chapter 2 - atomic structure & radioisotopes
2.1 - elements & structure of the atom
matter
pure substance → element or compound
element → atoms
compound → elements → atoms
mixture → homogeneous or heterogenous
structure & properties of the atom
protons (1+), neutrons (0), & electrons (1-)
electronic cloud - electrons on the outside (orbitals) & are being pulled towards nucleus
nucleus - protons & neutrons, have same amount to be a neutral atom, neutrons relieve the stress from so many protons
protons - 1.007 amu
neutrons - 1.008 amu
both determine the mass of an atom
electron - 0.00055 amu
element
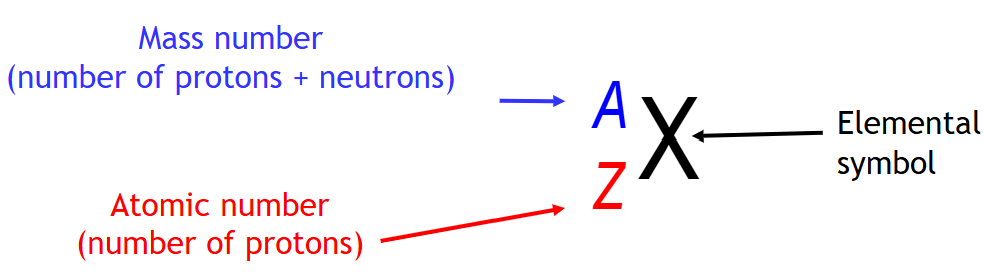
number of protons in contains → atomic number
ID of an element is given by the number of protons
every element has a 1 or 2 letter atomic symbol or chemical symbol
atomic symbol, atomic number, atomic mass → given on periodic table
protons + neutrons = mass of the atom
mass number → A
isotopes
same element with same number of protons but different number of neutrons
all elements have their own unique percent natural abundance of isotopes
symbol notation
a) 17 protons, 18 neutrons, 18 electrons
b) 17 protons, 20 neutrons, 17 electrons
c) 19 protons, 19 neutrons, 22 electrons
d) 6 protons, 6 neutrons, 8 electrons
average atomic mass
atomic mass of an element represents the average mass of the atoms that compose that element
calcium-44 is heaviest bc it has most neutrons
average mass closer to Ca-40 since it has the largest contribution (96.55%)
average of A:
4 (1$) + 4($2) = 4 + 8 = 12/8 = 1.5
4 + 4 8
average of B:
1($1) + 9 ($2) = 1 + 18 = 19/10 = 1.5
1 + 9 10
average: 24.23 × 36.97 + 75.77 × 34.97 / 100
895.7831 + 2649.6769 / 100
3545.46 / 100 = 35.45 amu
2.2 - navigating the periodic table
18 columns - groups
similar chemical & physical properties → noble gases, halogens, alkali metals (hydrogen is not a metal, react violently with water), alkaline earth metals (less reactive)
main groups or transition metals
7 rows - periods
metals, nonmetals, & metalloids
metals
shiny, malleable, ductile
good conductors of heat & electricity
exist as solids at room temp except mercury (liquid)
nonmetals
dull, brittle
poor conductors of heat or electricity → good insulators
exist as solids, gases & liquids at room temp
metalloids or semi metals
have intermediate properties
between metals & nonmetals
important elements in biochem & medicine
building block elements
hydrogen
carbon
nitrogen
oxygen
phosphorus
sulfur
macronutrients
sodium
magnesium
potassium
calcium
chloride
phosphorus
sulfur
micronutrients
iron
copper
zinc
iodine
fluoride
chromium
vanadium
molybdenum
selenium
silicon
2.3 - electron arrangement & valence electrons
modern picture of the electron in an atom comes from quantum mechanics
behavior of subatomic particles is not governed by the same physical laws as larger objects
describe the behavior of macroscopic objects
electrons are very small & light
valence electrons
electrons in highest energy level of an atom
responsible for the chemistry
group 7 has 7 electrons, etc
2.4 - radioisotopes
radioactive isotope (radioisotope)
has an unstable nucleus → all isotopes w atomic # > 82 are radioactive
undergoes decay to become more stable → spontaneous process
radiation emitted during decay
radiation can be
subatomic particle or high energy particle
a - alpha particle
b - beta particle
electromagnetic radiation or high energy light wave
y - gamma ray
x ray
parent nuclide (unstable) → daughter nuclide + radioisotope
alpha particle
slow-moving
high energy
2 protons & 2 neutrons
+2 charge
beta particle
high energy
electron
most radioactive decay includes gamma rays
only energy
half-life
time needed for ½ of the parent nuclides to decay into daughter nuclides