AP Biology Notes
Chemistry of Life
Properties of Water
Waters Properties are caused by its Polarity.
Because of this polarity it can bond with other water molecules via hydrogen bonds.
Hydrogen Bonds are a weak attraction between two highly electronegative atoms and a hydrogen atom taking part in different polar covalent bond.
* Hydrogen bonds are not technically a chemical bond but are able to stabilize biological structures.
Properties of water:
Cohesion: Provides surface tension (Water = Water)
Adhesion: When two unlikely molecules stick to one another
Capillary Action: Caused by both adhesion and cohesion allowing water to travel up the sides of a small tube
Reducing surface tension
- Soap molecules have a hydrophobic and hydrophilic end that helps breakdown the bonds between water molecules.
Water & Temp:
Water absorbs more heat than other regular molecules.
- Molecules move faster when gaining heat
- Molecules move slower when loosing heat
Takes longer to evaporate
Takes longer to heat up
Evaporative cooling = Surface temperature of water decreasing during evaporation
Ice will float on liquid water forming a blanket to protect other organisms from the freezing temperatures
Water is able to easily dissolve other substances because of its polar nature and ability to create hydrogen bonds.
Substances that are able to dissolve in water are called hydrophilic, meaning water loving, and substances that can’t dissolve in water are called hydrophobic, meaning it dislikes water.
Water is the basis for all things in life. Water helps moderate temperatures within organisms and environments.
Water helps animals regulate their internal temperatures through sweating or panting when doing something labor intensive.
H - 1 bond
O - 2 bonds
N - 3 bonds
C - 4 bonds
Key Terms:
Hydrophilic: A substance that dissolves in water
Polarity: A property of water where they have either a positive or negative end.
Hydrogen Bonds: Weak bonds between molecules, particularly important in water
Ionic Bonds: Strong bonds between positive and negative charged ions
Hydrophobic: Substances that cannot dissolve in water.
Hydrophilic Substances, due to their polar nature and ability to form interactions with water molecules, are able to easily dissolve in water.
Many nutrients are hydrophilic allowing them to be absorbed and transported into the bloodstream.
Intro to Biomolecules
All living things are composed of Oxygen, Hydrogen, and Carbon.
Molecules that contain Carbon are organic
Why Carbon?
Carbon is essential for living things because of its versatile bonding behavior.
-A single Carbon atom can form up to 4 covalent bonds with other atoms.
-The molecules can be polar or nonpolar.
-Carbon molecules often can form chains or rings
-Carbon atoms can be assembled and reformed into many different organic compounds.
Carbons rings are often represented as polygons.
Macromolecule Structure:
Monomers - Molecules used as repeated subunits to build larger molecules.
Polymers - Larger Molecules built from a monomer chain.
Dehydration Synthesis:
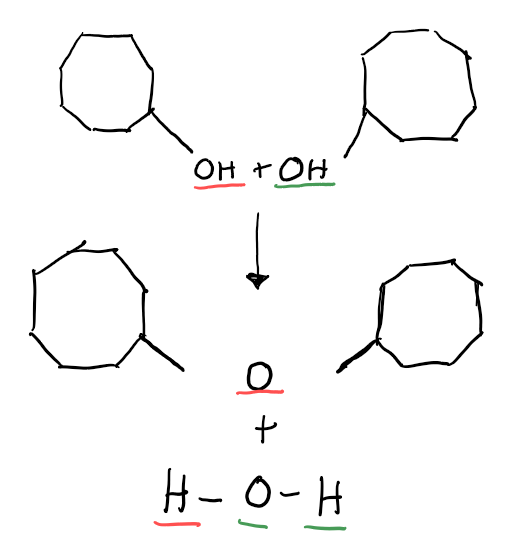
An Enzyme removes a hydroxyl group from one molecule and a hydrogen atom from another. A covalent bond forms between the molecules, and water also forms.
-A non polar molecule has an even distribution of electron charge while polar molecules have an uneven charge.
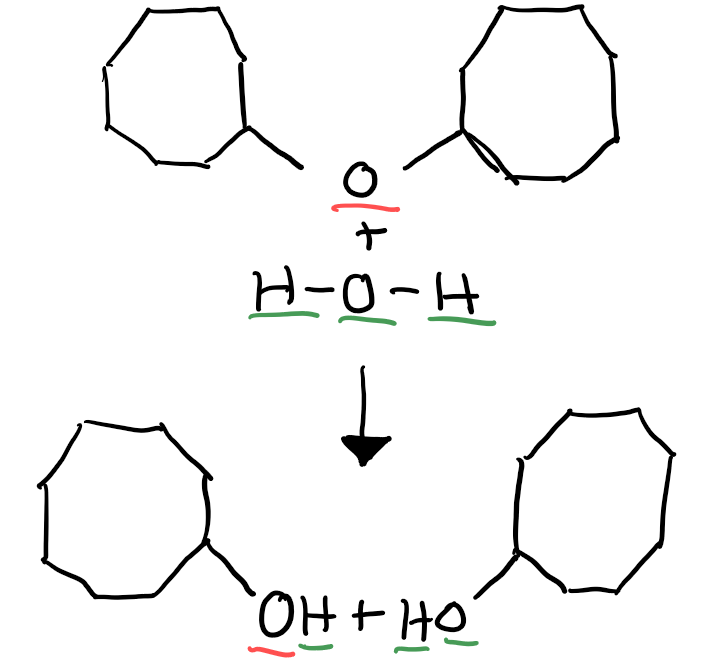
An enzyme attaches a hydroxyl group to a hydrogen atom at the cleavage site
The type of covalent bond formed between monomers depends on the monomer / macromolecule.
Macromolecule | Monomer(s) | Bond that connects monomers |
Carbohydrates | Monosaccharides | Glycosidic |
Lipids | Fatty acids and Glycerol | Ester |
Proteins | Amino Acids | Peptide |
Nucleic Acids | Nucleotides | Phosphodiester |
Proteins
All cellular processes involve proteins.
Proteins are the most diverse molecule and are used in:
-Structure
-Nutrition
-Enzymes
-Transport
-Communication
-Cellular defense
Proteins are large, complex molecules that are made up of building blocks called amino acids, which are one of the four major macromolecules of life.
Proteins have four levels of structure.
Primary structure –
The sequence of amino acids in a polypeptide chain.
→ Determined by the gene that encodes the protein.
Example: Met–Ala–Ser–Tyr–Gly...Secondary structure –
Folding of the chain due to hydrogen bonds between backbone atoms.
Two main types:α-helix (alpha helix) – coiled shape
β-pleated sheet – folded shape
Tertiary structure –
The overall 3D shape of one polypeptide chain, formed by interactions among R groups (side chains).
These include:Hydrogen bonds
Ionic bonds
Disulfide bridges (covalent bonds between cysteines)
Hydrophobic interactions
Quaternary structure –
When multiple polypeptide chains come together to form a functional protein.
Protein Function
Type of Protein | Function | Example |
Enzymes | Speed up chemical reactions | Amylase, DNA polymerase |
Structural | Support and shape | Collagen, Keratin |
Transport | Moving substances | Hemoglobin (O₂ transport) |
Defense | Protect the body | Antibodies |
Signal | Communication between cells | Insulin and growth hormones |
Motor | Movement | Myosin, actin |
The shape of a protein determines its function
If a protein loses its shape, it loses its function.
Enzymes
Enzymes are biological catalysts, they help speed up chemical reactions in cells without being used up in the process.
They are essential for metabolism, digestion, DNA replication, and other life processes.
Most enzymes are proteins
Enzymes and substrate fit together like lock and key
How do enzymes work?
-Substrate binds to the enzymes active site and the enzyme lowers the activation energy needed for the reaction to happen. The product is then formed and released, the enzyme is unchanged and can catalyze more reactions.
-Enzymes DON’T add energy, they make reactions happen faster and more efficiently.
Factors that affect enzyme activity:
Tempature
- Too low → Slow activity
- Optimal Temp → Highest activity
- Too High → Enzyme looses shapepH
-Each enzyme has an optimal pH
-Too acidic or basic → Denaturation (Loss/Looses of shape)Substrate concentration
-More substrate → more activity, up to a pointEnzyme Concentration
More enzymes → Faster reaction (If enough substrate is present)
Enzyme Inhibitors:
- Competitive inhibitors
-Bind to the active site, blocking the substrate
-Noncompetitive Inhibitors
-Bind somewhere else and change the enzyme’s shape, reducing function.
Key terms to know:
Catalyst:
A substance that speeds up a chemical reaction without being consumed in the process.Inhibitor:
A molecule that reduces enzyme activity. It can either block the active site or change the enzymes shape.Competitive Inhibitor:
A molecule that competes with the substrate for the active site.Noncompetitive Inhibitor:
A molecule that binds to a different part of the enzyme.Allosteric Site:
A site other than the active site where a molecule can bindInduce fit model:
A model of enzyme function where the enzyme slightly changes to better fit the substrate where it binds.
Enzymes and Metabolic Pathways
Enzymes often work in sequences, where the product of one enzyme becomes the substrate for the next, called a metabolic pathway. these are regulated by feedback mechanisms.
Feedback inhibition (Negative Feedback)
-The end product of a pathway inhibits an earlier enzyme, preventing overproduction. It is a form of noncompetitive inhibition that helps maintain homeostasis.
EX: In cellular respiration, ATP can inhibit enzymes involved in glycolysis.
Carbohydrates
v
Lipids
f
Nucleic Acids
f