation.
Why high speeds are necessary in particle accelerators
To overcome electrostatic forces of repulsion.
To break intermolecular forces and discover new particles.
Mass increases when travelling at relativistic speeds (E=mc^2 )
Atomic Structure
John Dalton
Modern theory of atom: each element is made up of its own indivisible atoms.
J.J Thompson
Plum pudding model: atoms were spheres of positive charge with tiny negative electrons stuck in them.
Rutherford Scattering Experiment
Observation | Conclusion |
Most of alpha particles went straight through the foil | Made up of mostly empty space |
Some of the alpha [articles were deflected through large angles | Small, dense positively charged |
Very few of the alpha particles were deflected by angles greater than 90 degrees | Most of the mass is concentrated in the nucleus |
Particle | Relative mass | Actual mass | Relative charge | Actual charge | Position |
Proton | 1 | 1.6726\cdot10^{-27}\operatorname{kg} | +1 | +1.6\cdot10^{-19}C | Nucleus |
Neutron | 1 | 1.6749\cdot10^{-27}\operatorname{kg} | 0 | 0 | Nucleus |
Electron | 1/1842 | 9.1094\cdot10^{-31}\operatorname{kg} | -1 | -1.6\cdot10^{-19}C | Shell |
Standard Notation

Fundamental Forces
Gravitational Force: Acts on anything that has mass and is always attractive. Only obvious with very massive particles.
Electromagnetic force: the force holds atoms together.
Strong nuclear force: Binds atomic nuclei together. Works over very small distances (femtometres).
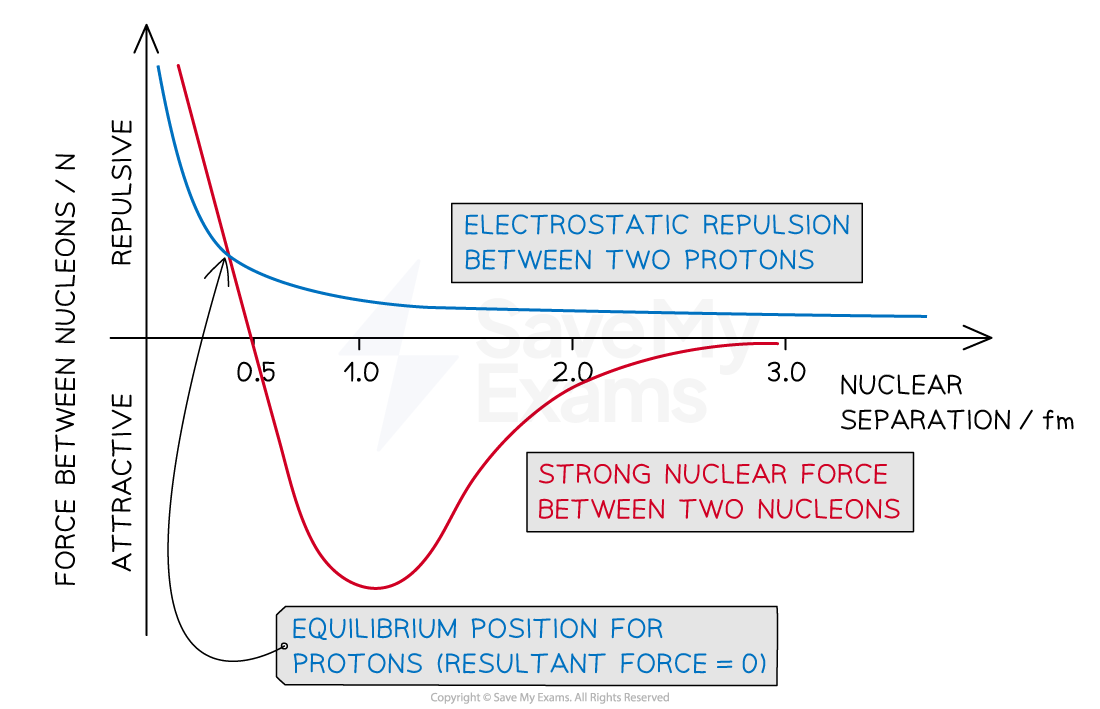
\le0.5fm
Repulsive so protons do not collapse into each other each other.
\le2fm
Attractive and binds protons together.
>5fm
Has no effect. EM force becomes dominant so protons will repel each other.
Weak nuclear force: Responsible for radioactive decay of the nucleus. Defines how unstable atoms become stable.
Force | Relative Strength | Range | Force carrier |
Strong | 1 | .10^{-15} | Gluons |
Electromagnetic | 0.0013 | Infinite | Photon |
Weak | 0.000001 | .10^{-18} | Bosons |
Gravitational | \cdot10^{-39} | Infinite | Graviton? |
Classification of Particles
Antimatter
Same mass, opposite charge.
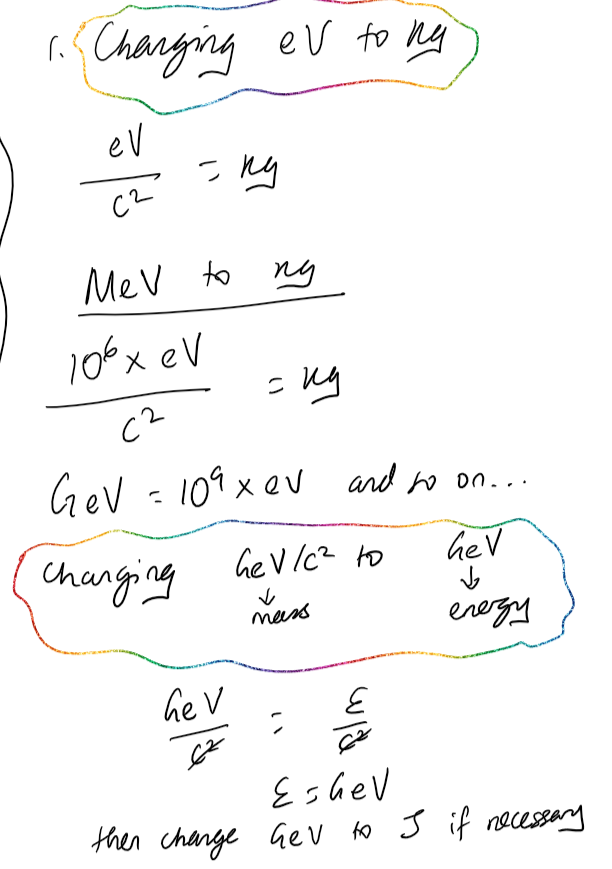
Rest Energy (E_{o} )
The equivalent energy of all the mass of a particle.
Energy into mass (Pair Production)
Energy is converted into mass and equal amount of matter and antimatter are produced.
Pair production will always produce a particle and an antiparticle
Scenarios
The minimum energy needed is the total rest energy of all particles produced.

Mass into energy (annihilation)
Particle - antiparticle collision always results in annihilation.
The entire mass of all particles involved becomes energy (gamma ray photons).
The minimum energy produced will be the same as the rest energy of the particles originally involved.
Fundamental Particles
Cannot be broken up to anything smaller. The two fundamental particles are quarks and leptons.
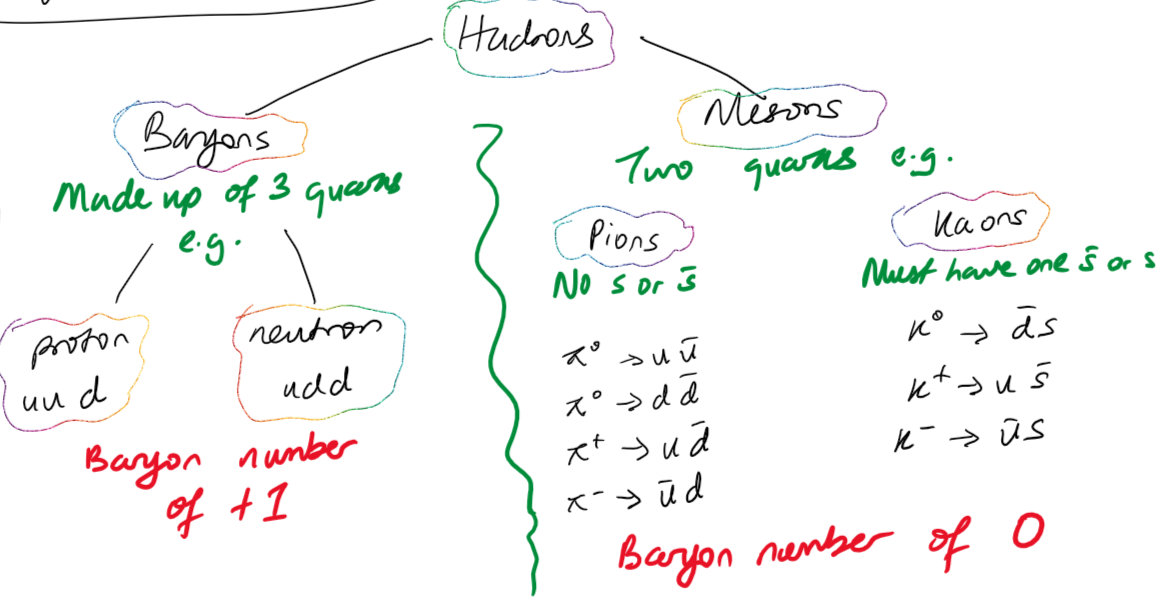
Quarks
Have different masses and charges
Has an anti-quark
Feels the strong force
Cannot exist on their own (exist in twos or threes) - Quark confinement
Name | Charge | Baryon Number | Strangeness |
up | +\frac23 | +\frac13 | 0 |
down | -\frac13 | +\frac13 | 0 |
strange | -\frac13 | +\frac13 | -1 |
Strangeness
A property of strange quarks and strange antiquarks.
Strange particles are created via the strong force. Strangeness is conserved during creation.
They decay via the weak nuclear force. Strangeness isn't conserved in the decay.
Leptons
Do not feel the strong force. Only interact via the weak force.
Electrons are the only stable leptons.
Muon and tau decay into an electron.
Each lepton has a neutrino.
Lepton number must be conserved.
Name | Symbol | Charge | Lepton e number | Lepton \mu number | Lepton \tau number |
electron | e^{-} | -1 | +1 | 0 | 0 |
electron neutrino | v_{e} | 0 | +1 | 0 | 0 |
muon | \mu^{-} | -1 | 0 | +1 | 0 |
muon neutrino | v_{\mu} | 0 | 0 | +1 | 0 |
tau | \tau^{-} | -1 | 0 | 0 | +1 |
tau neutrino | v_{\tau} | 0 | 0 | 0 | +1 |
Detecting Particles
Cloud Chamber
Contains super-cooled vapours
Charged particles pass through and ionise the vapour leaving a trail of ions (vapour trails)
Bubble Chamber
Contains pressurised hydrogen liquid
Charged particles pass through, then we reduce pressure and bubbles of gas in the places where there are trails of ions
We can determine the mass and charge of particles by applying a magnetic field.
Short heavy trails: lots of ionisation. Could be a proton or alpha, etc.
Long, faint tracks: little ionisation eg. electron or positron
Rules for determining what tracks show
Visible tracks: Charged particles are moving
Straight line: Charged particles with so much momentum that they don’t interact
Curves
r=\frac{mv}{Bq}
Two curves in opposite directions are oppositely charged particles to conserve charge
Two curves in opposite directions with equal radius infer momentum is conserved
V shape appearing out of nowhere: Decay of a neutral particle into two oppositely charged particles.
Tiny single spirals from a straight track: shows a knock-on electron.