giant covalent structures
Not all non-metals exist as molecules. Some have a giant structure, there is a huge number of atoms held by a network of covalent bonds - but there are no molecules/intermolecular forces.
High melting and boiling points
All giant covalent substances have high melting and boiling points because there are strong covalent bonds between all the atoms in the giant covalent lattice structure and a lot of energy is required to break these bonds.
Silicon dioxide (silica/sand)
Silicon dioxide has a giant rigid covalent structure similar to diamond. Each oxygen atom is joined to two silicon atoms and each silicon atom is joined to four oxygen atoms. The formula is SiO*2. Most sand is made up of silica. Silica has a high melting point, is very hard and does not conduct electricity (no delocalised electrons). Silica is insoluble - sand does not dissolve in water.
Diamond, graphite and graphene are all allotropes of carbon, meaning they are different structures of the same element with the same chemical properties. However, they have different physical properties because some may have delocalised electrons and weaker forces between layers.
Diamond
In diamond, each carbon atom forms 4 covalent bonds ( maximum amount ) with other carbon atoms in the giant covalent structure. Due to this rigid network diamond is very hard. There are no moving charged particles so diamond does not conduct electricity. Melting point is very high, as with all giant covalent structures. Diamonds are also insoluble. A use of diamonds are in tools as they are very hard.
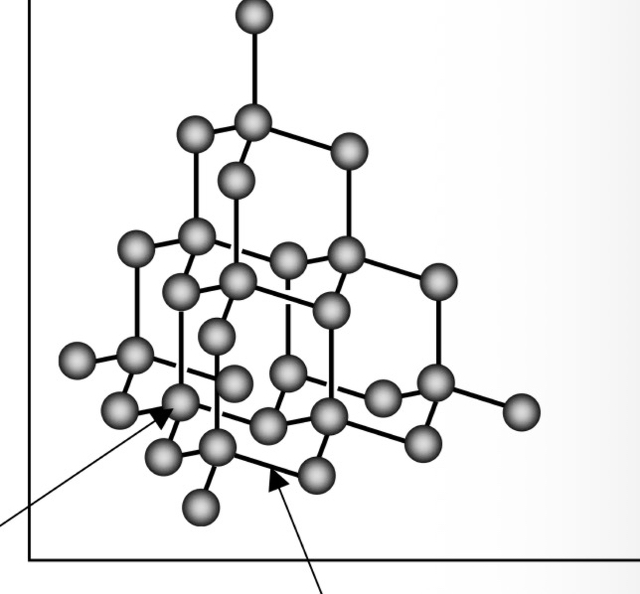
The grey balls are carbon atoms and the sticks covalent bonds, this is not a molecule and there are no intermolecular forces
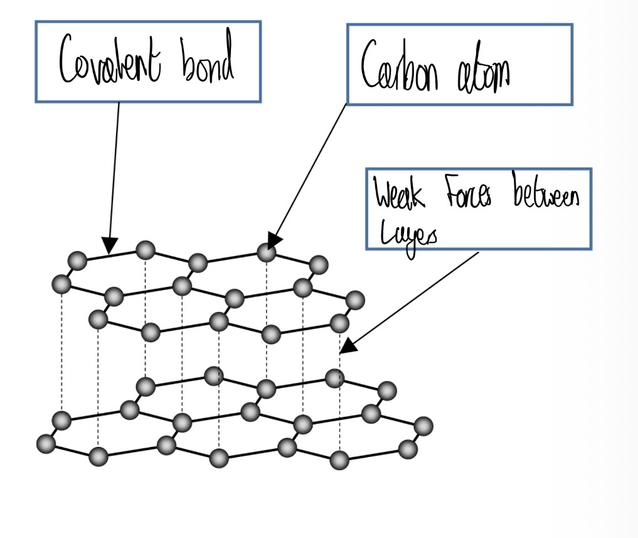
Graphite
In graphite, each carbon atom forms 3 covalent bonds with 3 other carbon atoms to form layers of hexagonal rings. There are weak forces ( NOT intermolecular) between the layers so they slide over each other. This makes graphite soft and slippery so it can be used as a mechanical lubricant and in pencils. Graphite has good conductivity because 1 delocalised electron is not covalently bonded so it can move and carry electrical charge/ heat through the structure.
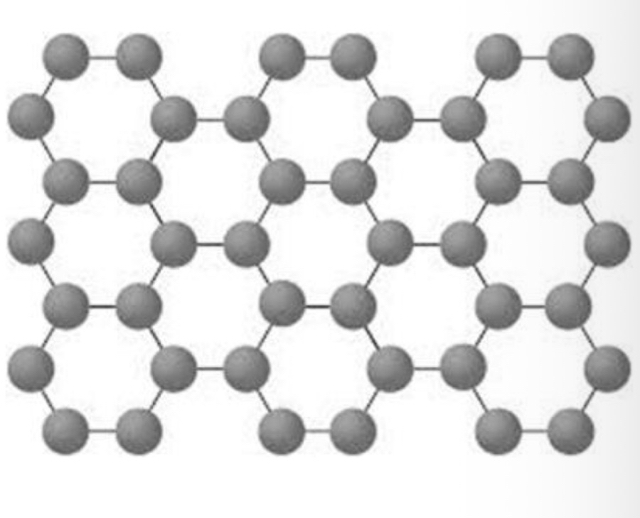
Graphene
Graphemes is simply one sheet/layer of graphite - it is just one atom thick. Graphene is an excellent conductor of both heat and electricity (delocalised electrons, but better than graphite). It has a low density and is very strong when compared to its mass. Graphene can be used in display screens, electric circuits, solar cells and more.
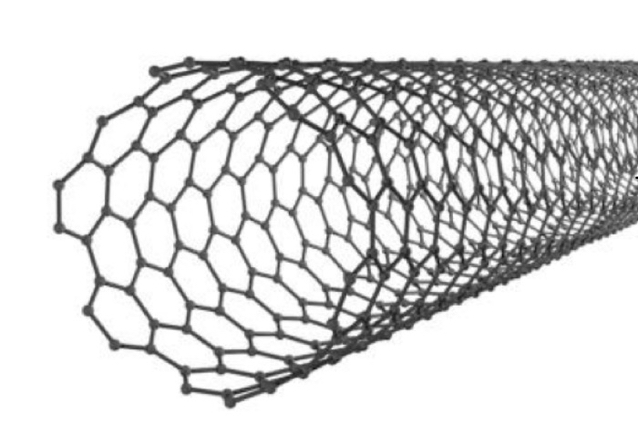
Fullerenes and carbon nanotubes
Fullerenes are molecules because they have a fixed structure that uses a finite number of carbon atoms. They are bonded in a spherical, hollow shape. The structure of fullerenes is based on hexagonal rings of carbon atoms but they may also contain wings with 5 or 7 carbon atoms. They can be used as mechanical lubricants because the molecules can roll over one another (individual spheres, not the carbons atoms).
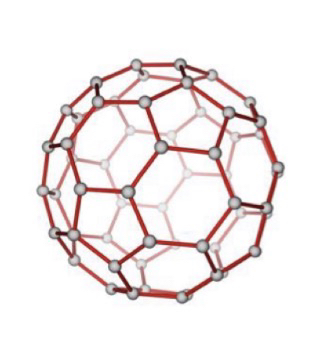
Carbon nanotubes are cylindrical fullerenes, like one layer of graphite/graphene as a cylinder. They have high tensile strength, so they are often used to strengthen materials. They also have high electrical and thermal conductivity (delocalised electrons). They can also be used for reinforcing materials.