bio
LOOK AT OBJECTIVES
biology = study of life
the seven traits that define living organisms
A. characteristics of living organisms
have organization: atoms = molecules - organelles - cells - tissues - organs - organ systems (not all organisms have all of these things)
acquire and use energy
maintain constant internal conditions (homeostasis)
have inherited information (DNA) that determines form and function
DNA has the instructions on how to make protein
all these organisms are composed of 1 or more cells
all organisms will respond to their environment (ex. walking outside when it’s hot makes you sweat. when it’s cold, you shiver)
all organisms reproduce
humans (the big six):
things we need (on a consistent basis) to survive: sleep, nutrition, exercise, socialization, learning, time outside
B. Science = collection of unified insights about nature, the evidence for which is an array of facts
facts are explanations thought to be true
science does not deal with absolute truths
list of scientific sources best to worst:
peer reviewed journals
reviewed texts (textbook)
science books
science magazines (like national geographic)
newspapers/TV
web/social media
theory = generalization that explains many observations - has been tested and researchers have yet to find evidence that disproves it - usually generates many hypotheses
hypothesis = tentative, testable explanation for observed phenomenon
C. Scientific method (describe it and be able to apply it to a given problem):
steps of ^
observation
hypothesis (never say it’s true cause you can’t ever prove anything is absolutely true)
experiment/test
conclusion
independent variable, dependent variable, and control samples
independent variable - factor of the experiment being tested (whatever is being changed or made different)
dependent variable - (what we use to measure the change, for ex. measuring the heart rate, which is the DV) response or change that occurs due to the IV
control sample - a sample where a the IVs are omitted
chapter 2 - chemical basis of life
A. element - a substance that can’t be reduced to a simpler substance (composed of atoms)
B. atom - basic unit of matter composed of protons, neutrons, and electrons
nucleus = very little space, but contains nearly all the mass of the atom
protons (+ charge, inside the nucleus) determine the atomic # of an element
ex. hydrogen - atomic # of 1, 1 proton - the number of protons never changes, therefor the atomic number is always equal to the number of protons
neutrons - (in nucleus) have no charge
isotopes - are just atoms of the same element with different weights because they have a diff number of neutrons but same number of protons.
ex. carbon (atomic # of 6) ~ C12 = 6 protons + 6 neutrons
12 is the mass number - it’s the protons + the neutrons
C14 is radioactive which means it’s a radio isotope - an isotope that has an unstable nucleus - loss of particles from the nucleus
ability to form four covalent bonds so it can bond with all types of things
electrons (surround the nucleus), ions and electron shells
electrons (- charge) have very little mass
ions - atoms that differ in the # of protons and electrons = pos or neg (more protons than electrons, will be pos. more electrons than protons, will be neg.)
electrons are found in shells, and each atom attempts to fill its outer shell
two electrons fill up the first shell
every shell after the first is full with eight electrons
atoms will form bonds to satisfy the requirements of their outer shell (they want to have a full outer shell, whether that means losing electrons to go back to a full shell or gaining electrons)
a neutral atom has an equal number of protons and electrons
C. Molecule - when two or more atoms join together to form something new. (they are bound together and they want to fill their outer shells)
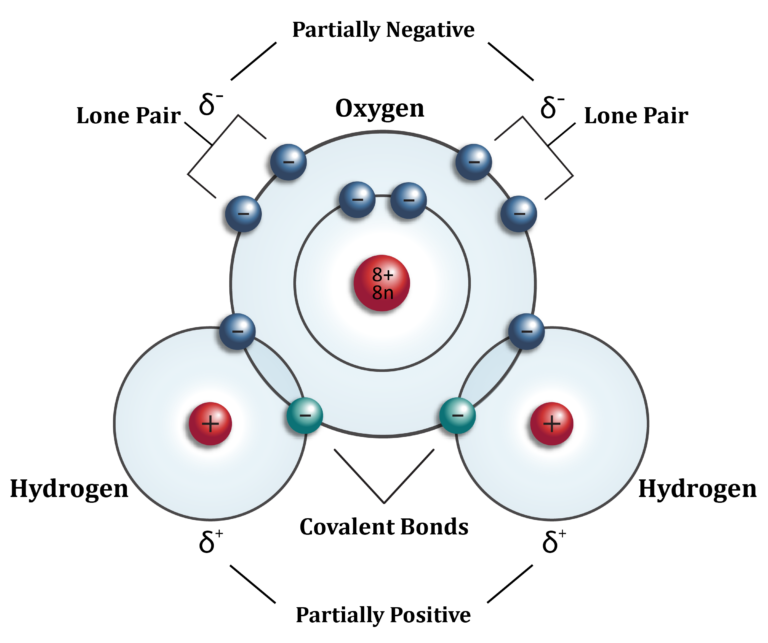
covalent bonds - bonds in which atoms share electrons
polar covalent bonds - shared electrons spend more time near the larger nucleus - the charge is on 1 end of the molecule
hydrogen is the biggest weakling and it will be pulled anywhere
hydrogen will be the positive end because all of its electrons are being pulled away from it
ex. h2o
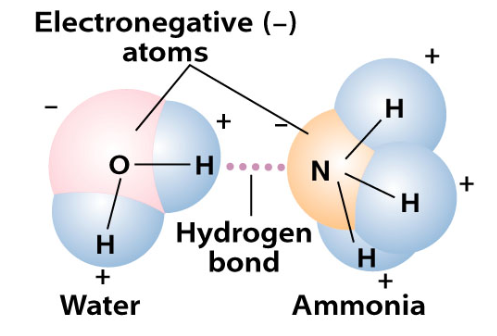
hydrogen bonds - formed when partially positive hydrogen atom in a polar covalent bond is attracted to a partially neg. atom in another polar covalent bond - opposites attract - ex. water and ammonia
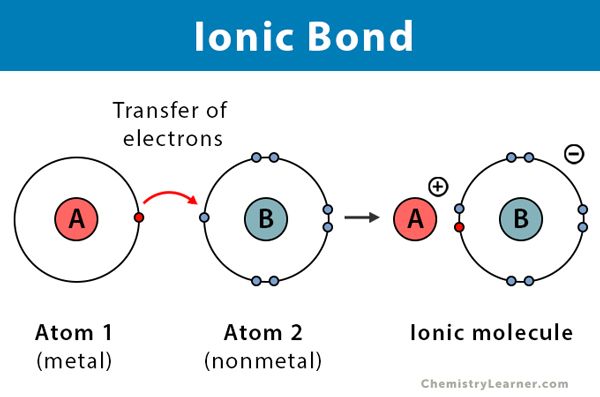
ionic bond - atoms bonded through attraction of oppositely charged particles (ions) - exchange electrons
D. solutions, solutes, solvents
solution - mixture of 2 or more substances (ex. solute dissolved in a solvent)
solute - substance that is dissolved in a solvent
solvent - the substance in which a solute is dissolved
sugar water - the solution
sugar is the solute
water is the solvent
E. water - provides the basis for life
freezes at 0 degrees celsius (32 degrees F) - ice = less dense dense than water - molecules spread apart
water has high specific heat
buffers climates - areas that are close to big bodies of water change temp more quickly(?)
june 21, dec 21 solecists
surface tension - water molecules bond together more strongly than water molecules to air
hydrophilic molecules - interact with water - anything that dissolved in water is a hydrophilic molecule - ex. salt
hydrophobic molecule - doesn’t interact with water - ex. oil
F. acids and bases
the pH of our blood is about 7.4
acids - lower pH
bases - raise pH
alkaline is a synonym for basic
measuring acidity or alkalinity
pH scale is from 0 - 14
0 = most acidic
more hydrogen ions (H^+)
14 = most basic
more hydroxide ions (OH^-)
raise pH and makes things neutral
7 = neutral
living organisms usually thrive in a pH of 6-8
some parts of organisms are very acidic - ex. stomach
when the pH in your body is too low it’s called acidosis
when the pH in your body is too high it’s called alkalosis
your bodily pH is very important - must stay close to neutral
G. Organic Compounds - always contain carbon
carbon has two electrons in its inner shell and four electrons in its outer shell
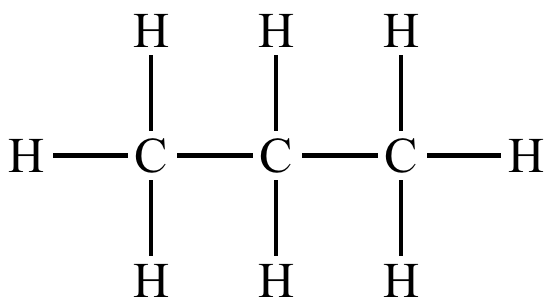
hydrocarbons - chain of carbons and hydrogens
ex. propane
functional groups - the groups of atoms in an organic molecule - where chemical reactions take place
FG - hydroxyl, carbonyl, carboxyl, amino, phosphate (know what molecules they are found in and how to draw)
H. Most organic molecules are repeated units
monomers - single units
polymers - string of monomers
I. Carbohydrates
contain C, H, O
monosaccharide (type of monomer):
C6H12O6 - Glucose
other ex. fructose, deoxyribose
Polysaccharide (polymer) - many monosaccharides linked together
starch - carbohydrate storage in plants
glycogen - how animals store carbohydrates
excess glucose that’s in bloodstream converts to glycogen and vise versa
cellulose - primary structural component of plants (provides rigidity). Animals can’t digest, only bacteria can
humans can’t digest cellulose so we don’t get any energy from it
cellulose increases how fast things move through your digestive system
chitin - (second structural polysaccaride) strengthens the exoskeleton of many organisms
ex. insects and arthropods
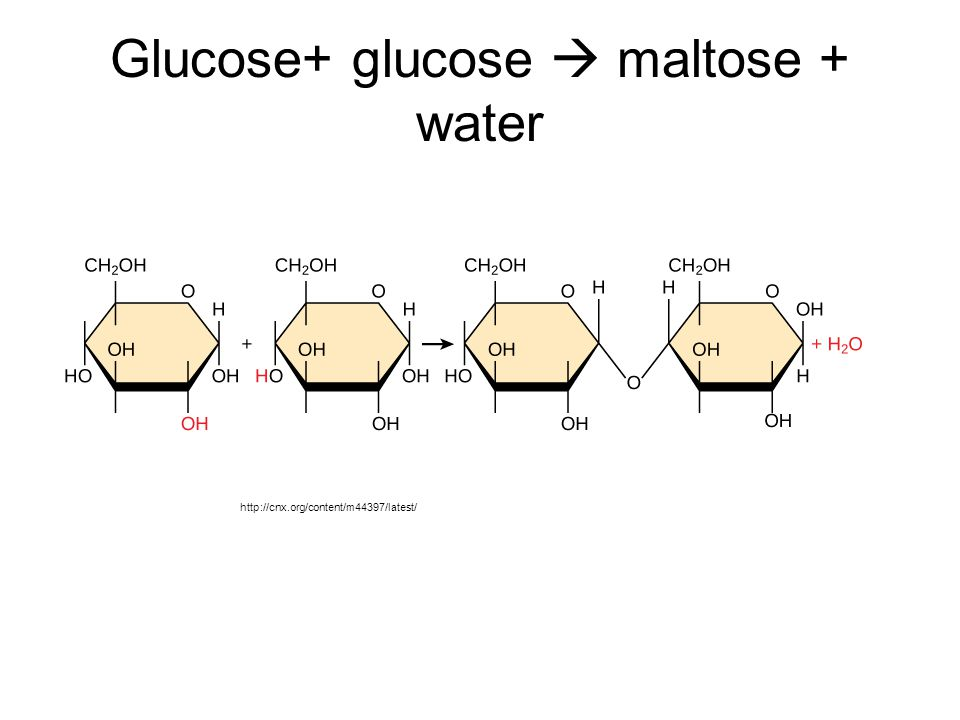
J. dehydration (cause giving off water in the process) synthesis —> when molecules bond to one another & produce H2O
ex. glucose + glucose = maltose
K. hydrolysis —> when a more complex molecules is split into simpler molecules and water is required
ex. maltose is broken down into two glucose molecules
L. lipids - oils, fats, steroids
characteristics of lipids
composed of C, H, O - more H
used for energy storage and insulation
hydrophobic
fat (type of lipid) - glycerol + fatty acids
fatty acids = a carboxyl and a long hydrocarbon chain
saturated fat vs unsaturated fat: single bonds vs double bonds from carbon to carbon
double bonds = unsaturated
single bonds = saturated
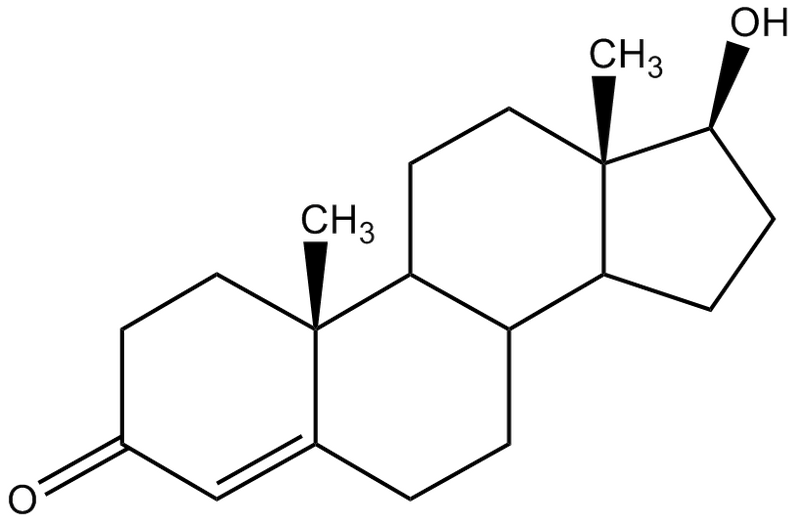
steroids (type of lipid): all have a set of 4 linked carbon rings
try to be able to identify a molecule by looking at
side chains are different
cholesterol is a steroid
ex. testosterone - every point of the hexagons have a carbon attached
phospholipids - make up the outer membrane of the cell
the cell membrane allows certain things to go in and out of the cell
2 fatty acids and a phosphate group
fatty acid tails are hydrophobic, but phosphate heads are hydrophilic
M. Protein - polymer made up of many amino acids (which are monomers)
all amino acids have an amino group and a carboxyl group attached to a central carbon
proteins consists of polypeptide chains made up of amino acids
protein functions are numerous and depend on the protein’s shape
monosaccharides make up the polysaccharides
amino acids are the monomers that make up protein, which are the polymers
nails and hair are made out of proteins like keratin
change in the environment can change the shape of a protein = denature the protein
ex. frying an egg
lipoproteins - capsule of protein that surrounds a globule of lipid
important for transporting lipids in and out of the bloodstream
LDL = low density lipoprotein - carry cholesterol to coronary arteries of the heart
really want this number to be low, less than 100
HDL = high density lipoprotein - cleans the cholesterol off your arteries and to your liver
want this number to be high, more than 40 for guys and more than 60 for girls
Apo’B - needs to be less than 90 - tested for people who have a family history of heart disease
N. Nucleic Acids - DNA and RNA is what we’ll talk about most
deoxyribonucleic acids (DNA) contains the instructions to make proteins
ribonucleic acid (RNA) takes DNA encoded instructions to the sites in cells where protein
DNA and RNA are composed of nucleotides
nucleotides are composed of a phosphate group, a 5 carbon sugar, and a nitrogen containing base (Guanine, Cytosine, Thymine, and Adenine)
half of DNA comes from mom, the other half is your dad
Chapter 3: Cells
A. Prokaryotes vs Eukaryotes
Prokaryotes:
archaea or bacteria
no nuclei
single celled
many do not use oxygen
no organelles
Eukaryotes:
everything else
nuclei
many are multicellular
most use oxygen
organelles
B. Eukaryotic Cell
nucleus - membrane bound department - contains DNA
plasma membrane - outer boundary of the cell
cytoplasm - region in side the plasma membrane but outside the nucleus (it’s everything in the cell but the nucleus)
organelles - structures within the cell which carry out specific functions
nucleolus - within nucleus - contains RNA & proteins - they combine to make ribosomal subunits - will make up ribosome
ribosome - structure that translates RNA to proteins
cytoskeleton - internal scaffolding that maintains the shape of the cell
cytosol - fluid in in the cytoplasm
rough endoplasmic reticulum (in cytoplasm) - network of plasma membranes that has ribosomes embedded in the surface
smooth ER - detoxifies potentially harmful substances and makes lipids
golgi complex - distribution center for proteins - tale proteins in, modifies them, and then ships them out to their final destination
vesicle - tiny membranous sacs in the cytoplasm
important vesicle: lysosome - organelles that contain enzymes that break down organelles and return them to the cytosol so they can be reused - also digest nutrients
peroxisomes - vesicles containing enzymes which break down fatty acids and amino acids
mitochondria - converts food energy into ATP - ATP is essential to our life
cytoskeleton
microfilaments - changes in cell shapes (actin, myosin)
intermediate filaments - skeleton
microtubules - move organelles from one area to another also cell extensions - cilia and flagella (which eat and destroy bacteria)
plant cells
central vacuole
may contain 90% of the cell volume
contains mostly water, but also nutrients and waste
cell wall - our cells just have cell membranes, not cell walls
located outside the cell membrane
cell walls are found in plants, bacteria, algae, and fungi
made of cellulose (polysaccharide)
plastids
chloroplasts - where photosynthesis takes place