Topic 11 Regents Chemistry Review: Organic Chemistry
Topic Overview
Organic Chemistry is the study of carbon and most carbon compounds. The name organic is a remnant of time when it was thought that carbon compounds could only be made by living things; hence the term organic. Today it is widely recognized that organic chemistry contains far more compounds than those made by living things. The number of organic compounds are enormous. Tens of thousands of new organic compounds are discovered every year, and there seem to be no end in sight to future discoveries.
Bonding of Carbon Atoms
The ability of carbon to form many different compounds is based, to a large extent, on the tendency of carbon atoms to covalently bond with other carbon atoms and form chain. This process can be continued indefinitely, leading to chains of thousands of carbon atoms.
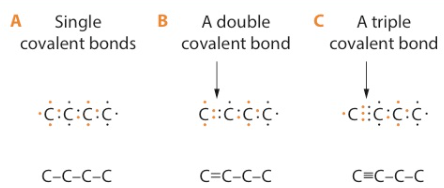
In such diagrams, a single line is often used to represent the pair of shared electrons (C-C). When one pair of electrons is shared between two carbon atoms, the bond is called a single bond. Organic compounds containing only single bonds are said to be saturated.
Sometimes carbon atoms can share two pairs of electrons, forming a double bond or even three pairs of electrons in a triple bond. Compounds containing one or more double or triple covalent bonds are unsaturated compounds.
Carbon atoms can also bond with other carbon atoms forming three-dimensional networks. Diamonds are made of networks of carbon atoms in which each carbon atom is bonded to four other carbon atoms in a characteristic network structure. As recent discoveries have shown, carbon atoms can be arranged in large networks in which each carbon atom is bonded a single bond to two other carbon atoms and with a double bond to one other carbon atom. The most common of these forms is buckminsterfullerene, also called a bucky ball, which contains 60 carbon atoms forming a pattern similar to that on a soccer ball. The carbon atoms form a framework, and inside of the network is empty space.
Structural Formulas The molecular formula shows the kind and number of atoms in a compound. For example, the molecular formula C3H8 tells the reader that the compound contains three carbon atoms and eight hydrogen atoms. Structural formulas attempt to show not only the kinds and numbers of atoms but also the bonding patterns and approximate shapes of molecules.
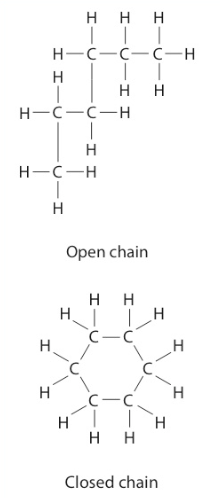
These pictures show the molecular formulas and structural formulas for two organic compounds. It is important to remember that these structural formulas are two-dimensional representations of three-dimensional molecules. Each carbon atom can be pictured as the center of a tetrahedron, and a short line can represent each of its covalent bonds.
Hydrocarbons
Although there are an extremely large number of organic compounds, the study of these compounds is simplified because they can be classified into groups called homologous series, the members of which have related structures and properties. A homologous series of compounds is a group of related compounds in which each member differs from the one before it by the same additional unit.
Hydrocarbons are organic compounds that contain only atoms of hydrogen and carbon. These compounds are the parent compounds from which many organic compounds are derived. Alkanes, alkenes, and alkynes are three important homologous series of hydrocarbons.
Alkanes
The alkanes are a homologous series of saturated hydrocarbons that release energy when burned. Methane (CH4), the first member of the series, comprises about 90% of natural gas, which is used to heat many homes. The second member, ethane (C2H6), is found in disposable lighters.
Alkenes
The same relationship between successive members can be found in the homologous series of alkenes. Each member of the alkene series contains one double bond. Alkenes are unsaturated, like alkynes because they contain one or more double or triple covalent bonds. Alkenes are named from the corresponding alkane by replacing the -ane of the alkane name to -ene.
Alkynes
The alkynes are a homologous series of unsaturated hydrocarbons that contain one triple bond. The naming of alkyne series repeats the pattern observed in alkenes. To find the alkyne name, use the corresponding name from the alkane series, and change the -ane ending to -yne. Thus the first member of the series (C2H2).