C1.2 Cell respiration Notes
C1.2.1 atp as the molecule that distributes energy within cells
ATP as the Molecule That Distributes Energy Within Cells
If your phone battery was running low you'd need a quick charge to keep it going.
In cells, adenosine triphosphate (ATP) acts like that quick charge, providing energy exactly when and where it's needed.
Definition
ATPATP is a nucleotide, similar to those that make up DNA and RNA.
Note
The three phosphate groups in ATP are negatively charged, creating repulsion between them.
This makes the bonds unstable and high-energy, perfect for quick energy release.
Why is ATP So Special?
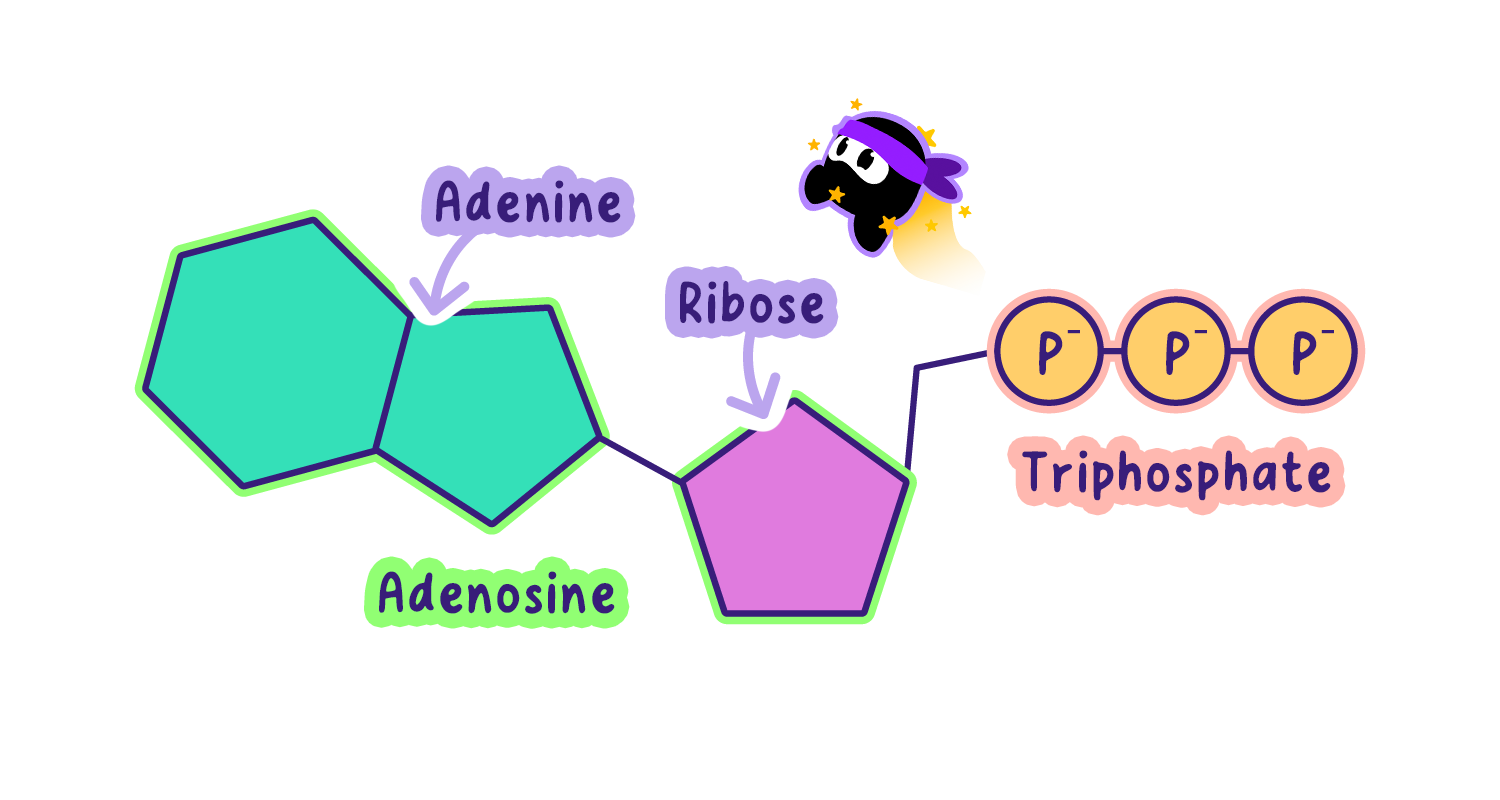
ATP consists of three parts:
Adenine: A nitrogen-containing base.
Ribose: A five-carbon sugar.
Three phosphate groups: Linked in a chain, with the last two bonds holding high-energy potential.
High-Energy Phosphate Bonds
The bond between the second and third phosphate groups in ATP is a high-energy bond.
Breaking this bond releases energy that cells can use for various functions.
This energy release makes ATP the perfect molecule for fueling cellular processes.
Recyclable
ATP is continually recycled within cells.
When it's used to power a process (e.g., muscle contraction or active transport), it's converted into ADP (adenosine diphosphate) and inorganic phosphate (Pi).
The cell then regenerates ATP by adding a phosphate group back to ADP through cellular processes like cellular respiration (in mitochondria) or photosynthesis (in plants).
Universal Energy Currency
ATP is used by almost all forms of life, from single-celled organisms to humans, to power biochemical reactions.
This universality highlights how crucial ATP is for the survival of life on Earth.
Warning
Students often confuse ATP with enzymes or proteins.
Remember, it is a nucleotide, not a protein.
Why is ATP Called the Energy Currency of the Cell?
Just as money is used to buy goods and services, ATP is used to "pay" for energy-requiring processes in cells.
Here's why ATP is perfectly suited for this role:
Solubility: ATP is soluble in water, allowing it to move freely through the cytoplasm and other aqueous environments within the cell.
Controlled Movement: ATP cannot easily cross cell membranes, ensuring it stays within the cell where it's produced and needed.
Reversible Reactions: The third phosphate group in ATP can be easily removed and reattached through hydrolysis and condensation reactions.
Efficient Energy Release: ATP releases a small, manageable amount of energy, minimizing waste. If too much energy were released at once, it would be lost as heat.
This reversibility allows ATP to be recycled continuously.
Analogy
ATP is like money in a cell's economy.
It is spent on "transactions" (biological processes) and can be "earned" (regenerated) through cellular respiration.
Example
When ATP is hydrolyzed to ADP (adenosine diphosphate) and a phosphate group, about 30.5 kJ/mol of energy is released.
This energy is used for processes like muscle contraction or active transport.
Warning
Don't confuse ATP with long-term energy storage molecules like glycogen or fats.
ATP is for immediate use, not storage.
How Does ATP Work?
ATP powers cellular processes by transferring its phosphate group to another molecule, a process called phosphorylation.
This transfer makes the recipient molecule more reactive, enabling it to perform work.
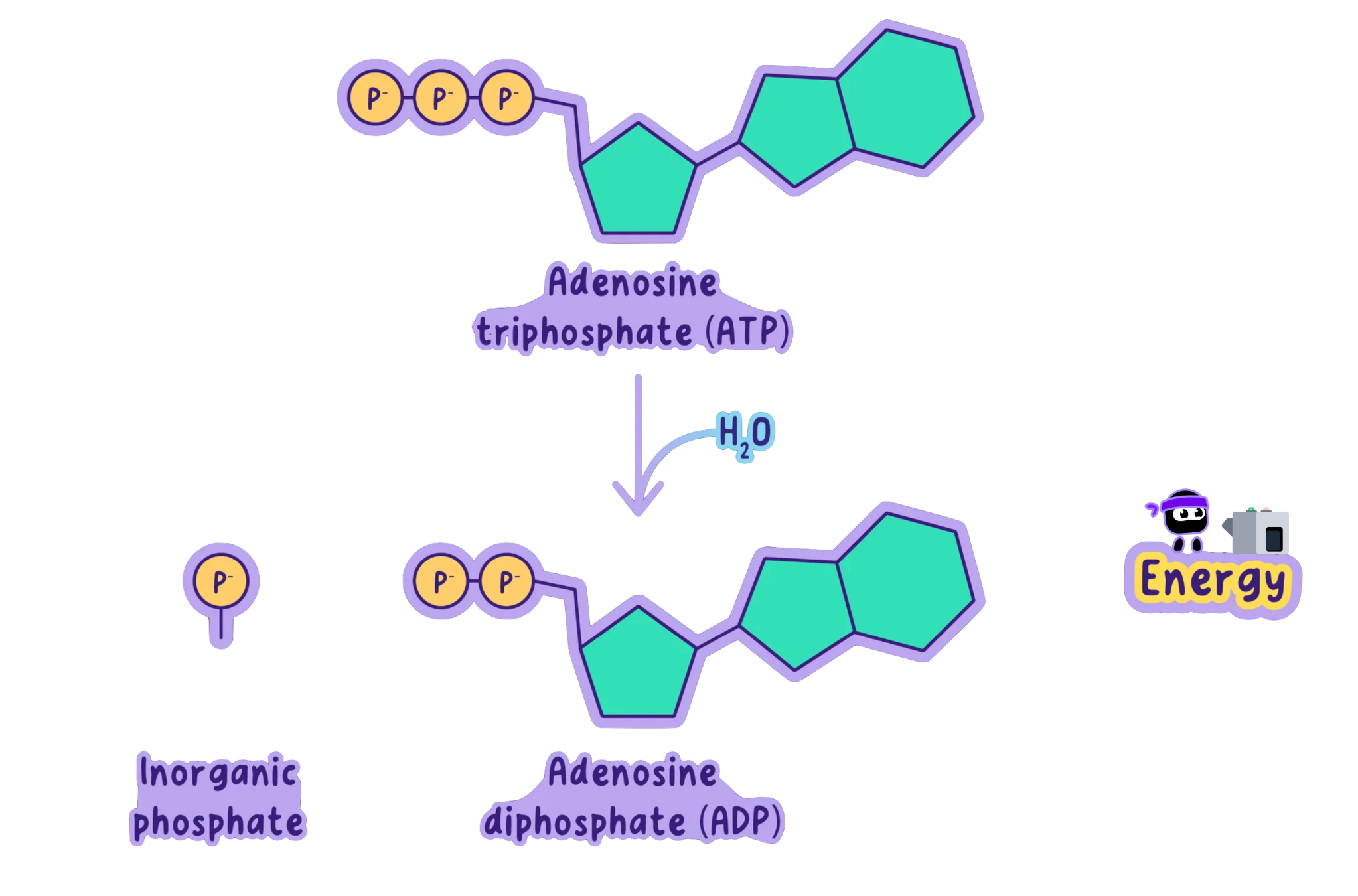
When ATP is used by the cell for energy, it undergoes a process called hydrolysis, in which the bond between the second and third phosphate groups is broken.
This releases energy and converts ATP into ADP and inorganic phosphate (Pi).
ATP + H₂O -> ADP + Pi + Energy
This reaction can be described as exergonic because it releases energy.
Note
The breaking of the third phosphate group in ATP releases enough energy to move muscle fibers, power nerve impulses, or build proteins.
Note
While ATP is crucial, it cannot provide energy indefinitely.
It needs to be continually regenerated through metabolic processes like cellular respiration (in mitochondria) and photosynthesis (in plants).
Analogy
Think of ATP as a rechargeable battery.
It releases energy when hydrolyzed to ADP, and is "recharged" back to ATP using energy from processes like cellular respiration.
Self Review
How does ATP release energy?
Describe the importance of ATP in cellular processes such as muscle contraction or protein synthesis.
How is ATP regenerated from ADP?
C1.2.2 life processes within cells that atp supplies with energy
ATP Powers Active Transport Across Membranes
Definition
Active transportActive transport is the movement of molecules or ions across a membrane against their concentration gradient (from low to high concentration), which requires energy.
Cells often need to move substances against their concentration gradient, from areas of low concentration to high concentration.
This process of active transport, requires energy.
Warning
Don't confuse active transport with passive transport.
Passive transport doesn't require energy because it moves substances down their concentration gradient.
Example
The sodium-potassium pump is a classic example.
It uses ATP to move sodium ions out of the cell and potassium ions into the cell, maintaining essential ion gradients for nerve impulses and muscle contractions.
ATP Powers The Synthesis of Macromolecules (Anabolism)
Cells constantly build complex molecules like proteins, DNA, and polysaccharides.
These anabolic reactions require energy because they are endothermic (energy-absorbing).
Each monomer added to a growing polymer, such as a nucleotide in DNA or an amino acid in a protein, requires ATP.
Definition
Anabolic ReactionsAnabolic reactions are metabolic processes in which smaller molecules are built into larger, more complex molecules using energy.
Examples of Anabolic Processes
DNA Replication: ATP provides energy to link nucleotides into long DNA strands.
Protein Synthesis: During translation, ATP powers the formation of peptide bonds between amino acids.
Polysaccharide Formation: ATP is used to assemble glucose molecules into glycogen or starch for energy storage.
Analogy
Remember that anabolism builds molecules (think "A" for "assemble"), while catabolism breaks them down.
Warning
Students often confuse catabolism (breaking down molecules) with anabolism (building molecules).
Remember, anabolism consumes ATP, whereas catabolism generates it.
ATP Powers the Movement of Cells and Cell Components
Movement is essential for many cellular processes, from dividing cells to immune responses.
Types of Movement Powered by ATP
Intracellular Movement: ATP drives the movement of organelles and vesicles along cytoskeletal tracks.
Chromosome Movement: During mitosis, ATP powers the spindle fibers that separate chromosomes.
Cell Locomotion: Cells like phagocytes use ATP to change shape and move toward infection sites.
Example
During cytokinesis, ATP is used to pinch the cell membrane inward, dividing the cell into two daughter cells.
ATP Powers Muscle Contraction
ATP is essential for muscle contraction, which involves the sliding of actin and myosinfilaments in muscle fibers.
ATP binds to the myosin head (a motor protein), allowing it to detach from actin, move, and reattach to another site on the actin filament.
Note
More on this in B3.3.2 Sliding filament model of muscle contraction (HL only)
Tok
When we call ATP the "energy currency" of the cell, we're using a metaphor from economics to explain a biological process.
How do metaphors shape the way we understand scientific concepts?
Can they also limit our understanding by making us think of living systems too much like human systems?
C1.2.3 energy transfers during interconversions between atp and adp
Energy Transfer Occurs During Interconversions Between ATP and ADP
ATP (adenosine triphosphate) is a high-energy nucleotide that serves as the primary energy currency of the cell.
The interconversion between ATP and ADP (adenosine diphosphate) involves energy transfer that drives essential cellular processes.
Hydrolysis of ATP: Releasing Energy
ATP consists of three components:
Adenine: A nitrogenous base.
Ribose: A five-carbon sugar.
Three phosphate groups: Linked in a chain, with the last two bonds being high-energy bonds.
Note
The high-energy bonds between phosphate groups are not "high-energy" because they store a lot of energy.
Instead, they release a significant amount of energy when broken due to the instability of the negatively charged phosphate groups repelling each other.
Hydrolysis: Breaking Down ATP
The hydrolysis of ATP to ADP releases energy that cells use for work.
This process involves breaking a phosphoanhydride bond in ATP, which releases a phosphate group (Pi) and generates ADP.
Definition
HydrolysisHydrolysis is a chemical reaction where water () breaks down a compound.
When ATP undergoes hydrolysis, it splits into:
ADP (adenosine diphosphate): ATP with one less phosphate group.
Inorganic phosphate (): The detached phosphate group.
Energy: Released for cellular work.
Note
Don't confuse ATP with long-term energy storage molecules like glucose or fats.
ATP is for immediate energy use, while glucose and fats store energy for later conversion into ATP.
Tip
Think of ATP as a compressed spring.
When the spring is released (hydrolyzed), it releases stored energy, just like ATP does when its phosphate bond is broken.
Synthesis of ATP: Storing Energy
While ATP hydrolysis releases energy, the reverse process, synthesis of ATP, requiresenergy.
Condensation: Forming ATP
Definition
Condensation Condensation is a reaction where two molecules combine, releasing water.
ADP+Pi+Energy->ATP+
ATP synthesis requires an input of energy, typically derived from the breakdown of glucose in cellular respiration (e.g., in mitochondria).
To rebuild ATP from ADP and :
Energy is needed to reattach the phosphate group.
Water is released as a byproduct.
Tip
The energy required to synthesize ATP is slightly greater than the energy released during hydrolysis.
This ensures that ATP acts as a reliable energy storage molecule.
Note
The ATP <-> ADP cycle is a constant process, as cells continuously hydrolyze ATP for energy and regenerate it to maintain energy balance.
Example
In muscle cells, ATP hydrolysis powers contraction, while mitochondria regenerate ATP using energy from glucose breakdown.
Why ATP is Ideal for Energy Transfer
Small, Manageable Energy Bursts: ATP releases just enough energy for cellular processes, minimizing waste.
Reversibility: ATP can be quickly regenerated from ADP, ensuring a continuous energy supply.
Solubility: ATP is water-soluble, allowing it to move easily within cells.
Localized Control: ATP cannot diffuse out of cells, ensuring energy is used where it's needed.
Analogy
Think of ATP as a rechargeable battery.
It releases energy when needed and can be recharged (synthesized) using energy from respiration or photosynthesis.
Self Review
Why does breaking the bond between phosphate groups release energy?
Name two processes that require ATP hydrolysis.
C1.2.4 cell respiration
Cell Respiration Produces ATP
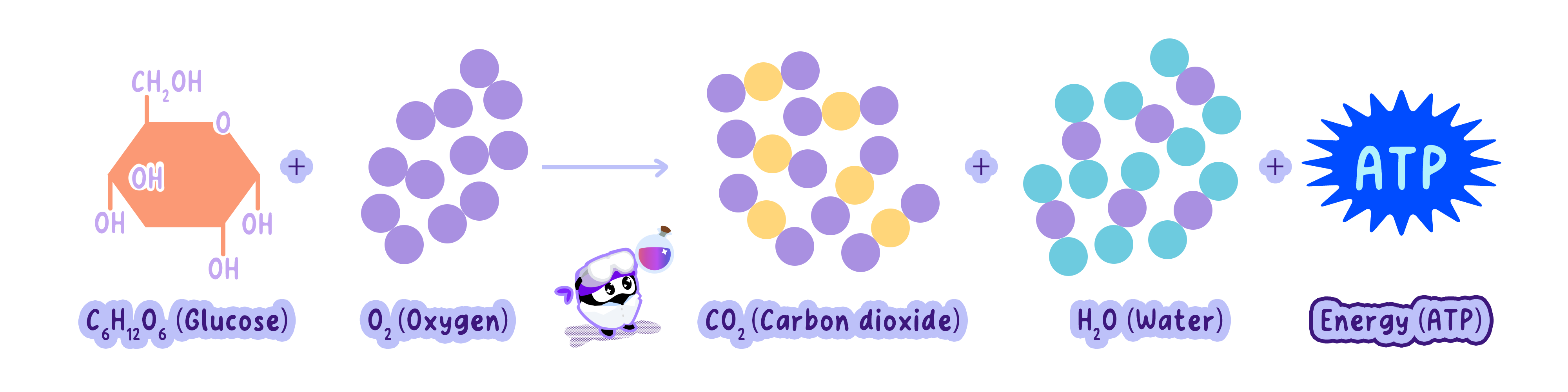
Cell respiration is the controlled release of energy from organic compounds to produce ATP, the energy currency of cells.
Cells break down carbon compounds (like glucose and fatty acids) and extract energy stored in their chemical bonds.
This energy is released through oxidation and captured in ATP molecules.
Definition
Cell respirationCell respiration is a series of metabolic processes that convert energy stored in carbon compounds, such as glucose and fatty acids, into ATP.
Tip
Oxidation involves the loss of electrons or hydrogen atoms, while reduction involves the gain of electrons or hydrogen atoms.
These reactions are coupled and form the basis of energy transfer in cells.
Main Substrates for Cell Respiration
While cells can use a wide range of organic compounds, glucose and fatty acids are the principal substrates.
Glucose
Glucose is a 6-carbon sugar and the most commonly used substrate.
It's broken down during glycolysis into two molecules of pyruvate (3C).
Pyruvate can then enter the mitochondria for complete oxidation in aerobic respiration, or be converted to other products in anaerobic respiration
Fatty Acids
Fatty acids are long carbon chains that are highly energy-dense.
They're broken down through beta-oxidation into 2-carbon acetyl groups, which form acetyl-CoA.Acetyl-CoA enters the Krebs cycle, just like the acetyl-CoA produced from glucose.
Fatty acids yield more ATP per gram than glucose because they contain more carbon-hydrogen bonds.Amino Acids
Other Carbon Compounds
Cells can also use amino acids (when glucose and fats are scarce), glycerol (from fat breakdown), and other organic molecules.
These compounds are converted into intermediates that can enter glycolysis, the link reaction, or the Krebs cycle at various points.
Hint
Cell respiration is flexible, meaning cells can use whatever carbon compounds are available to produce ATP.
Overview: How Cell Respiration Works
Cell respiration involves multiple stages that progressively extract energy from substrates.
Each stage will be explored more in depth in the succeeding articles (if you're an HL student), but here's an overview:
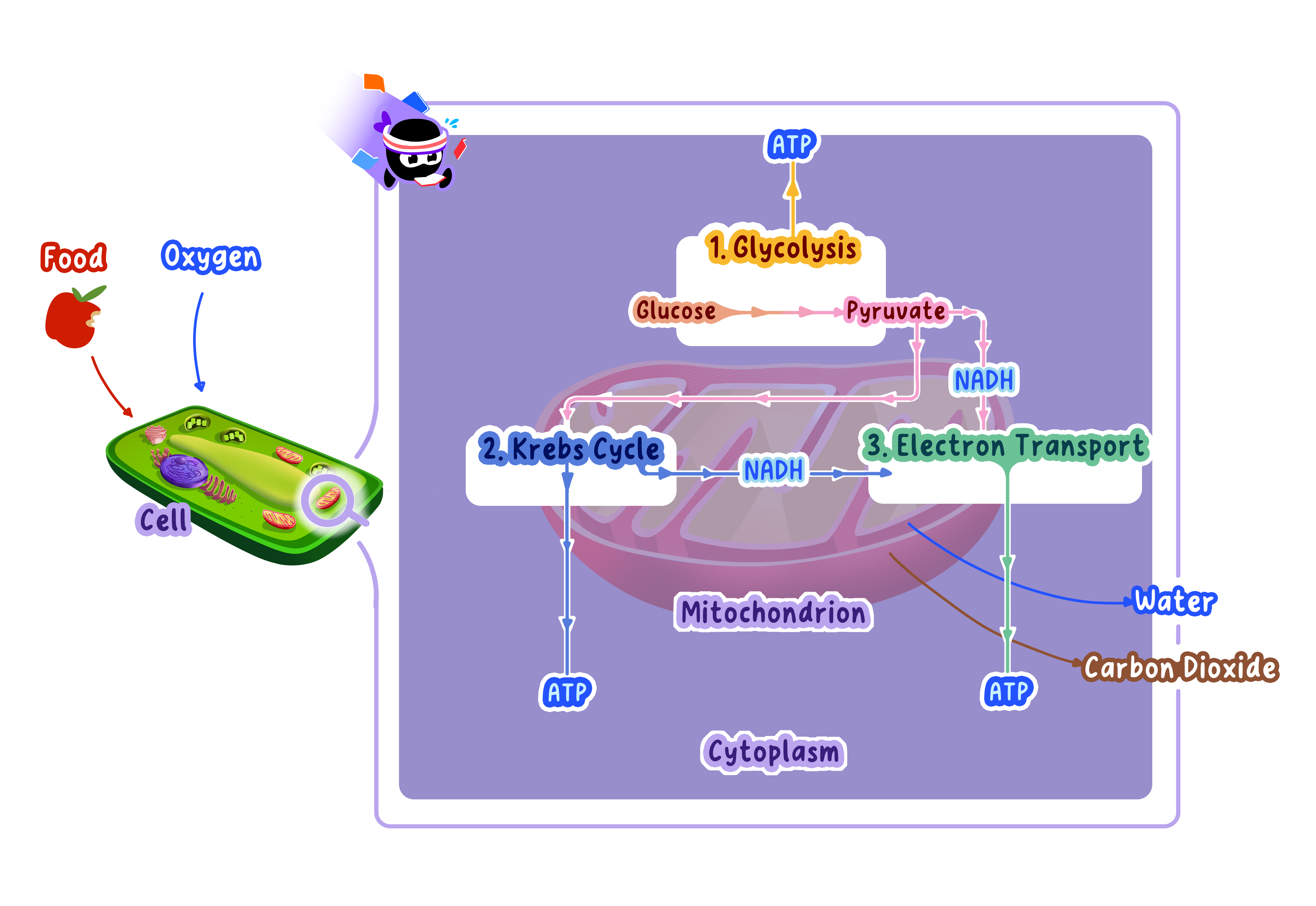
Stage 1: Glycolysis (Cytoplasm)
Glucose (6C) is broken down into two pyruvate (3C) molecules.
Produces 2 ATP (net) and 2 NADH.
Stage 2: Link Reaction (Mitochondrial Matrix)
Each pyruvate (3C) is converted into acetyl-CoA (2C).
Produces 2 NADH and 2 CO₂ per glucose.
Stage 3: Krebs Cycle (Mitochondrial Matrix)
Acetyl-CoA enters a cycle of reactions that completely oxidizes the carbon atoms.
Produces 6 NADH, 2 FADH₂, 2 ATP, and 4 CO₂ per glucose.
Stage 4: Electron Transport Chain and Oxidative Phosphorylation (Inner Mitochondrial Membrane)
NADH and FADH₂ donate electrons to the electron transport chain.
Energy from electron flow creates a proton gradient that drives ATP synthesis via chemiosmosis.
Produces approximately 34 ATP per glucose.
Oxygen acts as the final electron acceptor, forming water (H₂O).
Note
Total ATP yield per glucose: Approximately 38 ATP (though actual yields vary between 30-32 ATP in eukaryotic cells due to energy costs of transport).
Distinguishing Cell Respiration from Gas Exchange
Students often confuse these two processes, but they're distinct:
Feature | Cell respiration | Gas exchange |
|---|---|---|
Location | Inside cells (cytoplasm and mitochondria) | Across respiratory surfaces (lungs, gills, cell membranes) |
Process | Chemical breakdown of organic compounds to produce ATP | Physical movement of O₂ and CO₂ across membranes |
Purpose | Energy production | Supplying O₂ to cells and removing CO₂ waste |
Oxygen role | Used as final electron acceptor in ETC | Delivered to cells for use in respiration |
CO₂ role | Produced as waste product of carbon compound oxidation | Removed from cells and expelled from organism |
Self Review
What is cell respiration?
What are the two principal substrates for cell respiration?
Why do fatty acids yield more ATP per gram than glucose?
Name one example of another organic compound that can be used in cell respiration.
Where does glycolysis occur?
Where does the Krebs cycle occur?
What is the role of oxygen in cell respiration?
C1.2.5 differences between anaerobic and aerobic cell respiration in humans
Differences Between Anaerobic and Aerobic Cell Respiration in Humans
Cell respiration is the process by which cells convert biochemical energy from organic compounds into ATP, the energy currency of the cell.
There are two main types of cell respiration in humans: aerobic respiration and anaerobic respiration.
These processes differ primarily in the presence of oxygen, energy yield, by-products, and the location within the cell where they occur.
Analogy
Imagine sprinting up a hill.
Your muscles burn, and you're gasping for air.
What's happening inside your cells?
They're working hard to produce ATP, the energy currency of life.
But when oxygen runs low, they switch to a backup plan.
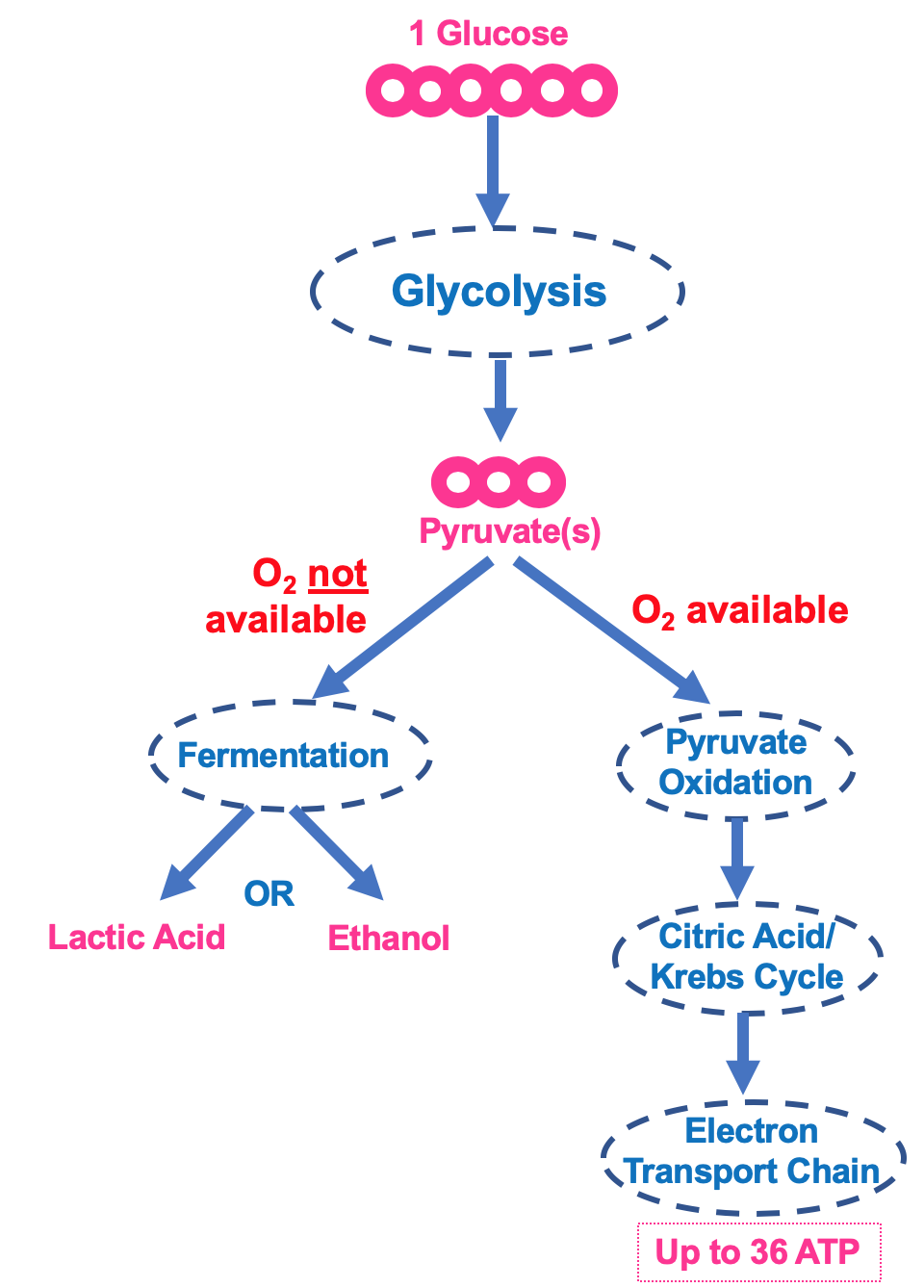
Tip
Understanding the differences between aerobic and anaerobic respiration is key to knowing how your body powers itself under different conditions.
Aerobic Respiration: The Oxygen-Powered Engine
Aerobic respiration is the primary way cells generate ATP when oxygen is available.
This process is highly efficient, producing a significant amount of ATP from each glucose molecule.
Glucose + Oxygen -> Carbon Dioxide + Water + ATP
Key Features of Aerobic Respiration
Oxygen Requirement: Aerobic respiration depends on oxygen as the final electron acceptor in the electron transport chain.
Location: Most of the process occurs in the mitochondria, specifically in the matrix (for the Krebs cycle) and across the inner mitochondrial membrane (for the electron transport chain)..
Respiratory Substrates: While glucose is the most common substrate, aerobic respiration can also use fatty acids and amino acids.
ATP Yield: For each molecule of glucose, 36-38 ATP molecules can be produced (depending on the efficiency of the cell's processes).
Waste Products: The process generates carbon dioxide and water, which are easily removed from the body.
The Process of Aerobic Respiration
Glycolysis: Glucose is broken down into pyruvate in the cytoplasm, producing a small amount of ATP and reduced NAD.
Link Reaction and Krebs Cycle: Pyruvate enters the mitochondria, where it is further broken down, releasing carbon dioxide and transferring energy to reduced NAD and FAD.
Electron Transport Chain (ETC): Electrons from reduced NAD and FAD flow through the ETC in the inner mitochondrial membrane, driving the production of ATP through chemiosmosis. Oxygen acts as the terminal electron acceptor, forming water.
Tip
Remember that aerobic respiration is the default pathway for most cells because of its high ATP yield.
It's why you breathe--to supply oxygen for this process.
Anaerobic Respiration: The Backup System
When oxygen is scarce, cells switch to anaerobic respiration to produce ATP.
This process is less efficient but provides a quick burst of energy.
Key Features of Anaerobic Respiration
Oxygen Independence: Anaerobic respiration does not require oxygen.
Location: All reactions occur in the cytoplasm, mitochondria are not involved.
Respiratory Substrates: Only glucose can be used as a substrate.
ATP Yield: Anaerobic respiration produces only 2 ATP molecules per glucose.
Waste Products: In humans, the process generates lactate (lactic acid), which can accumulate and cause muscle fatigue.
Note
Anaerobic respiration is critical during intense activities like sprinting, where oxygen delivery to muscles cannot keep up with demand.
However, it's only a short-term solution due to the low ATP yield and lactate buildup.
The Process of Anaerobic Respiration
Glycolysis: Glucose is broken down into pyruvate, producing 2 ATP molecules.
Lactate Formation: Pyruvate is converted into lactate to regenerate NAD, allowing glycolysis to continue.
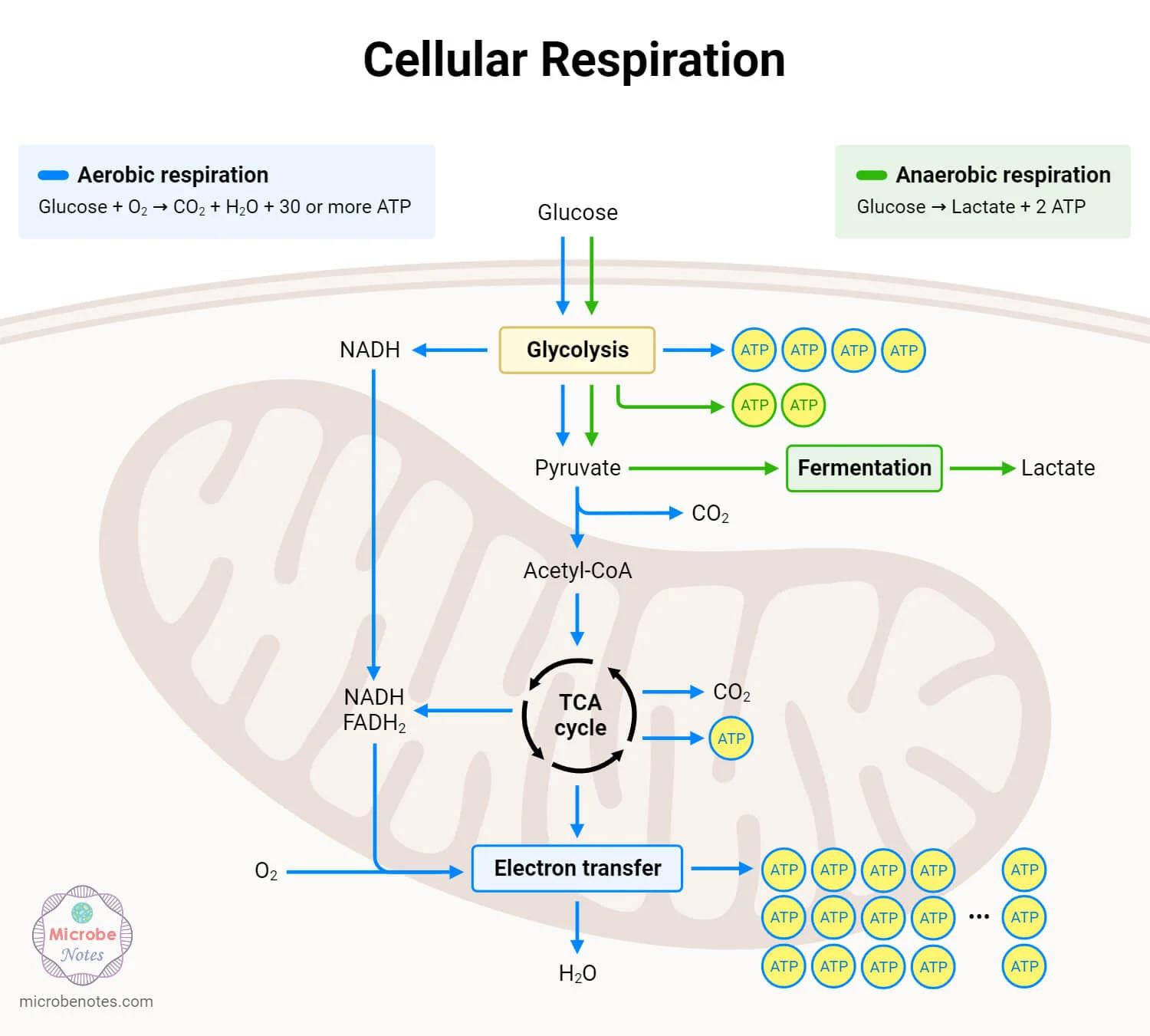
Example
Yeast uses anaerobic respiration to produce ethanol, a key step in brewing and baking.
Comparing Aerobic and Anaerobic Respiration
Feature | Aerobic respiration | Anaerobic respiration |
|---|---|---|
Oxygen Requirement | Requires oxygen | Does not requires oxygen |
Location | Mitochondria (Krebs cycle, electron transport chain) | Cytoplasm (Glycolysis and fermentation) |
Substrate | Glucose, fatty acids, amino acids | Glucose |
ATP Yield | High (36-38 ATP per glucose molecule) | Low (2 ATP per glucose molecule) |
By-Products | Carbon dioxide and water | Lactic acid (humans) or ethanol and CO₂ (yeast) |
Warning
Don't confuse anaerobic respiration with fermentation in yeast.
While both are anaerobic processes, yeast produces ethanol and carbon dioxide, not lactate.
Why Do Cells Use Both Pathways?
Aerobic respiration is the preferred method because of its high ATP yield.
However, anaerobic respiration provides a rapid energy boost when oxygen is limited, such as during intense exercise.
Analogy
Aerobic respiration is like a high-efficiency power plant using a renewable resource (oxygen) to produce large amounts of energy.
Anaerobic respiration is like a backup generator that can run without oxygen, but it produces much less energy and has a "cost" in the form of waste products like lactic acid.
Self Review
What are the main differences in ATP production between aerobic and anaerobic respiration?
Where does anaerobic respiration occur in the cell, and how does it differ from aerobic respiration in terms of location?
C1.2.6 variables affecting the rate of cell respiration
1. Temperature
Cell respiration relies on enzymes, which work best at optimal temperatures.
Higher temperatures increase enzyme activity, speeding up respiration.
However, if the temperature exceeds the optimum temperature, enzymes can denature and stop working.
Example
During intense exercise, muscles may not get enough oxygen, leading to anaerobic respiration and the production of lactate.
2. Type of Respiratory Substrate
Different substrates (e.g., glucose, lipids) yield varying amounts of energy.
Carbohydrates are often used first because they are easy to break down.
Lipids provide more energy per gram but are metabolized more slowly.
Tip
Lipids yield more ATP than carbohydrates, but they require more oxygen to break down.
3. Oxygen Availability
Oxygen is the final electron acceptor in the electron transport chain of aerobic respiration.
Limited oxygen forces cells to switch to anaerobic respiration, which is less efficient and produces much less ATP.
Animal cells produce lactate under low oxygen, while yeast produces ethanol and carbon dioxide.
4. Substrate Concentration
Higher concentrations of glucose or other substrates can increase the rate of respiration, up to a point.
Once enzymes are saturated, adding more substrate won't increase the rate further.
5. pH Levels
Enzymes involved in respiration have an optimal pH range.
Deviations from this range can slow down or halt the process.
Example
The Krebs cycle enzymes work best at a slightly alkaline pH (~7.4).
Note
Don't confuse aerobic and anaerobic respiration.
Aerobic respiration requires oxygen and produces more ATP, while anaerobic respiration occurs without oxygen and yields less ATP.
Measuring the Rate of Cell Respiration
1. Oxygen Uptake
Respirometers measure oxygen consumption, which reflects the rate of aerobic respiration.
A typical respirometer setup includes:
A sealed chamber with the organism or tissue.
An alkali (e.g., potassium hydroxide) to absorb carbon dioxide.
A capillary tube with fluid to measure changes in air volume, indicating oxygen uptake.
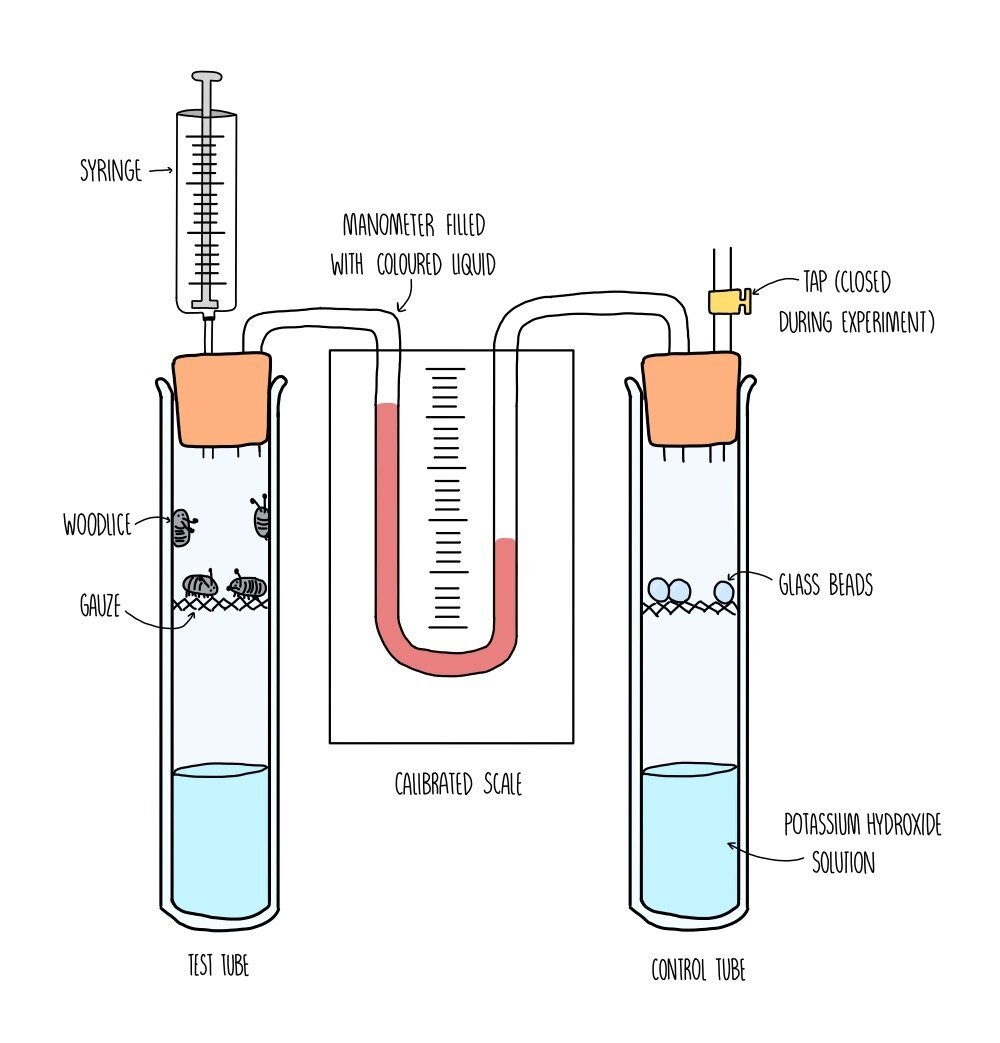
Note
The movement of fluid in the capillary tube shows oxygen consumption.
Carbon dioxide is absorbed, so volume changes are due to oxygen use alone.
2. Carbon Dioxide Production
During aerobic respiration, glucose is oxidized into carbon dioxide and water.
The amount of carbon dioxide produced can be measured with a gas sensor or by using acolor indicator in a closed system.
3. Substrate Consumption
By measuring the decrease in glucose or other substrates over time, you can infer the rate of respiration.
Here, other variables like temperature and pressure must always be controlled to ensure accurate results.
Designing Experiments to Investigate Variables
Respirometers are versatile tools for exploring how different factors affect respiration. Here are some experimental ideas:
Temperature: Compare respiration rates at different temperatures using a water bath to maintain constant conditions.
Substrate Type: Investigate whether yeast respires faster with glucose or sucrose.
Oxygen Availability: Measure respiration rates in aerobic versus anaerobic conditions.
Warning
In respiration experiments, failing to control variables like pH or glucose concentration can lead to inaccurate results.
Note
Ensure ethical treatment of organisms in experiments.
Avoid causing harm or stress to living subjects.
Calculating the Rate of Cell Respiration
To calculate the rate of cell respiration from experimental data, you need to:
Determine the amount of oxygen consumed or CO₂ produced.
Divide the change in oxygen or CO₂ concentration by the time to calculate the rate.
Example
If 0.5 mL of oxygen is consumed over a period of 5 minutes, the rate of respiration is:
Rate = 0.5 mL / 5 minutes = 0.1 mL/min
Self Review
How does temperature affect the rate of cell respiration?
What happens to the rate of respiration when oxygen is limited?
C1.2.7 role of nad as a carrier of hydrogen (hl)
NAD Acts As A Hydrogen Carrier in Cell Respiration
NAD (nicotinamide adenine dinucleotide) is a key molecule in cellular respiration.
It transfers electrons and hydrogen atoms between reactions, allowing cells to capture and use energy efficiently.
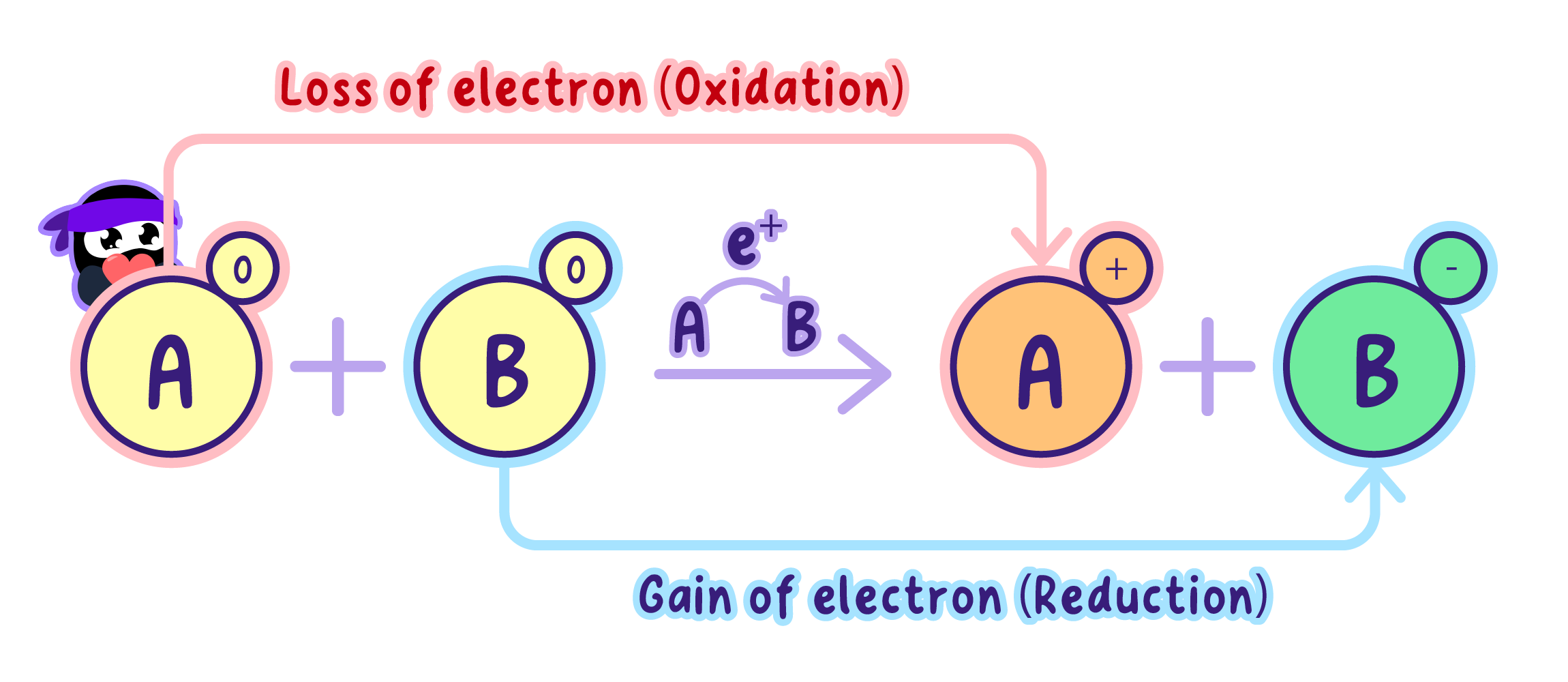
What is Oxidation and Reduction?
Before diving into NAD's role, let's clarify two essential concepts: oxidation and reduction.
Oxidation: The loss of electrons (or hydrogen atoms) from a molecule.
Reduction: The gain of electrons (or hydrogen atoms) by a molecule.
Analogy
OIL RIG, Oxidation Is Loss, Reduction Is Gain.
Note
These reactions always occur together, forming redox reactions.
NAD⁺ acts as an electron sink, capturing high-energy electrons during metabolic reactions.
NAD in Cell Respiration
NAD is a molecule that can carry electrons and hydrogen atoms from one molecule to another.
This is important in processes like cell respiration, where energy stored in glucose is released step by step.
In the first part of cell respiration, glycolysis and the Krebs cycle, NAD helps remove hydrogen from substrates (like glucose or fatty acids).
When NAD removes hydrogen, it becomes reduced (NAD+ becomes NADH), and the substrate it took the hydrogen from is oxidized.
Note
NAD+ is a coenzyme, meaning it helps enzymes but is not consumed in the process.
Once it becomes NADH, it can later release its electrons to help produce ATP.
Warning
NAD+ does not provide energy directly.
It carries energy in the form of electrons and hydrogen.
1. Glycolysis
Occurs in the cytoplasm.
Glucose is split into two molecules of pyruvate.
NAD⁺ is reduced to NADH as it accepts hydrogen atoms from glucose intermediates.
Example
In glycolysis, the oxidation of triose phosphate produces NADH, which is later used to generate ATP.
2. The Link Reaction and Krebs Cycle
Takes place in the mitochondrial matrix.
Pyruvate is converted to acetyl-CoA, then enters the Krebs cycle.
NAD⁺ is reduced to NADH multiple times, capturing high-energy electrons.
3. The Electron Transport Chain (ETC)
Located in the inner mitochondrial membrane.
NADH donates electrons to the ETC, returning to its oxidized form, NAD⁺.
The energy from these electrons is used to pump protons, creating a gradient that drives ATP synthesis.
Warning
Don't confuse NAD⁺ with NADH.
NAD⁺is the empty carrier, while NADHis the loaded carrier, ready to deliver electrons.
Analogy
Imagine NAD+ as a taxi that picks up passengers (electrons and hydrogen ions) and takes them to a molecular train station (the electron transport chain) where the energy from the passengers is used to generate power for the cell.
NAD+ is the empty taxi.
When it picks up the passengers (electrons and hydrogen), it becomes NADH (the full taxi).
The NADH then drives to the electron transport chain, drops off the passengers, and the cell can use the energy from those passengers (electrons) to produce ATP.
Tok
How does the concept of oxidation and reduction in biology connect to other fields, such as chemistry or physics?
Can you think of other systems where energy transfer is critical?
C1.2.8 conversion of glucose to pyruvate (hl)
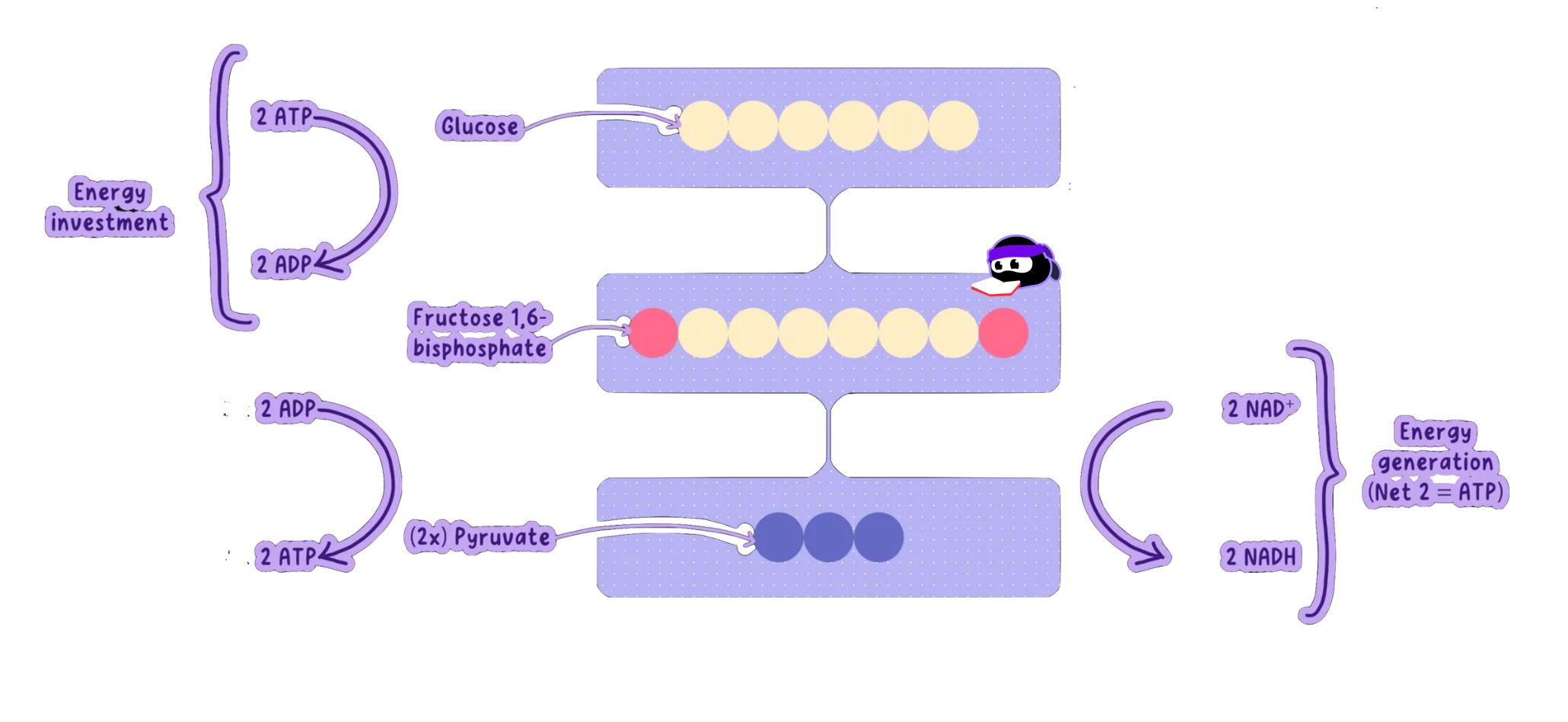
Glycolysis: The First Step in Cellular Respiration
Glycolysis breaks down one molecule of glucose (6C) into two molecules of pyruvate (3C).
It occurs in the cytoplasm and does not require oxygen, making it an anaerobic process.
It produces a small amount of ATP and NADH, which are used in later stages of cellular respiration.
Definition
GlycolysisThe metabolic pathway that breaks down one molecule of glucose (6C) into two molecules of pyruvate (3C), producing a net gain of 2 ATP and 2 NADH. It occurs in the cytoplasm and does not require oxygen.
Note
Each step in glycolysis is catalyzed by a specific enzyme, ensuring the process is efficient and tightly regulated.
Step-by-Step of Glycolysis
Glycolysis involves a series of enzyme-catalyzed reactions that can be divided into fourkey stages.
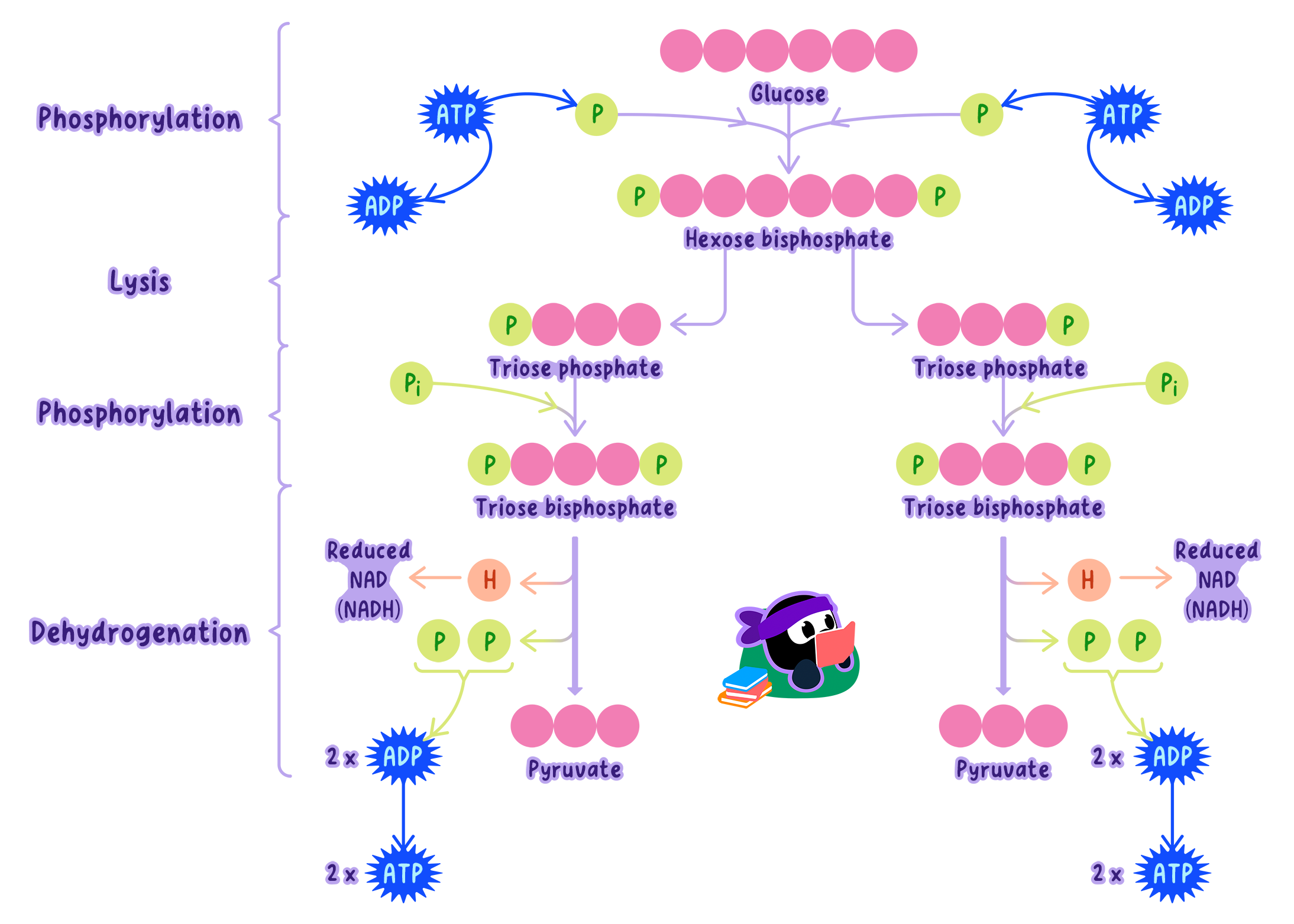
Stage 1: Phosphorylation (Energy Investment)
Definition
PhosphorylationPhosphorylation refers to the addition of a phosphate group to a molecule
Glucose is phosphorylated, meaning a phosphate group is added to it.
The enzyme hexokinase adds the first phosphate, forming glucose-6-phosphate.
A second phosphate is added in a subsequent step, forming fructose-1,6-bisphosphate.
This requires 2 ATP to be used (energy investment).
Tip
Adding phosphate groups makes glucose more reactive and traps it inside the cell (phosphorylated glucose can't cross the membrane).
Think of it as "priming" the molecule for breakdown.
Stage 2: Lysis (Splitting the Sugar)
The 6-carbon sugar (fructose-1,6-bisphosphate) is split into two 3-carbon moleculescalled triose phosphates.
Each triose phosphate is converted into glyceraldehyde-3-phosphate (G3P).
Analogy
Imagine cutting a log into smaller pieces before burning it for energy.
Lysis is like chopping the glucose molecule into manageable parts.
Note
From this point forward, every reaction happens twice per glucose, once for each G3P molecule.
Stage 3: Oxidation
Each G3P is oxidized, hydrogen atoms (with their electrons) are removed.
The hydrogens are transferred to NAD⁺, reducing it to NADH.
Simultaneously, an inorganic phosphate (Pi) is added to the molecule.
Warning
Don't confuse NAD+ with NADH.
NAD+ is the oxidized form, while NADH is the reduced form that carries electrons.
Stage 4: ATP Formation (Energy Payoff)
The remaining reactions convert the 3-carbon molecules into pyruvate (3C).
During these steps, phosphate groups are transferred directly from intermediates to ADP, forming ATP.
This is called substrate-level phosphorylation (direct transfer of phosphate, not using the ETC).
Example
If you invest USD 2 in a project and earn USD 4 in return, your net profit is $2. Same logic here.
Products of Glycolysis (Per Glucose)
Starting with one glucose molecule, glycolysis produces:
2 pyruvate (3C) (one from each G3P)
2 ATP (net) (4 produced − 2 used)
2 NADH (one from each G3P oxidation)
Note
Glycolysis is the universal first step of cellular respiration, it occurs in nearly all living organisms.
It works without oxygen, so cells can extract some energy even in anaerobic conditions.
The pyruvate, ATP, and NADH produced are essential for the later stages of aerobic respiration, which produce the bulk of cellular ATP.
Tip
These products are the starting materials for the next stages of cellular respiration (link reaction and Krebs cycle in aerobic conditions, or fermentation in anaerobic conditions).
Self Review
Where does glycolysis occur in the cell?
Does glycolysis require oxygen?
What is phosphorylation and why is it necessary in glycolysis?
How many ATP are used in the phosphorylation stage?
What does "lysis" mean in the context of glycolysis?
How many ATP are produced in total during glycolysis?
What is the net ATP yield per glucose and why?
How many NADH are produced per glucose?
What are the three end products of glycolysis per glucose molecule?
C1.2.9 conversion of pyruvate to lactate (hl)
Pyruvate is Converted to Lactate to Regenerate NAD⁺ in Anaerobic Respiration
Glycolysis requires NAD⁺ to continue, but cells have a limited supply.Without oxygen, the electron transport chain can't regenerate NAD⁺, so glycolysis would stop.
To solve this, pyruvate acts as an electron acceptor, allowing NAD⁺ to be recycled.
Analogy
Think of glycolysis as a factory assembly line producing ATP.
NAD acts like a conveyor belt, carrying electrons to keep the line moving.
Without oxygen, the conveyor belt gets stuck unless lactate production steps in to clear the jam.
Lactic Acid Fermentation Converts Pyruvate to Lactate and Regenerates NAD⁺
The enzyme lactate dehydrogenase catalyzes the conversion of pyruvate to lactate.
NADH donates its electrons to pyruvate, oxidizing NADH back to NAD⁺.
Reaction: Pyruvate + NADH + H⁺ -> Lactate + NAD⁺
This does not produce ATP directly--its sole purpose is to regenerate NAD⁺ so glycolysis can keep running.
Pyruvate + NADH + H⁺ -> Lactate + NAD⁺
This reaction has two important outcomes:
NAD⁺ is regenerated, allowing glycolysis to continue and produce ATP.
Lactate (lactic acid) is produced as a waste product.
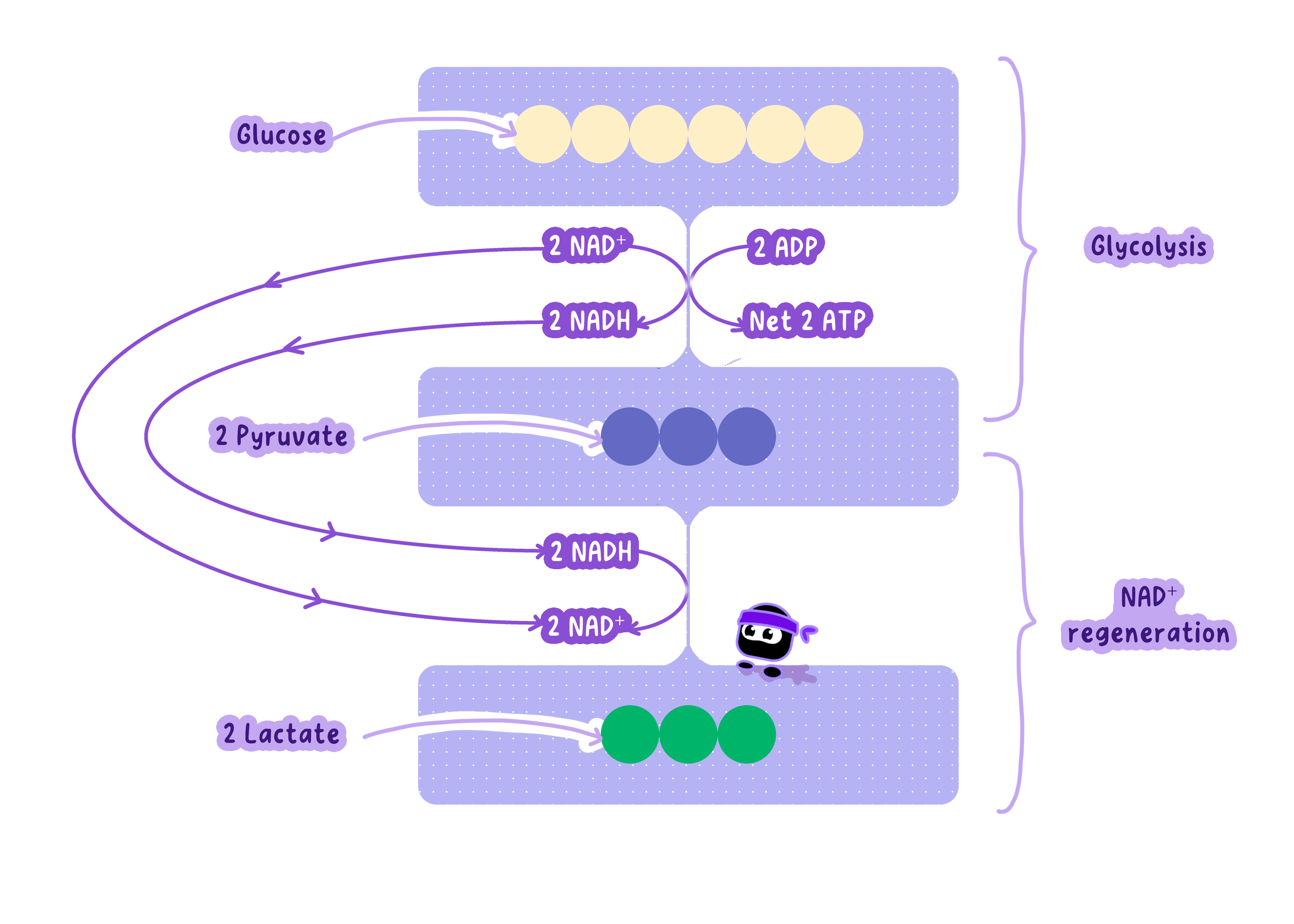
Lactate Is a Temporary Solution To Keep ATP Production Going
Lactate accumulates in muscles during intense exercise, causing a pH drop and muscle fatigue.
Once oxygen is available, lactate is converted back to pyruvate or transported to the liver for glucose synthesis.
Note
Anaerobic respiration only yields 2 ATP per glucose (from glycolysis alone), far less efficient than aerobic respiration's 30+ ATP.
Tok
How does the concept of "oxygen debt" during intense exercise relate to the broader idea of resource management in biology and beyond?
Can you think of other systems where temporary solutions are used to address immediate needs?
Self Review
Why is the regeneration of NAD essential for glycolysis to continue?
What happens to lactate after anaerobic respiration?
C1.2.10 anaerobic cell respiration in yeast and its use in brewing and baking (hl)
Yeast Uses Alcoholic Fermentation to Produce Ethanol and CO₂
Yeast regenerates NAD⁺ by converting pyruvate into ethanol and CO₂, not lactate like humans.
This process is called alcoholic fermentation.
The Two-Step Process
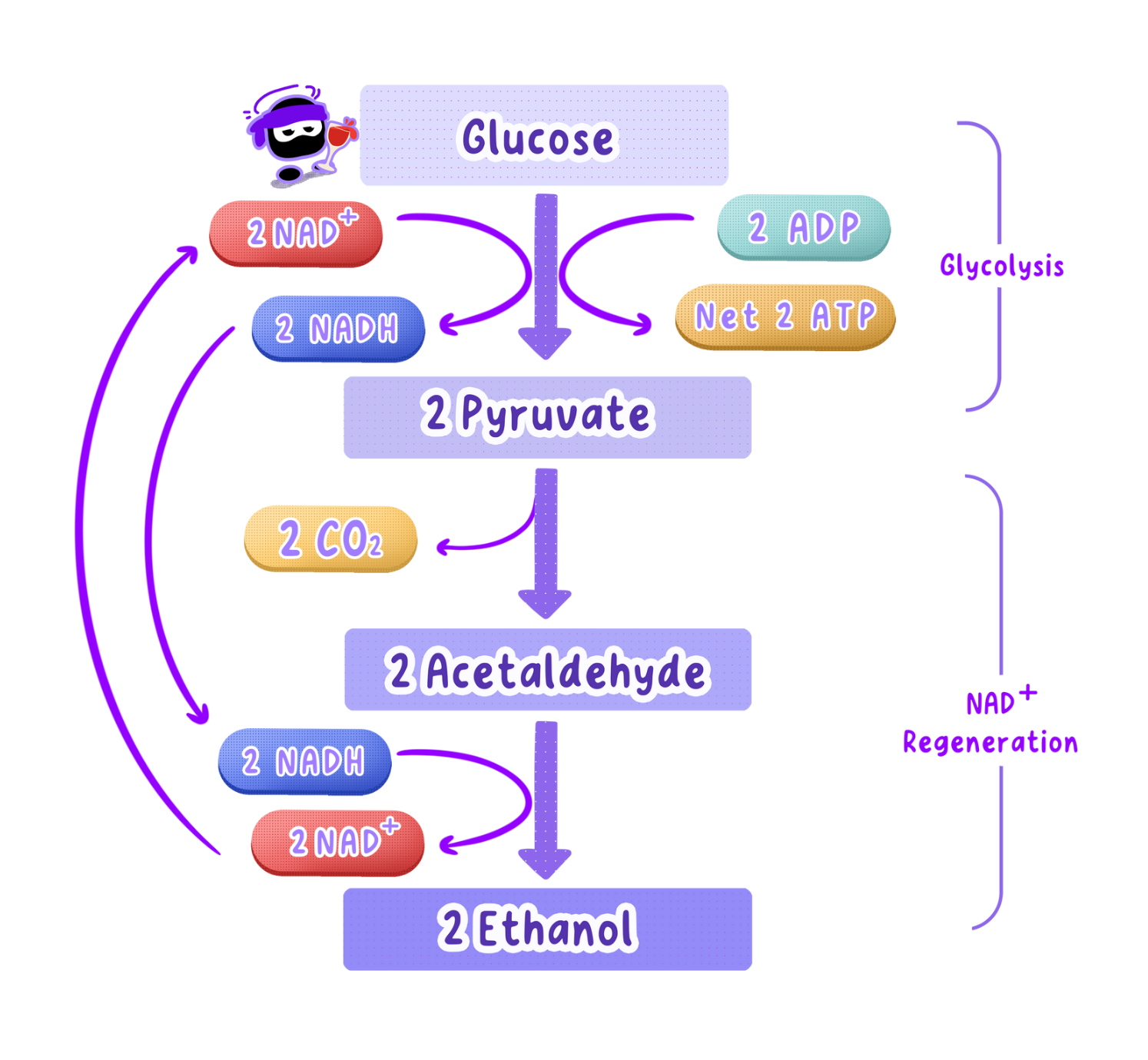
Decarboxylation of Pyruvate: Pyruvate loses a carbon atom, releasing carbon dioxide and forming ethanal (acetaldehyde).
Reduction of Ethanal: Ethanal is reduced to ethanol by accepting hydrogen from NADH, regenerating NAD.
Warning
Don't confuse anaerobic respiration with fermentation.
Fermentation refers to the broader process of energy production in the absence of oxygen, while anaerobic respiration specifically describes the metabolic pathway.
Commercial Applications
Brewing
Yeast ferments sugars in grape juice (wine) or malted barley (beer) under anaerobicconditions, producing ethanol and CO₂.
Fermentation stops when ethanol concentration reaches ~15% (toxic to yeast).
Baking
CO₂ produced by yeast gets trapped in dough, causing it to rise.
The ethanol evaporates during baking, leaving no trace in bread.
Note
The ethanol produced during baking evaporates completely, leaving no trace in the finished bread.
Yeast vs. Humans
Both use glycolysis to break down glucose.
Yeast: Pyruvate -> Ethanol + CO₂
Humans: Pyruvate -> Lactate
Tip
Humans produce lactate, while yeast produces ethanol and CO₂.
Self Review
What are the final products of anaerobic respiration in yeast?
Why is NAD⁺ regeneration important in anaerobic respiration?
How is anaerobic respiration used in baking?
Why is the ATP yield from anaerobic respiration lower than from aerobic respiration?
C1.2.11 oxidation and decarboxylation of pyruvate (hl)
The Link Reaction Connects Glycolysis to the Krebs Cycle
Glycolysis (in the cytoplasm) breaks down glucose into pyruvate (3C).
The Krebs cycle (in the mitochondrial matrix) processes acetyl groups (2C).
There's a mismatch: pyruvate has 3 carbons, but the Krebs cycle needs 2-carbon acetyl groups.
The link reaction bridges this gap by converting pyruvate (3C) into a 2-carbon acetyl group by removing one carbon and some hydrogens.
The acetyl group is then attached to coenzyme A, forming acetyl-CoA.
Analogy
Think of the link reaction as a bridge connecting two cities: glycolysis in the cytoplasm and the Krebs cycle in the mitochondrial matrix.
Without this bridge, energy flow would be interrupted.
Hint
Acetyl-CoA eventually carries the acetyl group into the Krebs cycle (more in C1.2.14)
Step-by-Step: How the Link Reaction Works
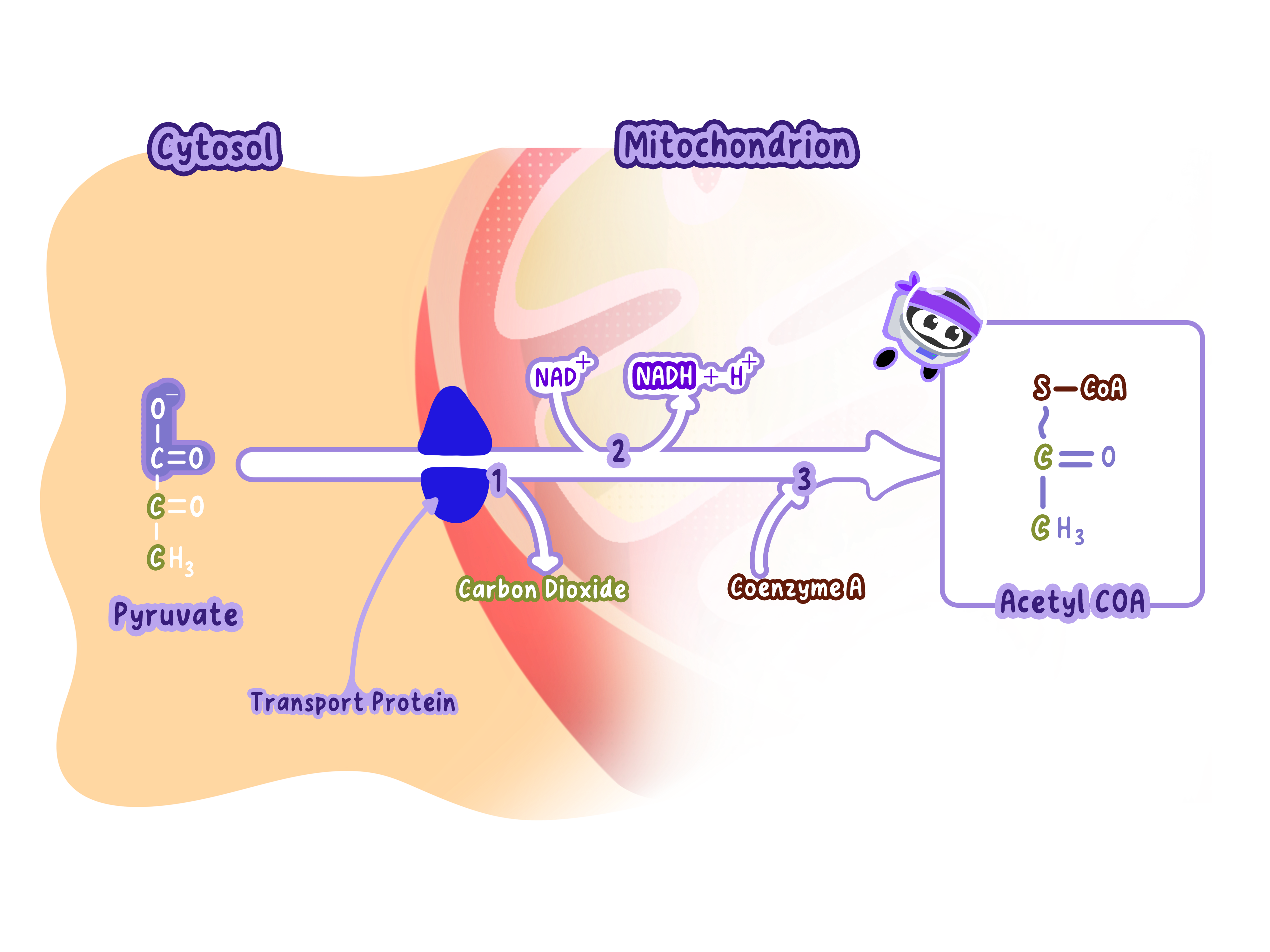
Step 1: Decarboxylation (Removing Carbon)
One carbon atom is removed from pyruvate (3C) as carbon dioxide (CO₂).
This reduces pyruvate from 3 carbons to 2 carbons.
Step 2: Oxidation (Removing Hydrogen)
Hydrogen atoms are removed from the 2-carbon molecule.
These hydrogens (with their electrons) are transferred to NAD⁺, reducing it to NADH.
Step 3: Acetyl Group Attached to Coenzyme A
The remaining 2-carbon acetyl group is attached to coenzyme A (CoA).
This forms acetyl-CoA.
Coenzyme A acts as a carrier, transporting the acetyl group to the Krebs cycle.
Tip
The Link Reaction occurs twice per glucose molecule, since each glucose molecule produces two pyruvate molecules.
Both Carbohydrates and Lipids Produce Acetyl Groups
Carbohydrates are broken down through glycolysis -> pyruvate -> link reaction -> acetyl-CoA.
Lipids (fatty acids) are broken down through beta-oxidation directly into acetyl-CoA, bypassing glycolysis and the link reaction.
Both pathways converge at acetyl-CoA, which enters the Krebs cycle.
Note
Regardless of whether the original substrate is carbohydrate or lipid, the Krebs cycle always processes 2-carbon acetyl groups carried by coenzyme A.
Self Review
Where does the link reaction occur?
What molecule accepts the hydrogens removed during oxidation?
What is the role of coenzyme A?
How many times does the link reaction occur per glucose molecule?
How do lipids enter the pathway that leads to the Krebs cycle?
Why is the link reaction called a "link"?
C1.2.12 oxidation and decarboxylation of acetyl groups in the krebs cycle (hl)
From The Link Reaction To The Krebs Cycle
In the link reaction (covered in C1.2.11), pyruvate is oxidized and decarboxylated to produce acetyl-CoA.
Acetyl-CoA carries a 2-carbon acetyl group that still contains significant chemical energy.
The Krebs cycle is where this acetyl group is completely broken down to extract that energy.
Definition
The Krebs cycle The Krebs cycle (also known as the citric acid cycle) occurs in the mitochondrial matrixand is a key part of aerobic respiration.
Step-by-Step: How the Krebs Cycle Works

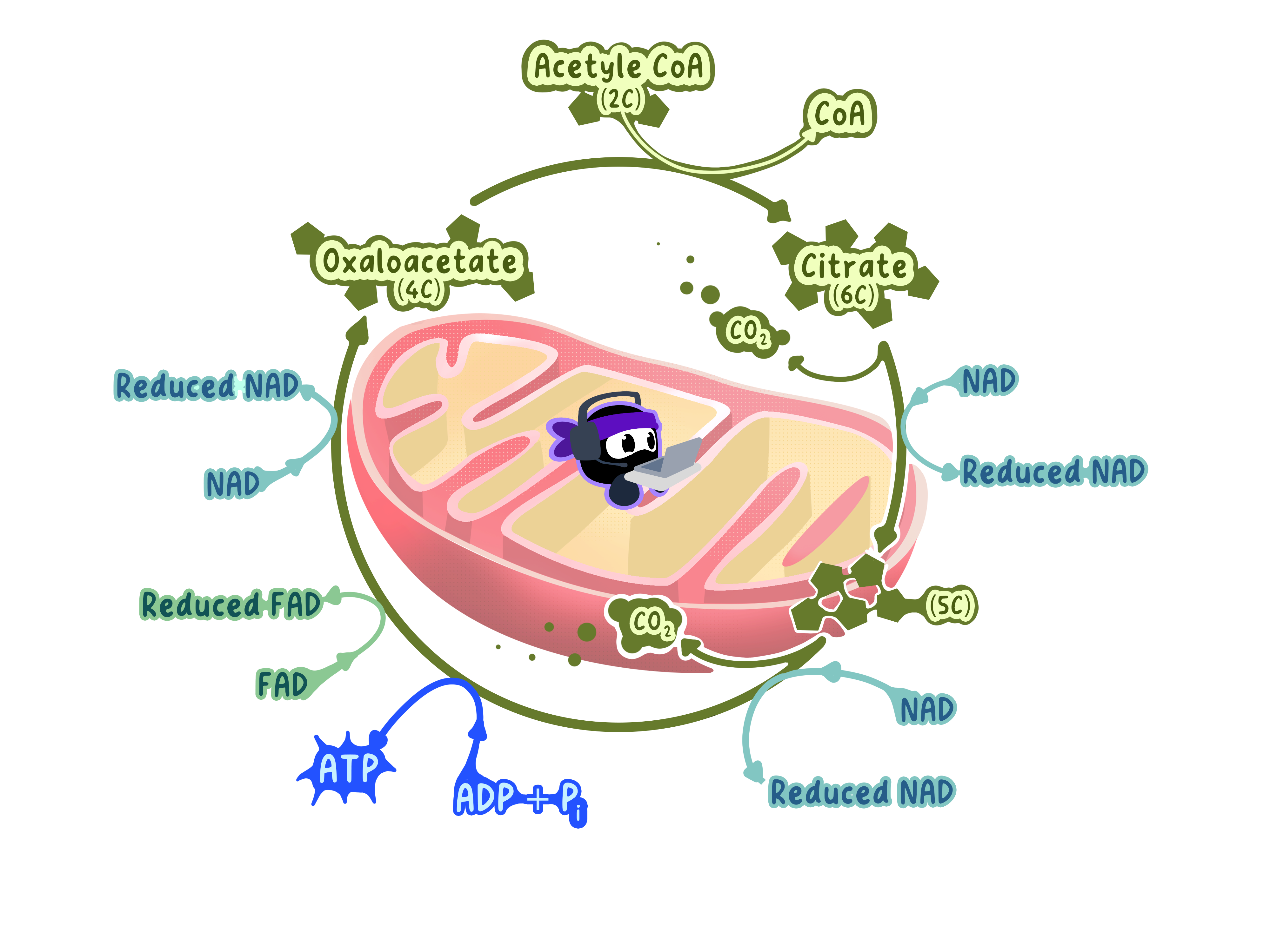
Step 1: Acetyl Group Enters the Cycle
The acetyl group (2C) from acetyl-CoA is transferred to a 4-carbon molecule called oxaloacetate.
This forms a 6-carbon molecule called citrate.
Coenzyme A is released and recycled back to the link reaction.
Hint
2C (acetyl) + 4C (oxaloacetate) = 6C (citrate)
Exam_technique
You only need to remember two molecules for the exam: citrate (6C) and oxaloacetate (4C).
Step 2: Removing Hydrogens (Oxidation/Dehydrogenation)
As citrate moves through the cycle, hydrogen atoms are systematically removed.
Each hydrogen atom consists of a proton (H⁺) and an electron (e⁻).
These hydrogens are transferred to carrier molecules:
NAD⁺ accepts hydrogens and becomes NADH (happens 3 times).
FAD accepts hydrogens and becomes FADH₂ (happens 1 time).
Hint
Total oxidations per cycle: 4 (3 produce NADH, 1 produces FADH₂)
Step 3: Removing Carbons (Decarboxylation)
The cycle removes carbon atoms as carbon dioxide (CO₂).
This happens twice during the cycle.
Each decarboxylation releases one carbon as waste.
Hint
The carbon count drops: 6C (citrate) -> loses 1C (first decarboxylation) -> 5C intermediate -> loses 1C (second decarboxylation) -> 4C
Note
The two carbons from the original acetyl group are released as waste.
They leave the mitochondria, enter the bloodstream, and are exhaled.
Step 4: Direct ATP Production
During one step of the cycle, 1 ATP (or GTP) is produced directly.
A phosphate group is transferred from an intermediate to ADP, forming ATP.
This is called substrate-level phosphorylation.
Step 5: Oxaloacetate is Regenerated
After four oxidations and two decarboxylations, the molecule is back to 4 carbons.
This is oxaloacetate (4C), the same molecule from Step 1.
Oxaloacetate can now accept another acetyl group and restart the cycle.
Note
The cycle is self-renewing.
Oxaloacetate isn't consumed but regenerated, allowing the cycle to continue indefinitely as long as acetyl-CoA is available.
Summary of Products Per Acetyl Group
Each acetyl group that goes through the Krebs cycle produces:
3 NADH (from Step 2: oxidations)
1 FADH₂ (from Step 2: oxidations)
1 ATP (from Step 4: substrate-level phosphorylation)
2 CO₂ (from Step 3: decarboxylations--waste)
Exam_technique
Remember that one glucose produces 2 acetyl-CoA in the link reaction, so the cycle runs twice per glucose.
Multiply all products by 2 for totals per glucose.
Self Review
What enters the Krebs cycle from the link reaction?
What is formed when the acetyl group combines with oxaloacetate?
How many oxidations occur per turn of the Krebs cycle?
How many decarboxylations occur per turn of the Krebs cycle?
What happens to the 2 carbons from the acetyl group?
Why can the Krebs cycle keep running indefinitely?
What is the main product of the Krebs cycle that fuels ATP production?
C1.2.13 transfer of energy by reduced nad (hl)
NADH Carries High-Energy Electrons
NADH is the link between the earlier stages of respiration (glycolysis, link reaction, Krebs cycle) and the ETC.
By transferring electrons to the ETC, NADH enables the production of most of the ATP in aerobic respiration.
Hint
The oxidation of NADH back to NAD⁺ ensures the entire process can continue running.
Energy Transfer When Electrons Are Donated
NADH donates a pair of high-energy electrons to the first carrier protein in the ETC.
When the electrons are passed to the carrier, energy is transferred from NADH to the ETC.
This electron donation oxidizes NADH back to NAD⁺.
The key reaction is: NADH -> NAD⁺ + 2e⁻ (donated to ETC)
Tip
Oxidation means losing electrons.
When NADH loses electrons, it becomes NAD⁺.
Why Regenerating NAD⁺ Matters
NAD⁺ is essential as an electron acceptor in glycolysis, the link reaction, and the Krebs cycle.
If NADH cannot be oxidized back to NAD⁺, these earlier pathways stop functioning because there's no NAD⁺ available to accept electrons.
The ETC continuously regenerates NAD⁺ by accepting electrons from NADH, allowing respiration to continue.
Note
The transfer of electrons to the ETC is also about recycling NAD⁺ so that glycolysis and the Krebs cycle can keep running.
Sources of Reduced NAD
Glycolysis (Cytoplasm)
Glucose is broken down into pyruvate.
Produces 2 NADH per glucose molecule.
This NADH must be transported into the mitochondria to donate electrons to the ETC.
Link Reaction (Mitochondrial Matrix)
Pyruvate is converted into acetyl-CoA.
Produces 1 NADH per pyruvate (2 NADH per glucose, since one glucose produces 2 pyruvate).
This NADH is already in the mitochondrial matrix, close to the ETC.
Krebs Cycle (Mitochondrial Matrix)
Acetyl-CoA enters the cycle and is fully oxidized.
Produces 3 NADH per acetyl-CoA (6 NADH per glucose).
This NADH is already in the mitochondrial matrix.
Hint
For every glucose molecule that undergoes aerobic respiration, a total of 10 NADHmolecules are produced (2 from glycolysis + 2 from link reaction + 6 from Krebs cycle).
All of these NADH molecules will donate their electrons to the ETC.
Exam_technique
Remember where NADH comes from using "GLK": Glycolysis, Link reaction, Krebs cycle.
Self Review
What does NADH donate to the electron transport chain?
What happens to NADH when it donates electrons?
Why is NAD⁺ important for cellular respiration?
Name the three stages of respiration that produce NADH.
How many NADH molecules are produced per glucose during glycolysis?
Where in the cell does each stage produce NADH?
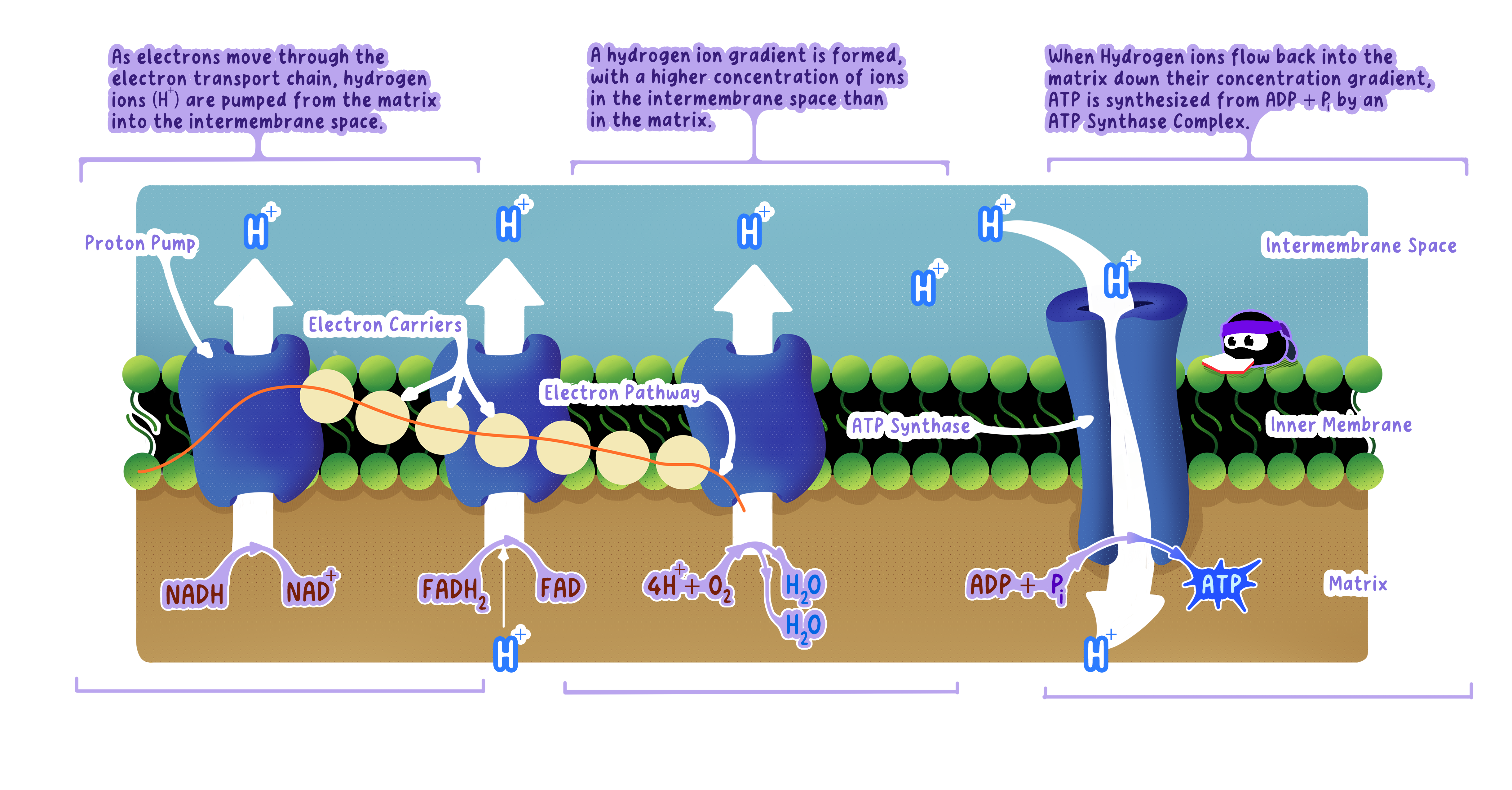
C1.2.14 generation of a proton gradient (hl)
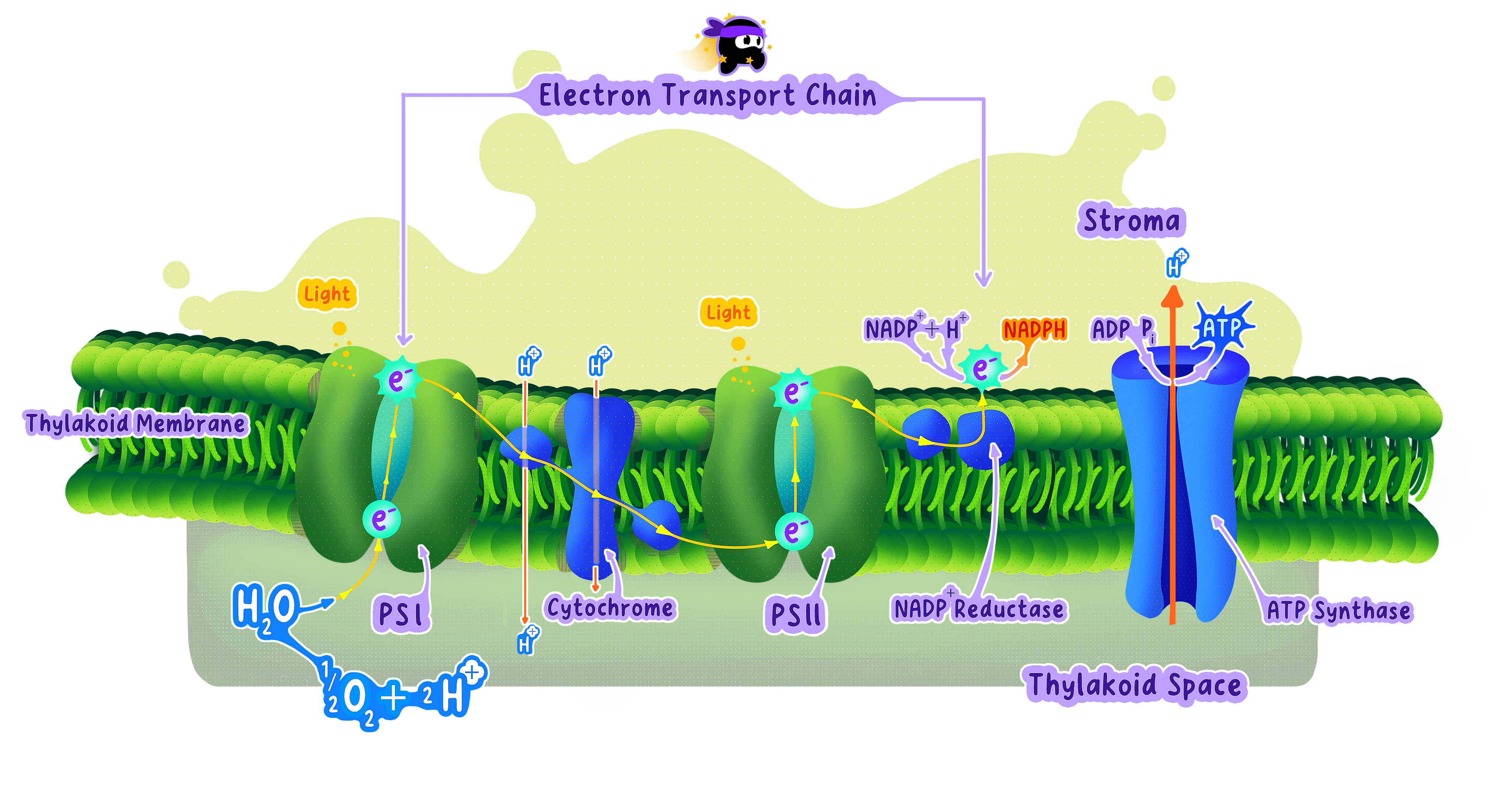
The Electron Transport Chain: A Series of Carriers
Definition
Electron transport chain (ETC) A series of protein complexes embedded in the inner mitochondrial membrane.
After NADH and FADH₂ donate electrons to the ETC (covered in C1.2.13), those electrons flow through these carriers in sequence.

Tip
Electrons don't release all their energy at once.
The stepwise transfer through multiple carriers allows the cell to capture energy efficiently at each stage, rather than losing it all as heat.
Step-by-Step: How Electron Flow Generates The Proton Gradient
The electron transport chain doesn't works as a sequence of controlled transfers that efficiently extracts energy from electrons.
Step 1: Electrons Enter the Chain
NADH donates electrons to the first carrier protein in the ETC.
FADH₂ donates electrons to a carrier further down the chain.
Hint
FADH₂ enters later in the chain than NADH, so it releases less energy overall.
This is why FADH₂ results in fewer protons being pumped and less ATP produced compared to NADH.
Step 2: Electrons Move Through Carriers
Electrons pass from one protein complex to the next.
Each transfer involves a redox reaction, one carrier is reduced (gains electrons), then oxidized (passes electrons to the next carrier).
As electrons pass from carrier to carrier, they move to lower energy levels.
Step 3: Energy is Released
Energy is released at each transfer step as electrons drop to lower energy levels.
This energy is captured by the protein complexes, it's not lost as heat.
Tip
Electrons don't release all their energy at once.
The stepwise transfer through multiple carriers allows the cell to capture energy efficiently at each stage, rather than losing it all as heat.
Step 4: Released Energy Drives Proton Pumping
The protein complexes in the ETC use the captured energy to actively pump protons (H⁺)from the mitochondrial matrix into the intermembrane space.
Protons are pumped against their concentration gradient (from low H⁺ to high H⁺).
This is active transport, requiring energy input.
Warning
Don't confuse where the energy comes from.
The energy for pumping protons doesn't come from ATP, it comes from the electrons moving down the chain.
The ETC creates the conditions for ATP synthesis; it doesn't use ATP.
Step 5: The Proton Gradient Forms
Continuous proton pumping creates:
High H⁺ concentration in the intermembrane space.
Low H⁺ concentration in the matrix.
This difference is the proton gradient (also called the electrochemical gradient).The gradient stores potential energy.
Hint
The gradient exists across the inner mitochondrial membrane, between the intermembrane space and the matrix.
Self Review
What happens to energy as electrons move through carriers in the ETC?
How is the energy released during electron transfer used?
In which direction are protons pumped? From matrix to intermembrane space, or vice versa?
Where exactly is the proton gradient located in the mitochondrion?
C1.2.15 chemiosmosis and the synthesis of atp in the mitochondrion (hl)
The Problem: Converting Gradient Energy into ATP
The electron transport chain pumps protons from the matrix into the intermembrane space, creating a concentration gradient.
This gradient stores energy, but gradients don't directly make ATP.
The cell needs a mechanism to convert the potential energy of this gradient into the chemical energy of ATP bonds.
That mechanism is known as chemiosmosis.
The Solution: Chemiosmosis
Definition
ChemiosmosisChemiosmosis is the movement of protons () across a membrane, driven by a concentration gradient, to power the synthesis of ATP.
Protons flow back into the matrix through a specialized enzyme called ATP synthase.
This flow directly drives ATP production.
Analogy
Imagine a dam holding back water.
When the gates open, water rushes through, turning turbines to generate electricity.
Now, replace the water with protons () and the turbines with ATP synthase--this is chemiosmosis in action.
How ATP Synthase Works
Definition
ATP SynthaseThe enzyme that uses the energy from the proton gradient to catalyze the synthesis of ATP from ADP and inorganic phosphate.
ATP synthase is an enzyme embedded in the inner mitochondrial membrane.
Protons flow through a channel in ATP synthase from the intermembrane space back into the matrix, moving down their concentration gradient.
This proton flow causes part of ATP synthase (the rotor) to spin physically.
The mechanical rotation forces conformational changes in the catalytic head of the enzyme.
These shape changes bring ADP and Pi together in the active site and compress them, forcing the formation of ATP.
Tip
Proton flow releases energy -> that energy immediately drives ATP synthesis through mechanical rotation.
If protons don't flow, ATP synthase doesn't rotate, and ATP isn't made.
If ATP synthase is blocked, protons can't flow back through it, and the gradient builds up.
Self Review
What creates the proton gradient across the inner mitochondrial membrane?
Where is the concentration of protons higher, in the matrix or the intermembrane space?
What physical movement occurs in ATP synthase when protons flow through it?
How does rotation of ATP synthase lead to ATP formation?
C1.2.16 role of oxygen as terminal electron acceptor in aerobic cell respiration (hl)
Oxygen Accepts Electrons to Sustain the Electron Transport Chain and ATP Production
The electron transport chain (ETC) transfers electrons through a series of protein complexes.
As electrons move through the chain, they must be continuously removed at the end to keep the process flowing.
Without a final electron acceptor, electrons would accumulate and the chain would stop.
Oxygen serves as the terminal electron acceptor, accepting electrons from the final carrier in the ETC.

Warning
A common misconception is that oxygen is directly involved in ATP synthesis.
In reality, oxygen's role is to accept electrons, enabling the electron transport chain to function and sustain the proton gradient needed for ATP production.
How Oxygen Accepts Electrons and Forms Water
Oxygen accepts electrons from the final complex in the ETC.
Simultaneously, oxygen combines with protons (H⁺) from the mitochondrial matrix.
This removes both electrons and protons from the system, preventing their accumulation.
This reaction produces metabolic water (H₂O):
Hint
By accepting electrons, oxygen allows the electron flow along the ETC to continue.
Continued electron flow sustains proton pumping from the matrix into the intermembrane space.
The proton gradient drives ATP synthesis through chemiosmosis (as covered in C1.2.15).
Without oxygen, electron flow stops -> proton pumping stops -> the gradient dissipates -> ATP production halts.
What Happens Without Oxygen
ETC stops: Electrons have nowhere to go, so the chain backs up.
NADH and FADH₂ accumulate: These cannot donate electrons to the ETC and remain reduced.
Krebs cycle stops: The cycle requires NAD⁺ and FAD to accept electrons. Without the ETC regenerating these carriers, the cycle halts.
ATP production drops: The cell relies on glycolysis alone, producing only 2 ATP per glucose instead of approximately 32 ATP from aerobic respiration.
Analogy
Imagine the electron transport chain as a conveyor belt carrying electrons.
Oxygen is the final station where the electrons are delivered.
If there is no oxygen, the belt backs up and stops functioning, halting the entire process.
Why Oxygen is the Ideal Terminal Electron Acceptor
High Electronegativity: Oxygen has a strong affinity for electrons, making it highly effective at accepting them at the end of the chain.
Energy Efficiency: The transfer of electrons to oxygen releases a significant amount of energy, which is harnessed to pump protons and drive ATP synthesis.
Byproduct Management: The formation of water as a byproduct is non-toxic and easily managed by the cell.
Self Review
What is the terminal electron acceptor in aerobic respiration?
What happens to oxygen when it accepts electrons from the ETC?
What is metabolic water and how is it formed?
Why does electron flow stop without oxygen?
How does the absence of oxygen affect NADH and the Krebs cycle?
C1.2.17 differences between lipids and carbohydrates as respiratory substrates (hl)
Lipids Yield More Energy Per Gram
Lipids yield ~37 kJ/g, while carbohydrates yield ~17 kJ/g.
Lipids contain more carbon-hydrogen (C-H) bonds and less oxygen compared to carbohydrates.
C-H bonds are highly oxidizable, they release substantial energy when broken down during cellular respiration.
Carbohydrates already contain more oxygen, meaning they are partially oxidized and release less energy per gram.
Hint
More oxidizable hydrogen and carbon = more electrons for the electron transport chain = more ATP produced per gram.
Note
Lipids are ideal for long-term energy storage because they pack more energy into a smaller mass.
Only Carbohydrates Can Be Used Anaerobically
Carbohydrates enter via glycolysis:
Glucose is broken down into pyruvate during glycolysis (occurs in the cytoplasm).
Glycolysis produces small amounts of ATP and reduced NAD (NADH).
This process does not require oxygen.
If oxygen is unavailable (anaerobic respiration):
Pyruvate is converted to lactate (in animals) or ethanol + CO₂ (in yeast).
This regenerates NAD⁺, allowing glycolysis to continue producing ATP.
If oxygen is available (aerobic respiration):
Pyruvate enters the mitochondria and is fully oxidized via the Krebs cycle and electron transport chain.
This produces significantly more ATP.
Note
Only carbohydrates can undergo anaerobic respiration because only they enter glycolysis, which is the only ATP-producing pathway that works without oxygen.
Lipids Require Oxygen and Cannot Be Used Anaerobically
Lipids Enter via Beta-Oxidation:
Fatty acids are broken down through beta-oxidation in the mitochondrial matrix.
The fatty acid chain is repeatedly cleaved, removing 2-carbon fragments.
Each fragment combines with coenzyme A to form acetyl-CoA.
This repeats until the entire fatty acid is converted into multiple acetyl-CoA molecules.
Acetyl-CoA Only Works Aerobically
Acetyl-CoA enters the Krebs cycle, producing NADH and FADH₂.
NADH and FADH₂ donate electrons to the electron transport chain, which generates ATP.
The ETC requires oxygen as the terminal electron acceptor.
Note
Why lipids can't be used anaerobically:
Lipids produce acetyl-CoA, which bypasses glycolysis entirely.
Acetyl-CoA can only be processed through the Krebs cycle.
Without oxygen, the ETC stops, NADH and FADH₂ accumulate, the Krebs cycle halts, and acetyl-CoA cannot be metabolized.
Key Differences Summarized
Feature | Carbohydrates | Lipids |
|---|---|---|
Energy Yield | ~17 kJ/g | ~37 kJ/g |
Anaerobic Respiration | Possible (via glycolysis) | Not possible |
Oxidation Process | Less hydrogen, more oxygen | More hydrogen, less oxygen |
Storage Form | Glycogen (short-term) | Triglycerides (long-term) |
Self Review
Why do lipids yield more energy per gram than carbohydrates?
What structural difference between lipids and carbohydrates explains their different energy yields?
Can lipids be used in anaerobic respiration? Why or why not?
Why can only carbohydrates undergo glycolysis?