Electrochemical and Potential Cells
Cell Diagram
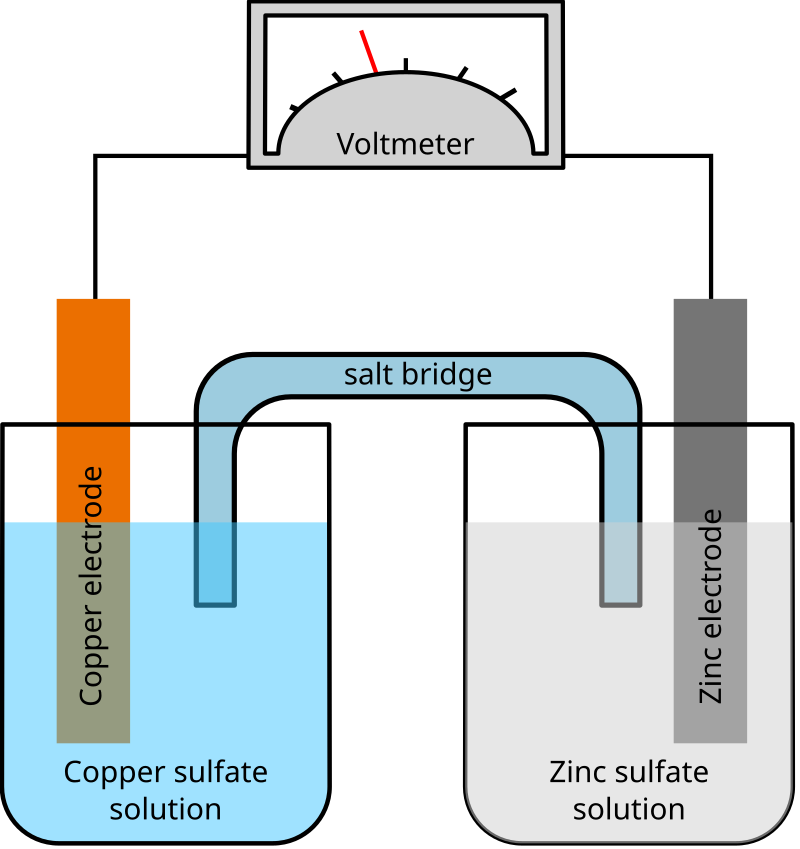
Key Points:
Anode: Zinc (Zn)
Cathode: Copper (Cu)
Electrolyte: Zinc sulfate (ZnSO₄) and copper sulfate (CuSO₄)
Cell Potential: Positive voltage indicates spontaneous reaction
Electron Flow: From anode (Zn) to cathode (Cu)
Salt Bridge: Maintains charge balance by allowing ion flow
Applications: Batteries, electroplating, corrosion studies
Half Equations:
Oxidation Reaction: Zn2+ (aq) + 2e ⇌ Zn (s) E° = -0.76 v
Reduction Reaction: Cu2+ (aq)+ 2e ⇌ Cu (s) E° = +0.34 v
Standard Hydrogen Electrode (SHE)
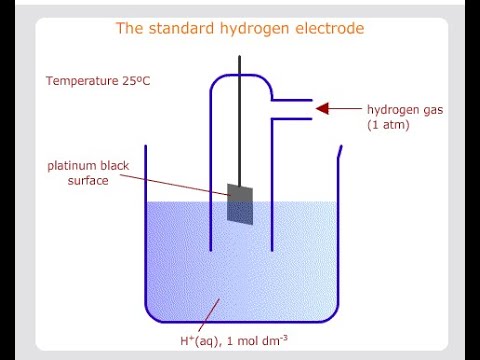
Key Points:
Standard Electrode Potential: 0.00 V
Reference Electrode: Used to measure other electrode potentials
Conditions: 1 M H⁺ concentration, 1 atm H₂ pressure, 25°C
Importance: Basis for electrochemical series
Applications: Used in pH measurement and electrochemical cells
Construction: Platinum electrode in contact with H₂ gas
Stability: Highly reproducible and reliable reference point
Half equation:
2H⁺ + 2e⁻ ⇌ H₂ (g)