Chemistry Unit 1:
Part 1: Kinetics
Collision Theory:
In order for particles to react, they must collide with sufficient energy(is what weakens the interatomic bonds since it makes them stretch and bend so they are temporarily easier to break) and the correct orientation(the more specific the orientation requirements are the fewer successful collisions there will be). They must collide to approach closely enough to disrupt some of their existing bonds to allow for the creation of new ones in the products. When we talk about sufficient energy, we mean particles must have equal or more energy than the activation energy(the minimum amount of energy required for a reaction to occur). Thus, the ways to speed up the rates of reaction involve making changes to the reactants to create more successful collisions(refers to orientation or energy) or the conditions they are in to increase the frequency of collisions which means there is a faster rate. The rate of reaction is the speed with which the reactants are turned into the products.
Changing Concentration(liquids) and Changing Pressure(gases):
When either increases, there are more reactive particles per unit volume thus, they collide more frequently, reacting faster because there are more reactions.

Changing Temperature:
Increases the number of particles with equal/greater energy than the activation energy, thus they can move faster leading to more frequent collisions and greater collision strength so there is a higher rate of successful collisions, so a greater rate of reaction.
Maxwell-Boltzmann diagram:
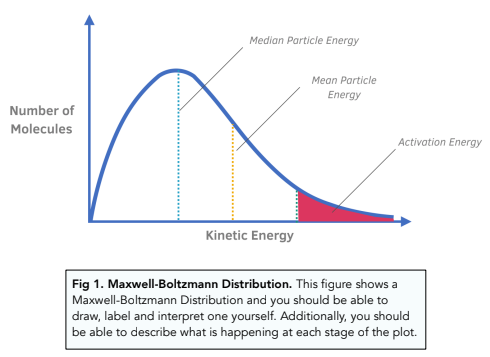
Note:
A few particles always have high kinetic energy.
If the temperature changes the peak will shift more to the right or more to the left.
And the curve heightens/lowers since the number of particles has to spread out as there is a bigger range of kinetic energies.
The curve always has to start from the origin and never crosses the x-axis.
Changing Surface Area:
Increasing the surface area exposes more particles thus, there is a greater number of particles that can react, allowing for more frequent collisions and thus a faster rate of reaction. Eg. by using magnesium powder instead of a solid peice.
Adding a Catalyst:
Catalysts are substances that speed up a reaction without being used up in it themselves by lowering the activation energy by providing an alternative chemical reaction pathway(shortcut) allowing for more particles to react thus a higher proportion of successful collisions. Represented by EA or EC on maxwell-boltzmann diagram.
Part 2: The Mole
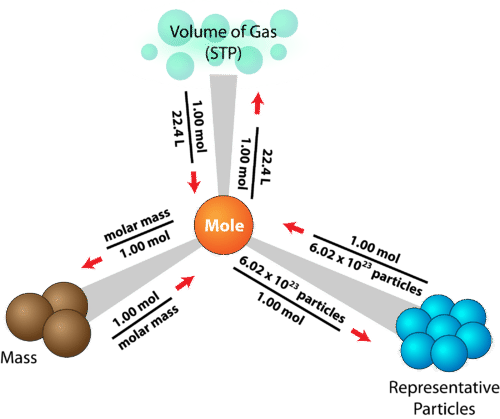
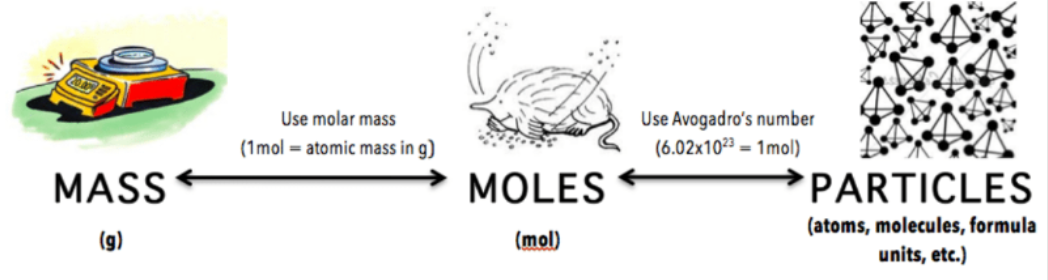
The Mole:
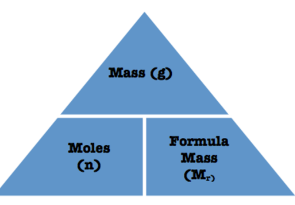
To measure how much of a substance chemists have they use the Mole as particles are too small to be counted. 1 mole of a substance= 6.02 × 1023 atoms(Avogadro’s number is the number of representative particles in a mole). We can calculate how many moles there are in a substance sample by doing its Mass(grams) divided by its Molar Mass(how many atoms there are in 1 mole of that substance which is the sum of the atomic masses in the formula).
The formula is Moles(mol)=Mass(g)/Mr(g/mol)
Example:
a)How many moles of carbon atoms is 4.72 × 1024 atoms of carbon?
1 mole= 6.02×1023
So, 4.72 × 1024 divided by 6.02×1023 equals 7.84 mol
b)How many moles are in 8.1g of HBr?
Moles= Mass/Mr = 8.1g/81 g/mol = 0.1 mol
HBr= 1+80= 81 g/mol
c)
Since Avogadro’s number is a measured quantity with three significant figures, the result of the calculation is rounded to three significant figures
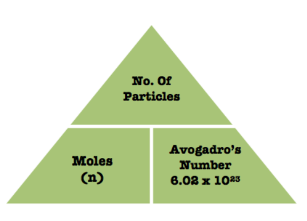
Number of particles in a sample:
To calculate the number of moles on a substance we do the total number of particles(atoms, molecules or formula units) in that substance divided by 6.02×1023(Avogadro’s number). Thus, if you want to get from having the mass of a sample of a substance to finding the number of particles in that sample you first have to convert the mass into moles and then you can convert it into a number of particles.
Formula= # of Particles/ moles x 6.02×1023
Example questions:
a) In 1 mol of Cabron atoms how many particles are there?
# of Particles/ 1 x 6.02×1023=6.02 x 1023 atoms
b) A lump of sugar contains 6 × 1022 molecules, what is the number of moles of sugar?
moles=# of particles / 6.02 x 1023 =6 × 1022 / 6.02×1023 = 0.1 mol (approximately to 1 dp)
c) How many atoms of fluorine are in 5.6 x 1022 molecules of MgF2?
Each molecule of MgF2 contains:
1 magnesium (Mg) atom
2 fluorine (F) atoms
So, for every molecule of MgF2, there are 2 fluorine atoms.
To find the total number of fluorine atoms, you multiply the number of MgF2 molecules by 2:
5.6 x 1022 molecules of MgF2×2 F atoms/molecule
=1.12× 1023 atoms
So, there are 1.12×1023 atoms of fluorine in 5.6×1022 molecules of MgF2
Leave answers in standard form
Polyatomic ions:
Polyatomic ions are a group of atoms bonded together with an overall charge and they move around as an entity so the subscript and charge NEVER change. You can put brackets around the polyatomic ion to help with clarity. When writing the formula for them in ionic compounds look at their net charge and the other atoms’ charge when forming an ion to determine if you need to add more of either to balance the charges out. Note that you never put the charge of the polyatomic ion inside the formula.
Remember that polyatomic ions contain more than one type of atom in the ion and the majority of polyatomic ions are anions that are named ending in “ate” or “ite.” Some anions have multiple forms and are named accordingly.
All of them you need to know for this unit:
Ammonium- NH4+
Carbonate- CO32-
Hydrogen carbonate- HCO3-
Sulphate- SO42-
Nitrate- NO3-
Phosphate- PO43-
Hydroxide- OH-
Example situations:
a) Sodium carbonate = Na+1 (multiply by 2 to balance out) + CO32- so final formula is Na2CO3
* The charge for elements when forming an ion is given by their group number, with group 4 being able to give or gain 4 electrons.
b) Explain why this is wrong: Ammonium diiodide
We don’t use prefixes when forming ions as the charges are balanced and in this case Ammonium has a +1 charge and iodine has a -1 charge this they balance out and you only need one of each.
c) Give the chemical formula for Barium hydrogencarbonate
Barium= Ba(group 2 so 2+ charge)
Hydrogen carbonate= HCO3- since it has a -1 charge we need two of it to balance out the +2 charge of Barium
= Ba(HCO3 )2
* added brackets around the polyatomic ion to help clarify added subscript
Converting between Moles, Concentration and Volume:
We can find the number of moles in a sample of a substance given its volume and concentration. Note that concentration is measured in mol/dm3 and volume is measured in decimetres cubed(dm3).
Examples:
a) If I have 20cm3 of 2.0mol/dm3 HCI, how many moles of HCI is that?
First, 20/1000 = 0.02dm3
Moles= conc. x vol. =0.02 × 2 = 0.0400 moles(3sf)
b) What is the volume of 79.3 g of neon gas at standard temperature and pressure?
79.3g / 40(molar mass of Ne2) = 1.9825 moles
1.9825 mol x 22.4 L = 44.4dm3 3sf
c)If 30cm3 of 0.5mol/dm3 potassium hydroxide reacted completely with 25cm3 of sulphuric acid, what is the concentration of the sulphuric acid?
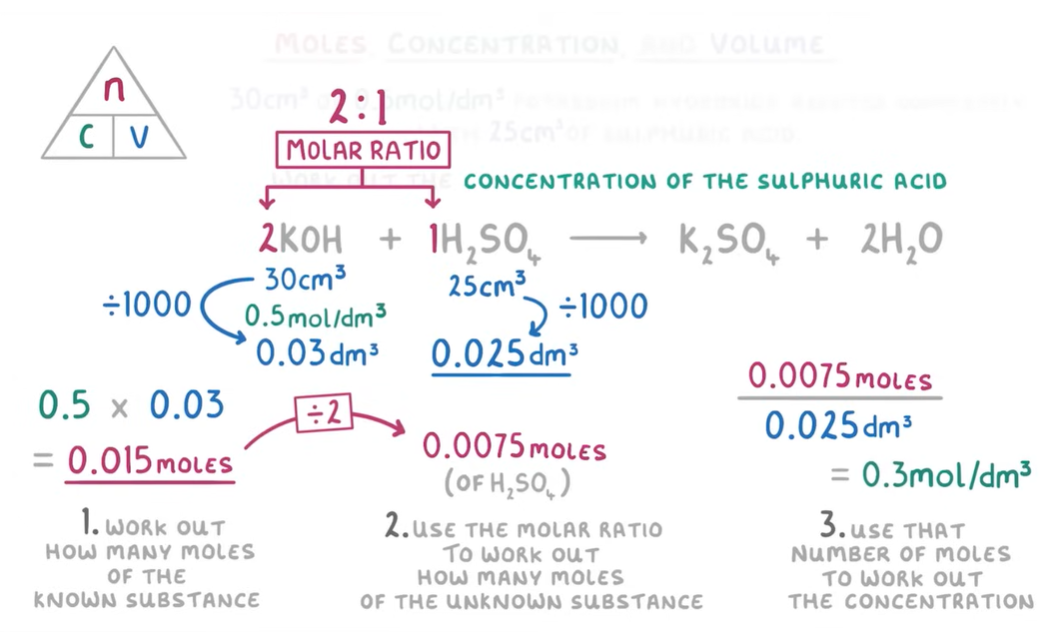
Note: you can calculate the mass you need of a substance reacting with another substance(A) if you have the mass of substance (A) and the balanced chemical formula by using the molar ratio.
You cannot convert directly from mass to number of particles(use mole road map).
Remeber that 1 mole= 22.4 L
Always make sure all of the known bits of your equations ar in the correct units- 1L= 1000 cm3= 1 dm3
Part 3: Energetics
Chemical Reactions:
Energy can never be created nor destroyed, only transferred and this occurs during chemical reactions. Firstly, energy is stored within the chemical bonds of the reactants and products(each bond stores a different and specific amount of energy). Note that energy is added to break bonds and energy is released to make new bonds.
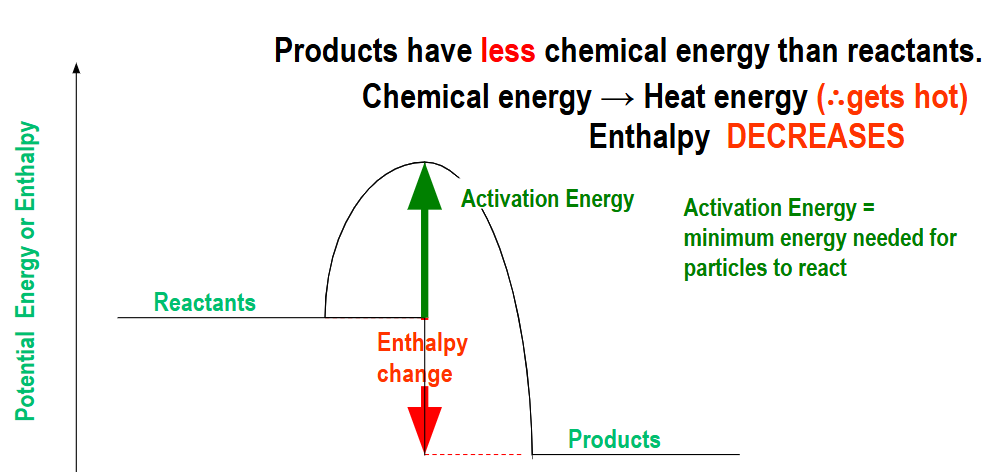
Exothermic Reactions:
They are reactions that release energy from the new bonds formed in the products into the surroundings, increasing the temperature of the surroundings(we can’t measure the substance only surroundings). Thus, the energy needed to break the bonds in the reactants is less than the energy released during the formation of new bonds in the products, which is why the energy profile looks like this:
Note:
They have a low activation energy which is why exothemric reactions are a lot more common eg. combustion reaction in the presence of oxygen, oxidation, neutralisation reactions and cellular respiration.
The enthalpy change is negative.
Reactants have more chemical energy than the products.
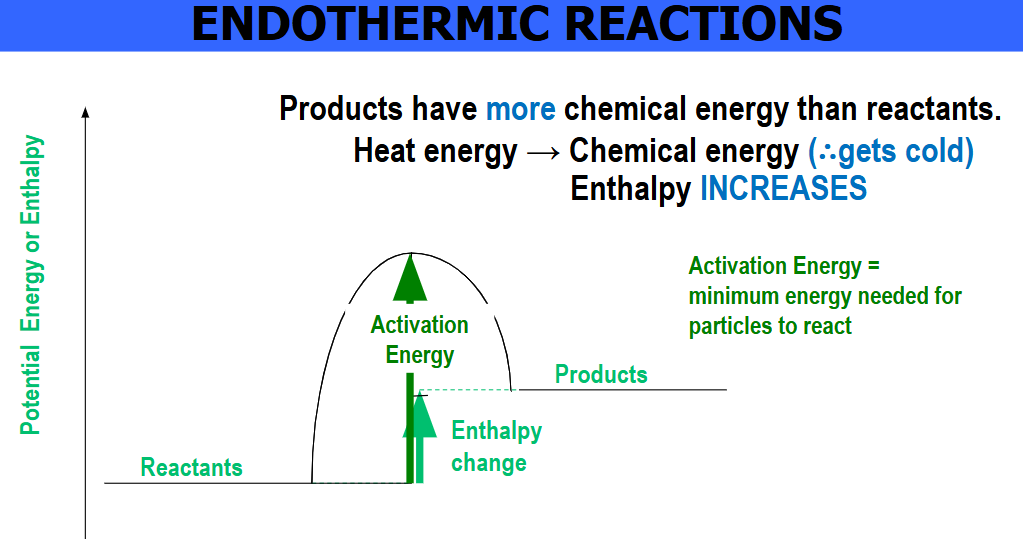
Endothermic reactions:
Reactions during which thermal energy is absorbed from the surroundings since the bonds in the reactants require more energy to break than the energy released when forming new bonds in the products. Consequently, the temperature of the surroundings typically decreases. You might need to supply heat for an endothermic reaction to occur eg. with a bunsen burner.
Note:
Activation energy is very high making these reactions less common, an example is photosynthesis(light energy into chemical energy).
Enthalpy change is positive.
Reactants have less chemical energy than products as indicated above.
Bond Energies:
Thus, we can find out whether a reaction as endothermic or exothermic by calculating the change in energy(known as enthalpy which is basically the chemical energy stored within the reactants law of thermal dynamics and is technically internal energy/potential energy). We measure the energy in Kilo Joules(kJ). The steps to follow when doing a bond energy calculation question are:
Balance the equation.
Draw the chemical formula to determine the type and number of each bond, this is important to understand how the atoms are bonded together eg. N2 is a triple bond.
Find the specific bond energies in the table provided.
Calculate total energy needed to break bonds in reactants and total energy needed to make bonds in the products.
Calculate the enthalpy change = Break-Make kJ/mol
Determine the type of reaction it is (-)= exo (+)= endo
Examples:
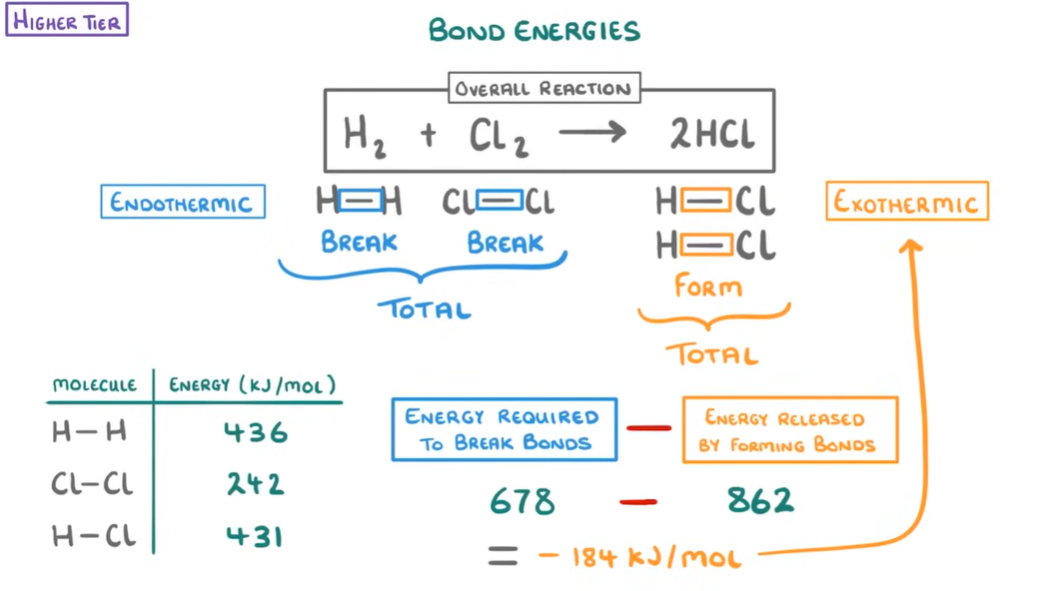
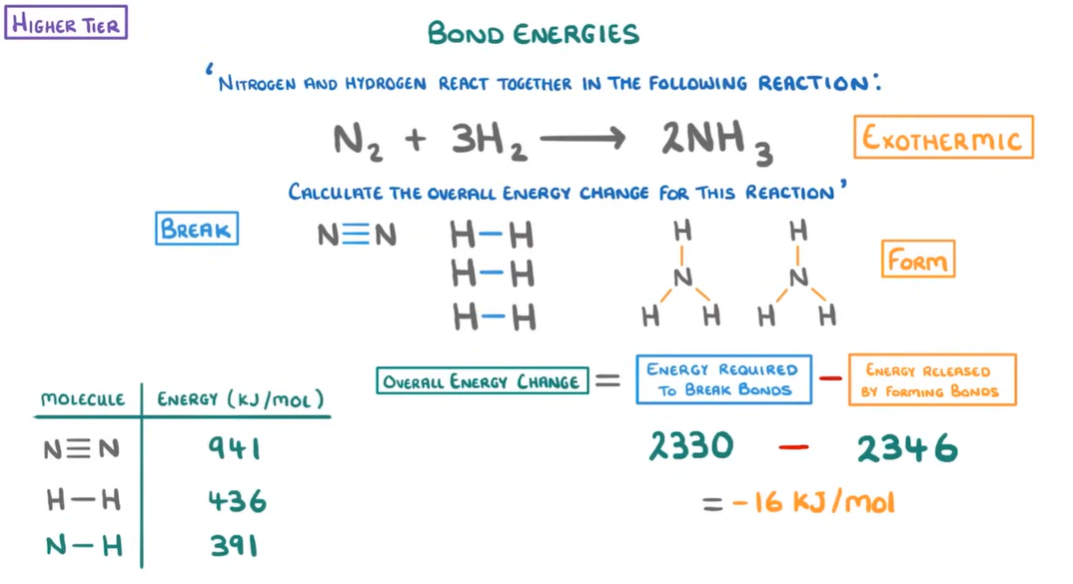
Balancing equations:
Catalyst:
If a catalyst is added the activation energy is lowered as an alternative reaction pathway is provided, making the jump from the reactants to products lower on the energy profile diargam.
Reminder from Last Year:
Heat capacity is the amount of heat required to raise the temperature of an object by 1°C(different substances and materials respond to heat in different ways, eg. water needs a lot of energy to increases its temperature whereas metals typically need little energy to increase in temperature).
Specific heat is the amount of energy required to raise the temperature of 1 gram of a substance by 1°C(precise).
Part 4: Equilibrium
Reversible Reactions:
Reactions where once the products are formed the reactants can be reformed, thus the reaction proceeds both forwards and in reverse(both directions). The fact that a reaction is reversible is indicated by the double headed arrow. If the reversible reaction is occurring in a closed system(no exchange of matter only exchange of energy) where the reactants/products can’t escape or react with something else, after a while at a certain point, the reaction will reach chemical equilibrium. A reaction has reached chemical equilibrium when the rates at which the forward reaction and reverse reaction is occurring at is equal(dynamic process), and thus the concentrations of the products and reactants are constant(don’t have to have the same concentrations) from now on. Since both reactions are occurring simultaneously there are no observable changes.
Phase equilibrium: occurs when a substance is in equilibrium between two states eg. stoppered flask of water attains equilibrium when the rate of evaporation= rate of condensation.
Solution equilibrium: ccurs when a solid substance is in a saturated solution. At this point, the rate of dissolution=rate of recrystallization.

Note:
No products at the start.
Reactants formed first here.
Examples:
a) When copper(II) sulfate (CuSO₄) is hydrated, it interacts with water to form copper(II) sulfate pentahydrate (CuSO₄·5H₂O), which is a blue crystalline compound(). Whereas when it is heated the water evaporates and is driven off, producing copper sulphate which is a white solid. Its a reversible reaction so as soon as any water is around the anhydrous copper sulphate will be converted back into the blud hydrated form.

b)Ammonium chloride is a white solid. It breaks down when heated, forming ammonia and hydrogen chloride. When these two gases are cool enough, they react together to form ammonium chloride again.
Ammonium chloride(NH4Cl(s)) ⇌ ammonia(NH3(g)) + hydrogen chloride( HCl(g))
c) CaCO3(s) ⇌ CaO(s) + CO2(g)
Le Chatelier’s Principle:
It explains how if a change(it is stressed) is made to an equilibrium the equilibrium will shift/adapt to counteract the change(stressed).The system can be stressed by a change in concentration, pressure(gases) and temperature.

Think of it as a scale where it shifts to keep it balanced, so if the number of products on the right is increased then the equilibrium will shift to the left, the reverse direction in this case, producing more reactants to return to equilibrium, balanced.
Concentration:
Equilibrium shifts to the side with fewer moles if the number of products/reactants being formed increases, to balance out the concentration(increase it or decrease it).
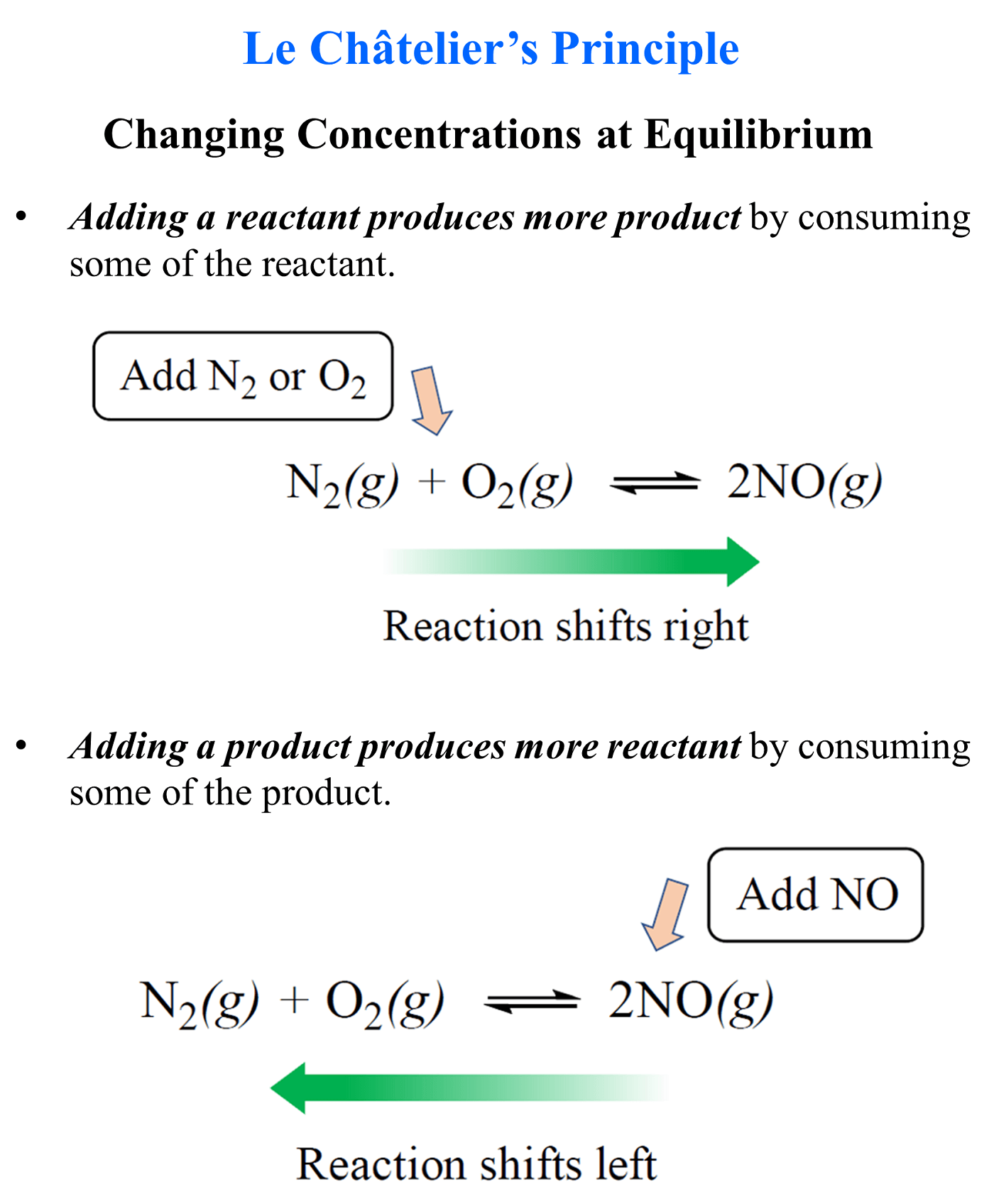
Pressure:
Equilibrium shits to the side with fewer moles thus an increases in pressure favours the side with fewer moles(lowest volume- least molecules) to decrease the pressure whereas a decrease in pressure favours the side with more moles(greatest volume) to increase the pressure back to normal.
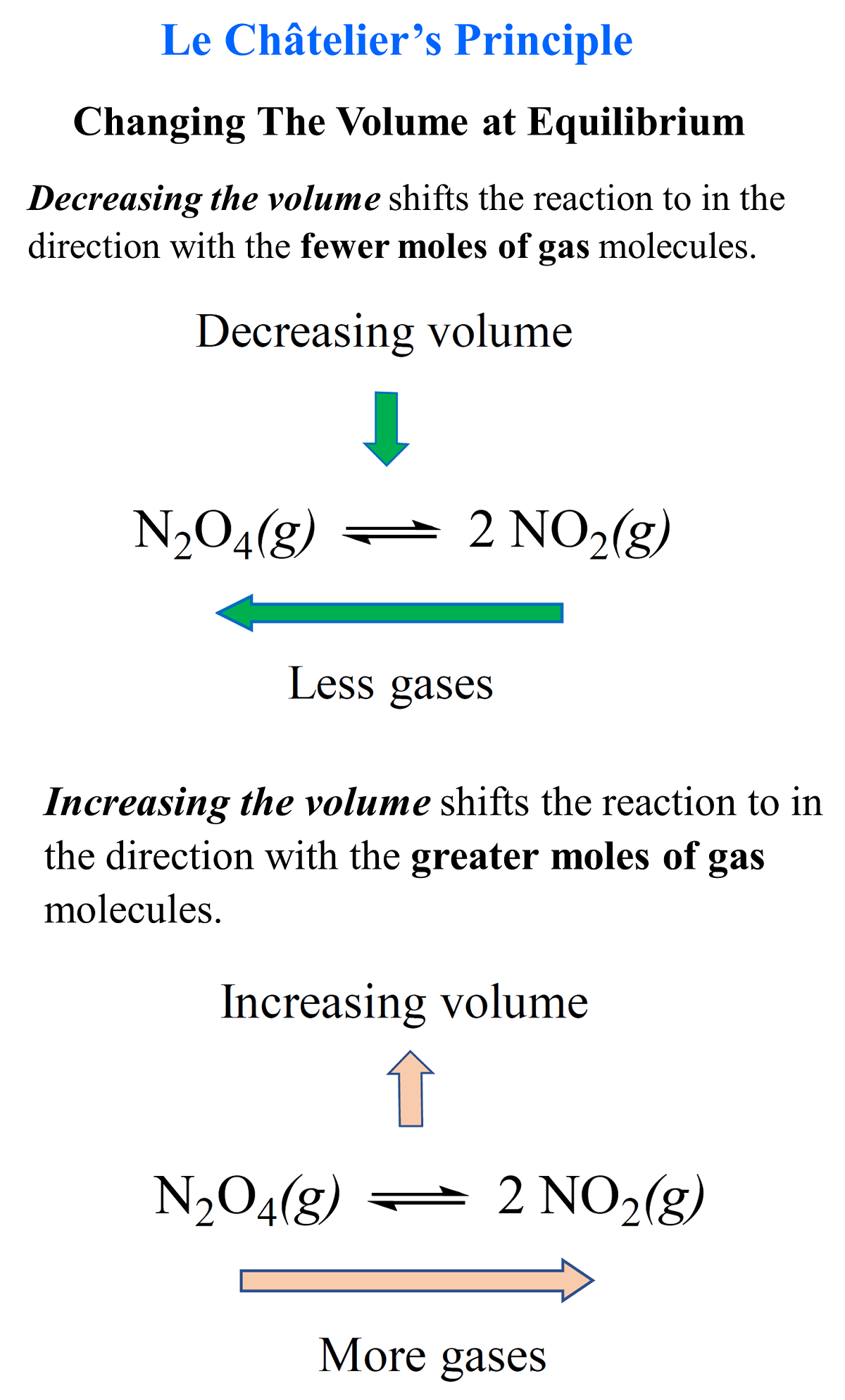
Temperature:
If there is an increase in temperature equilibrium will shift towards the side that absorbs energy which is forward/reverse (depends on reaction) direction, thus its the endothermic direction so to the left/right. Therefore, creating more reactants/products and less products/ reactants as energy is absorbed to restore the temperature. But if there is a decrease in temperature the opposite occurs, thus equilibrium shifts to the side that releases energy, which is the exothermic reaction which will be one of the directions.
Example:

Catalyst:
If a catalyst is added it speeds up both the forward and reverse reactions equally so it doesn’t affect the position of the equilibrium but reduces the time for it to be established/reached. Therefore there is no change in the amount of reactants/products being formed.
The Haber Process: N2+3H2 ⇌ 2NH3
The Haber process is an industrial method used to synthesize ammonia from nitrogen and hydrogen. The conditions typically used in the Haber process are set to increase the yield of Ammonia and make use of Le Chatelier’s principles:
High Pressure: The process operates under high pressure (around 200 atmospheres) to favour the formation of ammonia, as ammonia has less particles than nitrogen and hydrogen gas. Pressure can’t be super high due to costs and saftey.
High Temperature: The process uses intermediate temperatures (around 400-500°C) to increase the rate of reaction, even though it is an exothermic reaction. The heat favours the backward reaction, so a balance is needed between temperature and yield to ensure the rate of reaction is still relatively high(particles need the energy to collide and react) but they need a low temperature to favour the exothermic direction(produces ammonia) thus there is a compromise.
Catalyst: Iron is commonly used as a catalyst to speed up the reaction without being consumed by it. The catalyst helps achieve a good rate of ammonia production under elevated temperature and pressure conditions.
These conditions ensure that the maximum yield of ammonia is produced efficiently while balancing production rate and cost.
Chemistry Unit 2:
Part 1: Redox Reactions
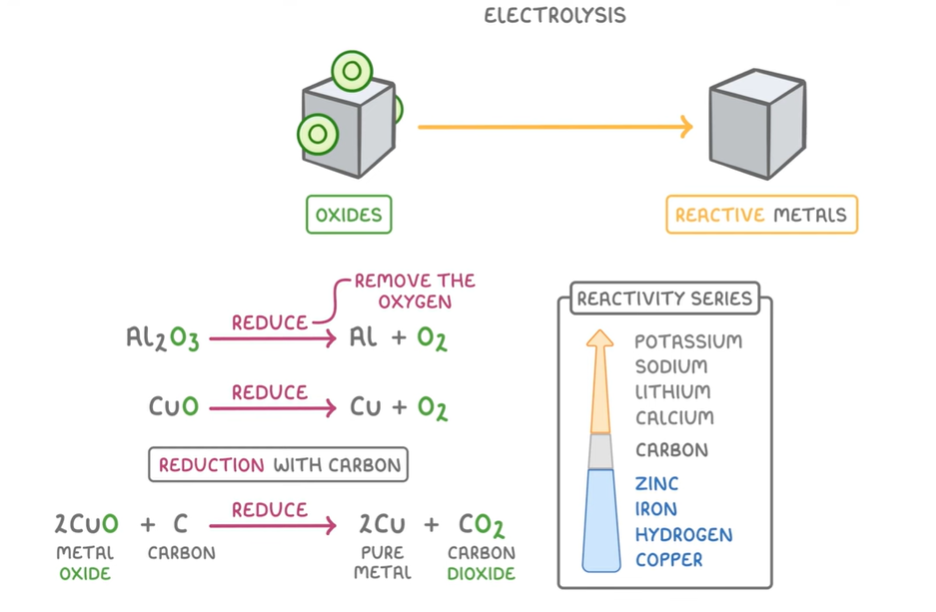
Reactivity Series:
All metals have electrons in their valance shell that they want to get rid of by reacting to form cations. The reactivity of a metal is defined by how easy it is for it to form cations, so often it does not have a lot of electrons to lose or one in a shell very far away from the nucleus of the atom so that the intermolecular attractive forces are very weak. The most reactive metals produce the most heat during the reactions with an acid or water. Therefore, Group 1 metals(alkali) are the most reactive because they only have 1 electron to lose.
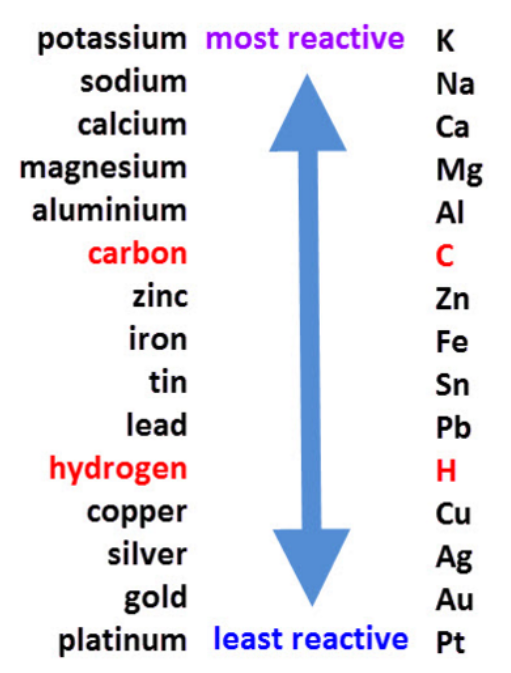
In a displacement reaction, which can be a redox reaction, a more reactive metal displaces a less reactive metal from its compound. No reaction will occur if the metal with the compound has the highest reactivity.
For example:
a) Mg+ FeSO4—> MgSO4 + Fe (Here Magnesium displaces the iron sulfate, making the sulfate bond to Magnesium instead as it’s higher up in the reactivity series.
b) Cu+ FeSO4 NO REACTION because copper is less reactive than iron
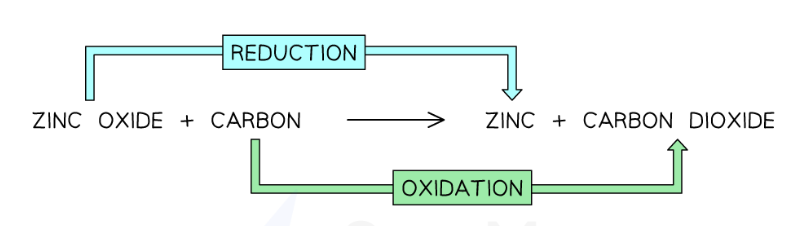
Redox Reactions:
Reactions in which oxidation and reduction take place at the same time, such as when a substance is reduced and the other substance is oxidised. Oxidisation is the gain of oxygen, meaning a loss of electrons, whereas reduction is the loss of oxygen, also meaning a gain in electrons.
Examples:
a) Here, Zinc is reduced and carbon is displacing the zinc oxide.
b) Here is the reaction between iron(III) and water and oxygen that causes the metal to rust, we can also see the individual half equations for each part of the reaction to clarify the electron transfer.
c) For the reaction Cu(NO3)2 + 2Na —> Cu +2NaNO3 deduce the half equations and the redox equation.
Reduction: Cu+2 + 2e- —> Cu
Oxidisation: Na —> Na+ + e-
Redox equation: The sum of the oxidation half + the reduction half, and then you must cancel out the electrons. DOESN’T INCLUDE SPECTATOR IONS*
Therefore: 2Na + Cu2++ 2e- —> 2Na+ + 2e- + Cu
. CANCEL OUT THE 2e-
. 2Na + Cu2+ —> 2Na+ + Cu
Change in Oxidation State:
When a substance is oxidised, it loses electrons, meaning it becomes more positive, thus we see an increase in its oxidation state(basically charge). In contrast, a substance that is being reduced will gain electrons so its charge will become more negative.
Rules for labelling charge:
Atoms of an uncombined element will have zero charge eg. Br2 and Ca
When an atom is bonded you write the charge for each individual element unless it’s a polyatomic atom.
Use the charges of other elements to figure out the charge transition metals have.
Examples:
Mg + Br2—→ MgBr2
(0) (0) (+2) (-1 bc each individual atom of Br has -1 charge)
Mg is oxidised bc its charge increased and Br is reduced bc its charge became more negative meaning it gained electrons.
3CuF2 + 2Al —→ 2AlF3 + 3Cu
(+2) (-1) (0) (+3) (-1) (0)
Cu is reduced because it becomes more negative, decreasing to 0 and aluminium is oxidised as it’s charge increases positively.
2Ag(NO3) + Cu —→ Cu(NO3)2 + 2Ag
(+1) (-1) (0) (+2) (-1) (0)
First, we work out Argon’s charge knowing that it doubled has to cancel out 2 x -1 and then we work out Copper’s charge after knowing that it has to cancel out 2 x -1. Thus, Argon is reduced and Copper is oxidised, note that Nnitrate’s charge doesn’t change.
Oxidising agent:
Gains electrons and oxidised the other substance in a chemical reaction
Reducing agent:
Loses electrons and reduces the other substance ina chemical reaction
Rusting:
It’s a type of corrosion(progressive breakdown of metals when reacting with substances in their environment) specific to Iron and steel(alloy of iron and carbon). In order for Iron to rust, both oxygen(O2) and water(H2O) must be present. Note that, only the surface of iron is exposed and rusts in the conditions but then gradually, flakes of rust fall off which reveals new metal underneath so the next layer begins to rust which makes the metal break down.
*Note: The mass of an iron piece rusting would increase when rusting if it is in a test tube and you measure the mass of it before and after since more elements become bonded and thus apart of it, increasing it’s mass whereas out in the open the flakes can fall which would decrease it’s mass.
Methods for Preventing Rust:
Barrier methods: Preventing H20 and O2 from touching the Iron by painting it(works well for large areas), oiling or greasing it(necessary if the object has moving parts like chains on a bike etc,) or Electroplating it which is when electrolysis is used to cover the iron in a thin layer of another metal(can still cause it to rust if scratched).
Sacrificial methods: Adding a more reactive metal to the Iron so the Iron won’t react and rust by eg. adding Zinc or Aluminium so they oxidise instead.
Example of both: Galvanising the Iron/Steel which means you coat the object in a layer of Zinc which acts as a protective coating(barrier method) but also if the surface is scratched the Iron still won’t oxidise because Zinc will still react which spares the Iron.
Half equations:
They contain electrons in the reactants or products and represent either the oxidation or reduction reaction part. In oxidisation reactions, the electrons are separated from the atom, which is why they are on the product side whereas, in reduction reactions, the electrons combine with the ion, adding them to the reactant side. Note that each side of the balanced half equation should cancel out to give 0, so you might have to balance the equation when eg. dealing with diatomic molecules (H O F Br I N Cl)
Example:
a) Ca + MgBr2 —> CaBr2 + Mg 1. Write out the full equation
(0) (+2)(-1) (+2)(-1) (0) 2. Calculate the charges of each atom
Ox: Ca —> Ca+2 + 2e- Red: Mg2+ + 2e- —> Mg 3. Write the half-equations
b) Ag2SO4 + Cu —> Ag + CuSO4
(+1) (-2) (0) (0) (+2)(-2)
Ox: Cu —> Cu+2 + 2e- Red: Ag+ + e- —> Ag
NOTE- IGNORE POLYATOMIC IONS IN HALF-EQUATIONS BC THEY ARE SPECTATOR IONS(NOT ACTUALLY CHANGING OR GIVING/LOSING ELECTRONS)
c) Ca + FeSO4 —> CaSO4 + Fe
(0) (+2)(-2) (+2)(-2) (0)
Ox: Ca—> Ca+2 + 2e- Red: Fe+2 + 2e- —> Fe
Electrolysis:
Is a process that allows us to split up compounds using electricity, which passes through the electrolyte—> split up into useful pure elements. For it to occur there must be an:
Electrolyte: a molten/liquid or a solution(dissolved) containing an ionic compound in which ions are free to move, thus it conducts electricity. eg. you have to melt insoluble compounds like lead bromide to allow the electrons to move more than in the solid form.
Anode: is the positive electrode that attracts anions, and it is where negatively charged ions lose electrons; thus, it’s the site of oxidation.
eg. 2Br- —> Br2 + 2e-
Cathode: is the negative electrode that attracts cations and is where positively charged ions gain electrons, thus it’s the site of reduction.
eg. Al3+ + 3e- —> Al
Wire + Battery
Needed to facilitate the electrons flow from the cathode to the anode.
At the anode and the cathode, ions are discharged to become their natural elemental form
Unless a hallide(groups 7) is present, the anode always discharges the hydroxide(OH-)
The cathode only discharges 1 ion, which is the ion of the least reactive element.
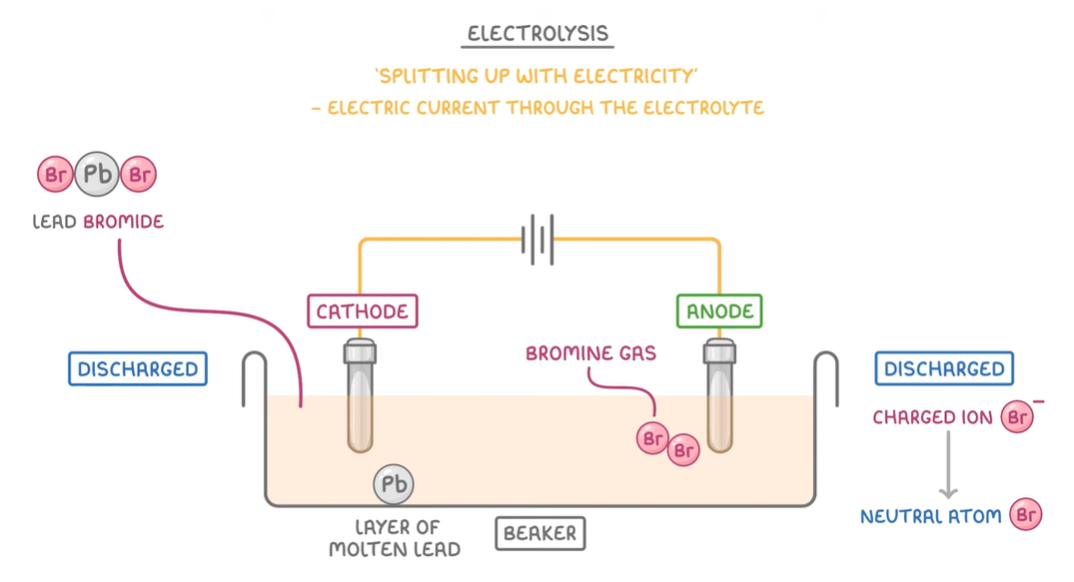
Example: Electrolysis of molten calcium bromide
Anode: bromide ions lose electrons and oxidise into bromine.
2Br- —> Br2 + 2e-
Cathode: calcium ions gain electrons from the cathode and reduce into pure calcium atoms.
Ca2+ + 2e- —> Ca
Electrolysis equations: have the ions on the reactant side with electrons cancelled out, and the pures on the product side.
2Br- + Ca+2 + 2e- —> Br2 + 2e- + Ca
CANCEL ELECTRONS BE ALREADY BALANCED
2Br- + Ca+2 —> Br2 + Ca
Electrolysis Calculations:
Metal extraction: Through these processes, we can extract useful metals from their oxides by either using carbon to displace the metal, which only works for less reactive metals or we can use electrolysis, where we purify and melt the metal before setting up he electrolysis reaction
Part 2: Reacting Masses
Molar Ratio:
The relationship between the coefficients in a balanced chemical equation can give us important info.
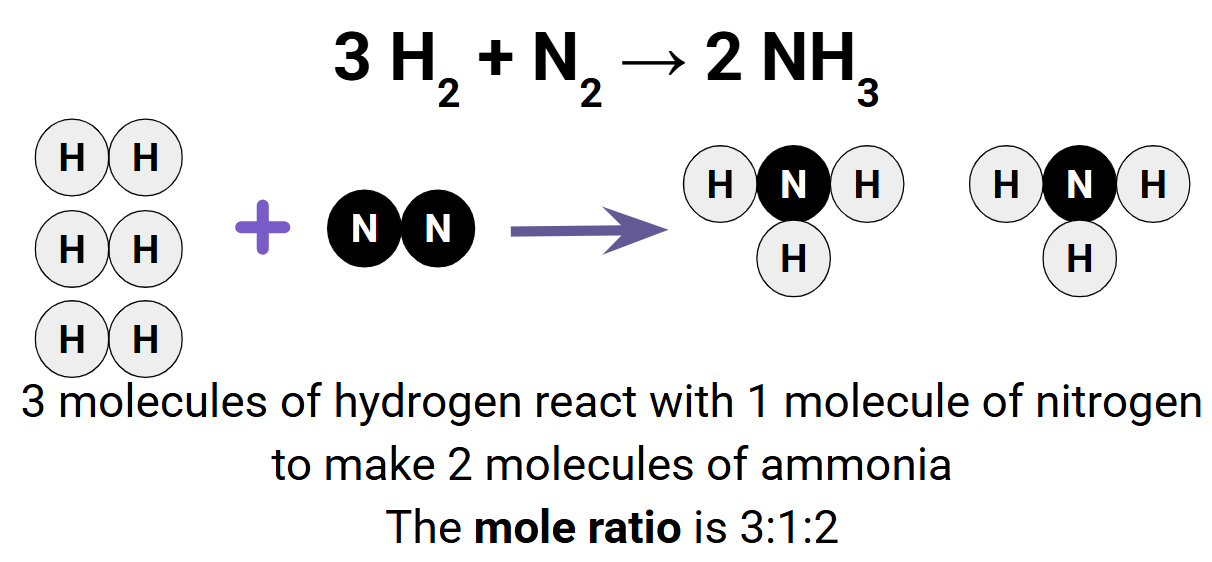
As you can see here the ratio is between the coefficients of each atom. Now if you have the mass of one of the substances you are reacting with you can figure out the mass of the other substances needed to form the products and how much product you will form.
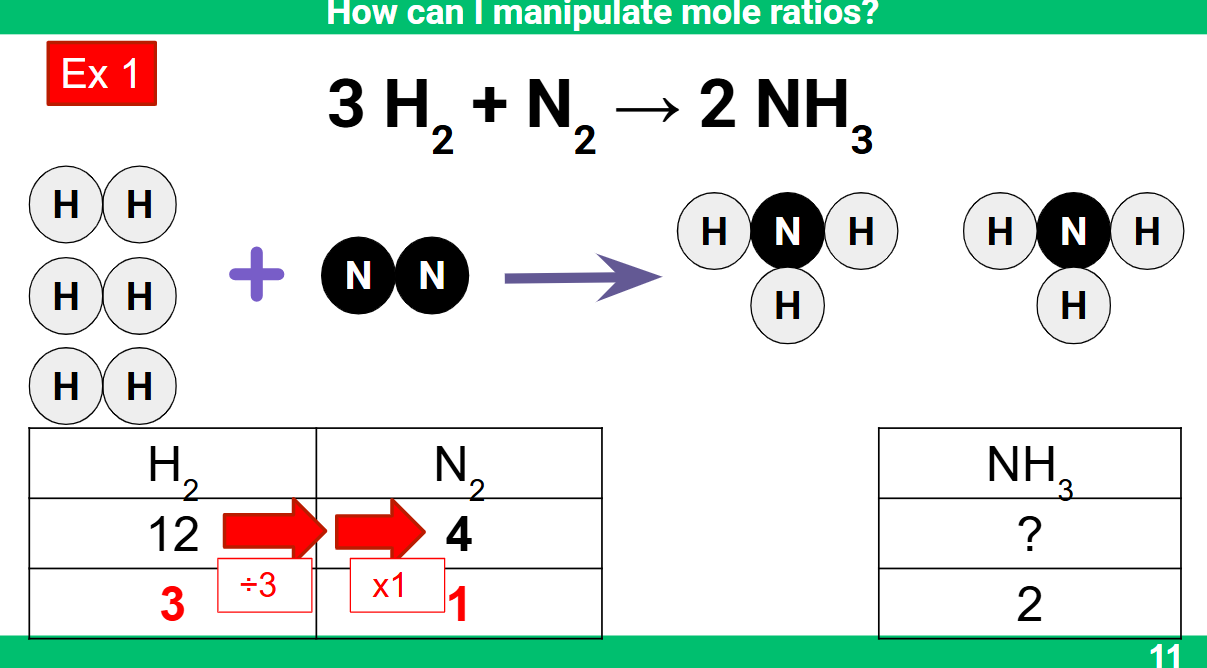
Calculating the mass:
Percentage yield:
Chemistry Unit 3:
Part 1: Organic Compounds
Organic Compounds
This is a large group of compounds which all contain carbon-hydrogen or carbon-carbon bonds but can also contain a small number of other atoms.
eg. Methane, C3H8O2N, C2H6O non examples: CO2 , Na2CO2
Homologous series:
Af family of organic compounds that have the similar chemical properties, but the physical properties can change(gradually) eg. as the carbon chain becomes longer the boiling point increases, and they share a general formula and change by a difference of CH2(gradual change) and the same functional group.
Hydrocarbons: type of organic compound but that only contains hydrogen and carbon eg. Butane, methane(CH4) and ethene etc.
Hydrocarbons with longer chains have a higher boiling point
Hydrocarbons with longer chains are less flammable
Hydrocarbons with longer chains are more viscous(thick and not runny) and are darker in colour
Hydrocarbons with longer chains create a smoky flame it burnt(not easyn to ignite tho
Alkanes: are the simplest type of hydrocarbons and are aturated compounds because no double carbon bonds
SUFFIX: -ane
GENERIC FORMULA: alkane= CnH2n+2
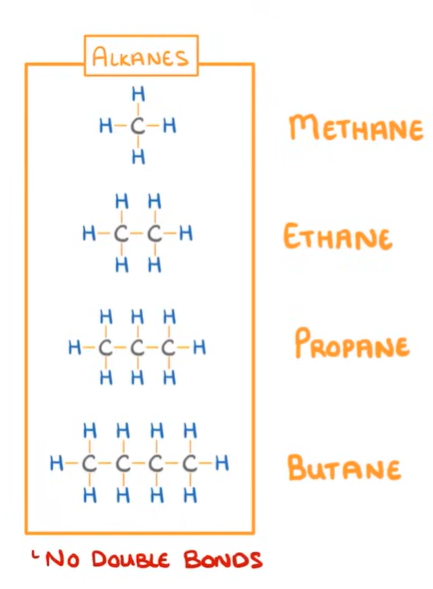
Trends in chemical properties—>
As you go down the group and chain length increases, boiling point increases
Shorter alkanes are more volatile because they evaporate more easily, whilst longer alkanes are viscous.
Shorter alkanes are more flammable
Uses:
Short alkanes are used as fuels(burn easily and release a lot of energy)
Alkenes: hydrocarbons that are unsaturated and contain a double carbon bond(this is their functional group).
eg.
SUFFIX: -ene
GENERAL FORMULA: CnH2n
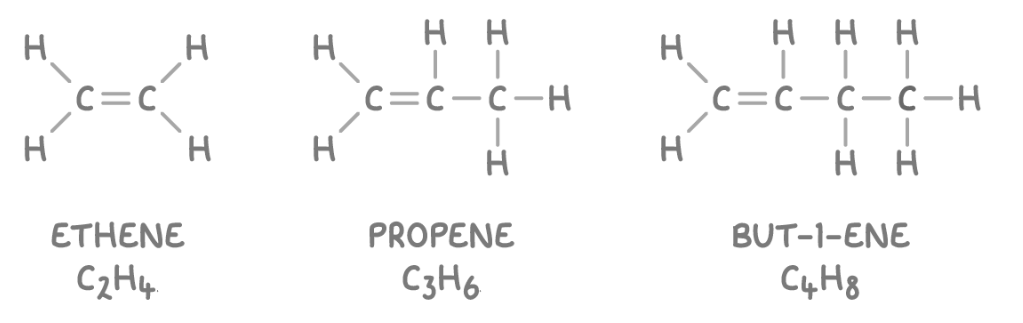
Chemical Properties(some cool stuff to know)—>
More reactive than alkanes due to the carbon double bond consisting of a sigma and pi bond.
Can participate in additional reactions.
Uses:
Make polymers-Alkenes serve as key monomers in the manufacture of plastics and synthetic fibers.
Ethanol production- Alkenes are involved in the hydration process to produce alcohols, which are widely used as solvents and in beverages.
Synthesis of fine chemicals- They are used as intermediates in the synthesis of pharmaceuticals and agrochemicals.
Addition reactions:
Polymerisation:
Carboxylic Acids: homologous series of organic compounds(not hydrocarbons tho) that has the functional group R-COOH.
SUFFIX: -anoic acid
GENERAL FORMULA: CnH2n+1COOH
—> put COOH at the end and take away one H because of the double bond between O=C to match valency of carbon.
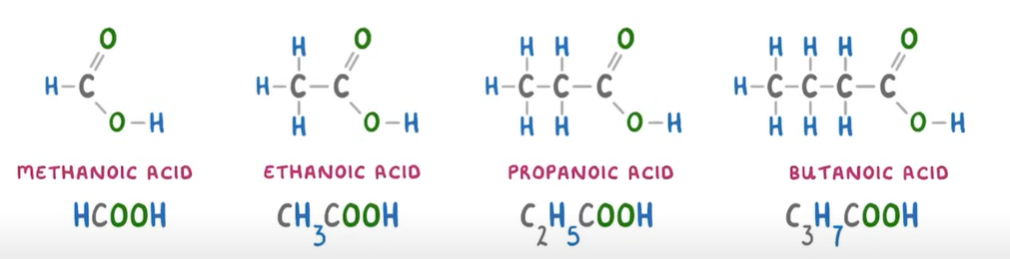
Chemical info—>
Are all weak acids that don’t fully ionise(the carboxylic acid molecules don’t all release their hydrogen ions), which means they only partially dissociate in solution, resulting in an equilibrium between the undissociated acid and the ions formed.
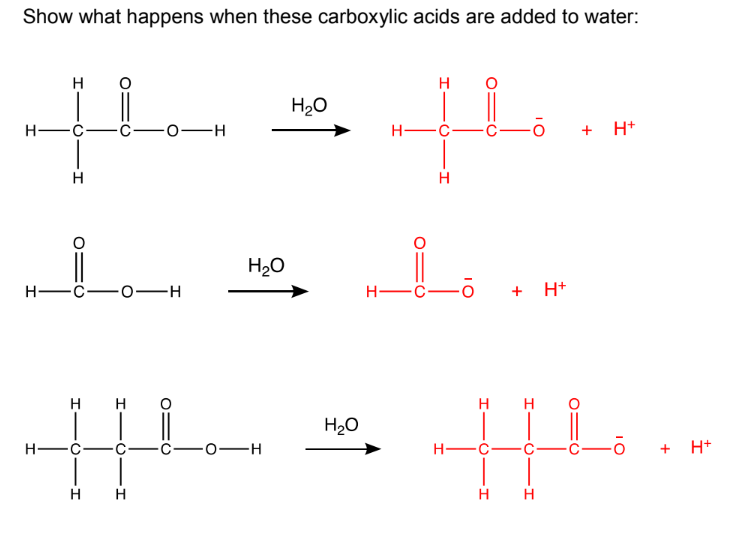
If reacted with a metal carbonate, it forms a salt, water and carbon dioxide (same for other acids).
Made by oxidising an alcohol using an oxidising agent
Alcohols: a homologous series of organic molecules with the functional group -OH(not hydrocarbons).
SUFFIX: -ol
GENERAL FORMULA: CnH2n+1OH
—>locate on which carbon the OH is at to name the alcohol correctly, put the number in between the root and suffix and always use the lowest number possible when counting. eg. Butan-1-ol or Butan-2-ol
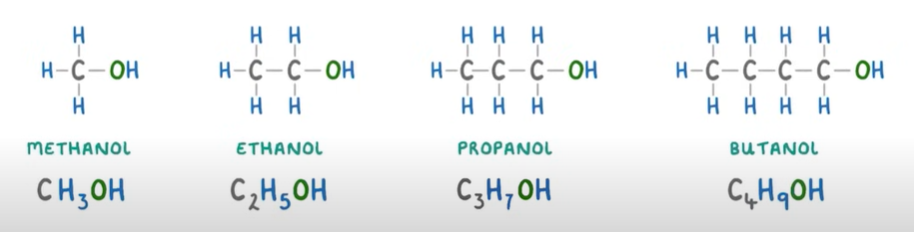
Chemical Properties—>
Flammable—> can undergo complete combustion with air(we can therefore use it as a fuel) and release a lot of energy
Soluble, however, alcohols are often used as solvents industrially bc they can dissolve hydrocarbons and lipids that water can’t dissolve(good solvents)
Can be oxidised to form carboxylic acids
ethanol is the alcohol used to make alcoholic drinks
Alkyl Groups: an R-group attached to the main chain and contains hydrogen and carbon only—>basically, you take the alkane form and chop off one of the “H”
Side groups:
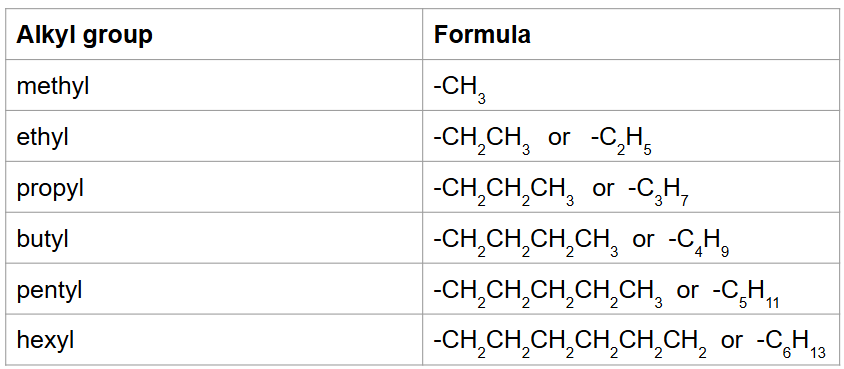
For naming:
Same root and suffix
Number the chain from the end closest to the alkyl group(lowest number)
—>include this number before the name of the alkyl group, separated by a dash ‘-’
eg. Here it’s a hexane bc it’s made of only carbon nd hydrogen, and there are no double c=c bonds, and the group added is a methyl group because there is only one carbon, the group is added on the 2nd carbon, so I need to add a 2, thus this compound is = 2-methylhexane
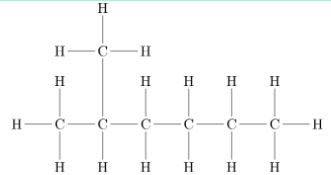
If there is more than 1 alkyl group—>
number from the end, which gives the lowest number
order the names of the alkyl groups alphabetically (ethyl before methyl)
if there are numerous of the same alkyl group on the same carbon you write di, tri- or tetra to indicate the number of similar alkyl groups ont’t hat carbon, NOTE the di and tri stuff is not considered int he alphabetical order.
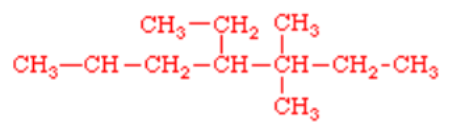
eg.
overall it’s an alkane and there are 7 carbons in the longest chain
—>heptane
There is 2 methyl groups on carbon 3—> I need to write di
There is 1 ethyl group on carbon 4
= 4-ethyl-3,3-dimethylhelptane
Halogenoalkane prefixes: organic compounds following the structure of an alkane but has one or more of it’s hydrogen atoms replaces by a halogen atoms.
Position of the halogen atoms on the carbon chain is annotated like for alkyl groups—> you say if it’s on the 1st, 2nd…. carbon and if there is more than one of the same you use the di, tri, tetra-
PREFIX: halogens name but with “o” at the end instead of “ine)
—> fluoro, chloro, bromo, iodo
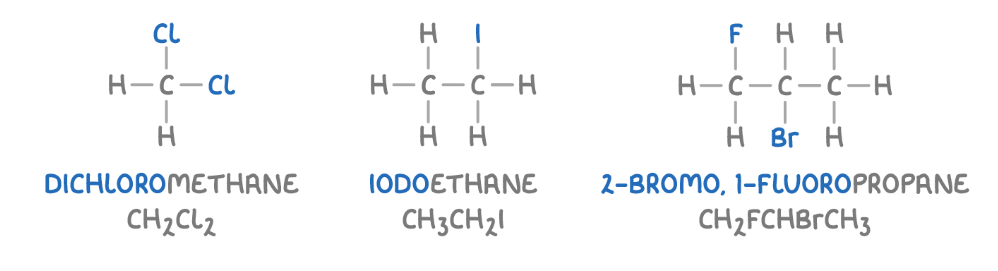
Eg for naming.
H and C atoms only and has 3 carbons in longest so it’s propane
Chloro on carbon 2 and a chloro on carbon 1 —> 1,2-dichloro
Branch with 1 carbon and replaced the H with methyl on carbon 2
= 1,2-dichloro-2-methylpropane
Isomers: molecules with the same molecular formula(same number of each atom) but different structural formulae.
—> usually differs in skeleton arrangement(branched or straight chain), position of functional group(different carbon atom) or the atoms form different functional groups(new bond but between atoms already there).
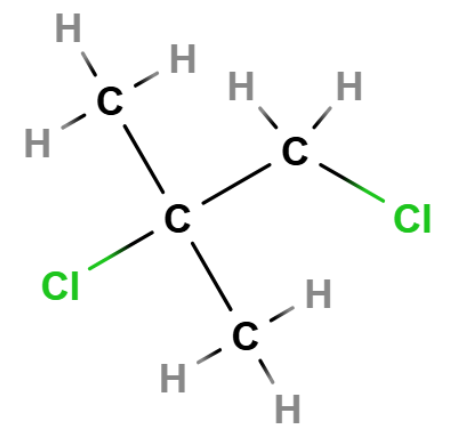
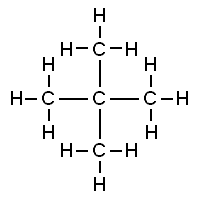
eg. pentane has 3 isomers—> molecular formula is C5H12
C-C-C-C-C in a chain with the hydrogens around—>pentane
Diamond shape with carbons to form dimethylpropane
methylbutane which has a methyl group on the first carbon so longest continuous chain is 4 not 5.
Types of isomerism: Chain, positional, functional group
Naming Organic Compounds Using the IUPAC Technique:
PREFIX=branches(any groups attached)
ROOT= name of the compound based on longest continuous chain of carbon atoms
SUFFIX= family-group it belongs to(name of functional group)
ICUPAC technique: an internationally recognised way to name organic and inorganic compounds by following the next steps:
Identifying the longest continuous carbon chain to determine the stem name.
Recognising the key functional groups to determine suffix names.
Numbering the carbon atoms based on the location of the functional group.
Including numbers and prefixes for any side chains present.
Applying multipliers like di-, tri-, and so on for multiple identical groups.
Assembling the complete name by integrating all these elements.
Breakdown Process:
Examples:
a)
b)
c)
Fractional Distillation:
A process that allows us to split up crude oil into its useful fractions by separating the hydrocarbons based on their boiling points to make them useful. This is an important industrial process since crude oil provides raw materials for fuels(via combustion) and plastics(via polymerisation).
Crude oil is a dark and viscous liquid that is flammable and is a mixture of different hydrocarbons with different chain lengths.
—>Fractions contain hydrocarbons with similar chain length, and so at each fraction, they are thus collected and filtered out.
The fractionating works by having a temperature gradient, which allows for the crude oil that is mostly in vapour form when entering the fractionating column, and so different parts of it condense at different levels of the column, separating the oil into fractions.
—> At the bottom, it is hottest, and that is where the longest chains of hydrocarbons condense back into a liquid because they have a higher boiling point than the other chains, since the amount of energy required to break the bonds is greater due to the greater intermolecular forces between the atoms. They form the first fraction, creating bitumen, which is used to cover roads.
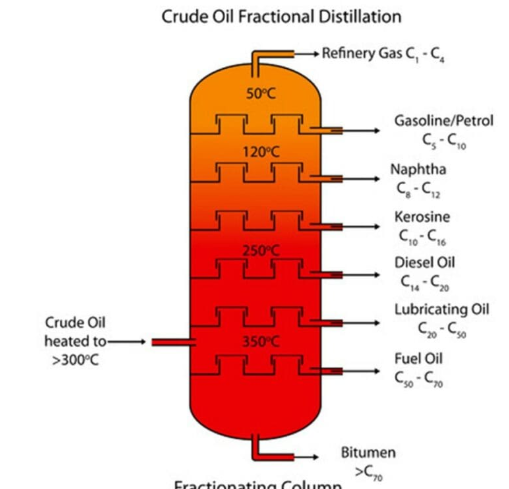
Process:
Crude oil is heated up at a high temperature(outside the fractionating column) and is then pumped into the column.
Some hydrocarbons condense inside the column at the bottom, forming bitumen.
The rest of the vapour continues to rise as it is hot and the bubble caps are slowing down the rate of the rising vapour to ensure that at each level, the fractions can be separated from the mixture.
Finally, short hydrocarbon chains condense at the top and higher up in the column where the temperature is lower because their boiling point is lower due to the weak intermolecular forces, meaning less energy is needed to break the bonds(which is what starts to happen when you boil something).
Distillation
condenser
Combustion
It’s burning by having a fuel that releases energy when it’s burnt since it stores potential energy when not burnt, it’s an exothermic reaction. For a combustion reaction to occur a fuel, oxygen and heat is required, if one is not present it won’t occur.
There are two types of combustion—>
Complete combustion: excess oxygen and thus produces carbon dioxide and water; it always produces these products(same). During this reaction, the air hole is open, so the flame is blue.
eg. Methane(CH4) when burnt= CH4 + O2 —> CO2 + 2H2O
Incomplete combustion: limited oxygen supply and thus produces a mixture of carbon(C), carbon monoxide(CO), carbon dioxide(CO2) and H2O and therefore, the products are not always the same(vary). During this type of combustion, the air hole is closed and the flame is yellow.
eg. 2CH4 + 2O2 —> 2CO + 4H2O
—> carbon monoxide forms because there isn’t enough O2 to fully oxidise the fuel so carbon atoms react only with 1 Oxygen molecule instead of 2.
Effects of these products:
CO2 is a GHG contributing to global warming
Carbon monoxide is toxic and deadly, known as a silent killer bc it’s odourless/tasteless/colourless
Unburnt carbon is a particulate contributing to air pollution
Polymerisation: Part 3
Polymers: large, chain-like molecules that can extend for thousands of atoms. They are made up of lots of repeting untis called monomers. Polymers are held together by:
Strong covalent bonds between atoms in molecules.
Weak intermolecular forces between molecules.
Because of the large size of polymer molecules, the intermolecular forces add up to be quite strong.
So, polymers are usually solid when at room temperature.
But, many polymers melt easily because the intermolecular forces remain less strong than chemical bonds.
Polymers: