intermolecular forces of attraction (imfa)
attracts molecules together
binds the compound together
binds the molecules of the compounds together
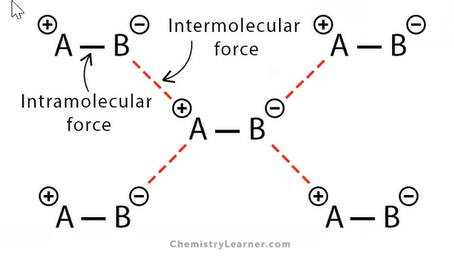 types of intermolecular forces
types of intermolecular forces
london dispersion force
temporary dipole
nonpolar molecules
low strength of attraction
dipole-dipole force
permanent dipoles
polar molecules
medium strength of attraction
hydrogen bond
permanent dipoles exclusive to F, O, and N
polar molecules
medium to high strength of attraction
ion-dipole force
full ion and dipole
ion and polar molecule
high strength of attraction
order of the strength of the bonds (lowest to highest)
london dispersion force → dipole-dipole force → hydrogen bond → ion-dipole force
what is the effct of imfa to the surroundings?
responsible for creating solid liquid and gas
the stronger the interaction between the bonds, the stronger the structure in room temperature
the tighter the bond, the more solid the object is
melting and boiling point
the stronger the intermolecular forces, the higher the melting point and boiling point
viscosity
refers to the ease with which molecules can move past each other
the stronger the intermolecular forces, the more viscous the substance is
the thicker the liquid… or something
cohesion and adhesion
cohesion - attraction between like molecules
adhesion - attraction between molecules
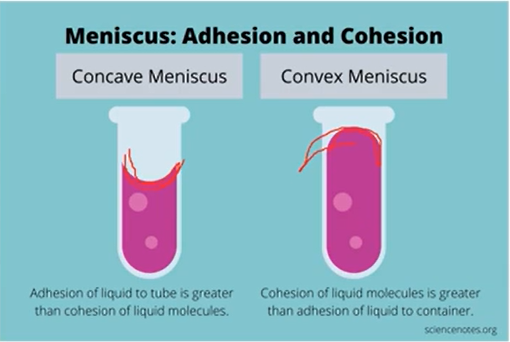
surface tension
results from the net inward attraction experienced by the molecules on the surface of a liquid
the stronger the imfa the higher the surface tension will be
capillary action
the ability of a liquid to flow up a thin tube against the influence of gravity
stronger the adhesion between galss and iquid, the higher the capillary action
solubility
ability of solute to dissolve in solvent “like dissolves ike”
nonpolar substances are hydrophobic and polar substances are hydrophilic
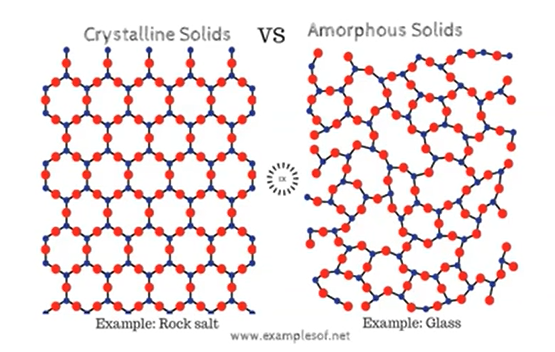
crystalline & amorphous solids
formation nung . material or something ganern .
higher the arrangement, the stronger the intermolecular force
 anitititiitiit fragile
anitititiitiit fragile