EBP Lec 8: Clinical Decision Analysis and Screenings
Learning Objectives
Recall considerations of diagnostic testing, including different types of cost and maximising patient benefit
Apply the concepts of Clinical Decisions Analysis in the context of screening (such as the UNHS and adult ABR)
Recall outcomes of screening (hit, correct rejection, false alarm, miss) and applicability of sensitivity and specificity in a diagnostic context
Correctly outline what the current UNHS screening process is in Victoria
Recall the role of ABR in adult diagnostic testing, including when to refer for ABR and management pathways after ABR testing
Clinical decision analysis framework
Diagnostic testing in audiology must balance: time to perform tests, patient time, test costs, availability, and test accuracy (sensitivity, specificity).
Core question: Will the test outcome change patient management?
If yes, perform the test; if no, avoid it to save costs and time.
Example discussed: testing reflexes at 4 kHz is possible but often does not change management because it is time-consuming and uncomfortable, so it’s not routinely done.
Costs in healthcare (types of costs to balance with patient outcomes)
Financial for diagnostics (healthcare and personal): cost to healthcare system, out-of-pocket costs for patients
Loss of productivity
Financial for disease burden
Emotional costs (anxiety, distress for families, newborns during screening)
Cost of time (time off work, waiting, etc.)
Cost of not doing anything (i.e. costs of untreated conditions)
And
Transportation costs and access (getting to appointments)
Waste and sustainability (consumables, environmental impact)
Side effects or adverse events from tests
Examples in newborn screening context: emotional cost of referral for a baby who is later found not to have a condition; cost of missed diagnosis with potential long-term impacts if a condition is not treated early.
Clinical Decision Analysis
Two components of CDA:
Measure and Maximise
→ Evaluate usefulness of a test and ensure best outcome for patients
Examples:
Screenings for bowel cancer, hearing loss in newborns
Drug trial (cost of drug vs benefit)
Home vs hospital-based nursing program
We want the most efficient and cost-effective test that will detect the most pathology
Screenings
identifies individuals at risk of a target condition (what we’re screening for), then go on to have diagnostic testing for the condition
only have positive or negative outcome (“Pass” or “Refer”)
Diagnostic tests investigate further:
determine presence/absence
may determine additional information (e.g. hearing loss severity)
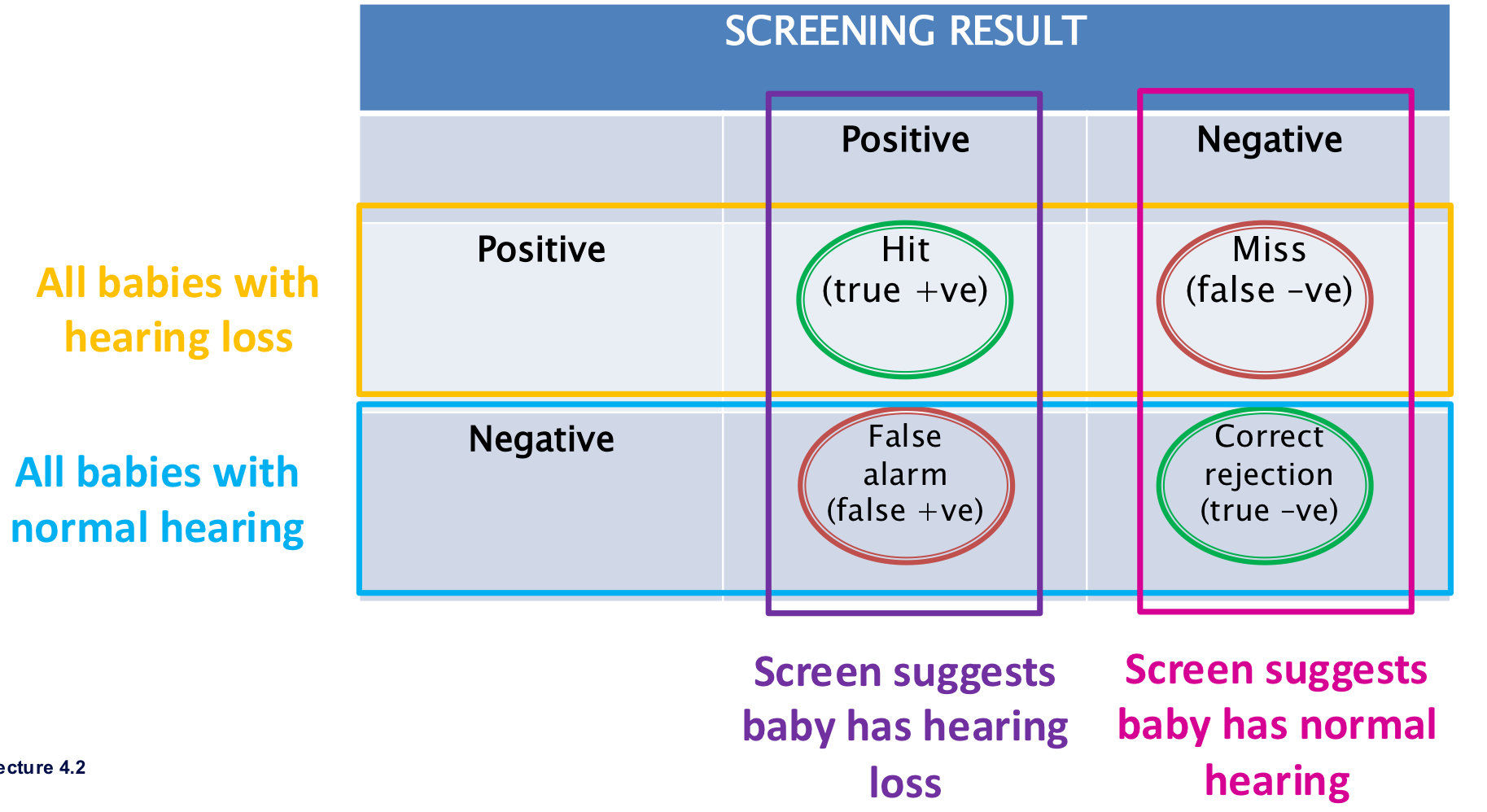
Outcomes (two-by-two table in newborn hearing screening example):
True Positive (TP): baby has hearing loss and screen is positive (referral).
False Positive (FP): baby does not have hearing loss but screen is positive (referral). *Error
True Negative (TN): baby does not have hearing loss and screen is negative (pass).
False Negative (FN): baby has hearing loss but screen is negative (pass). *Error
Consequences of errors:
False Positive: emotional cost, time and resource use for unnecessary diagnostic testing, possible less trusting.
False Negative: delayed diagnosis and management, potential higher downstream costs (e.g., speech-language delays, education costs and lower productivity in workforce).
Sensitivity and Specificity
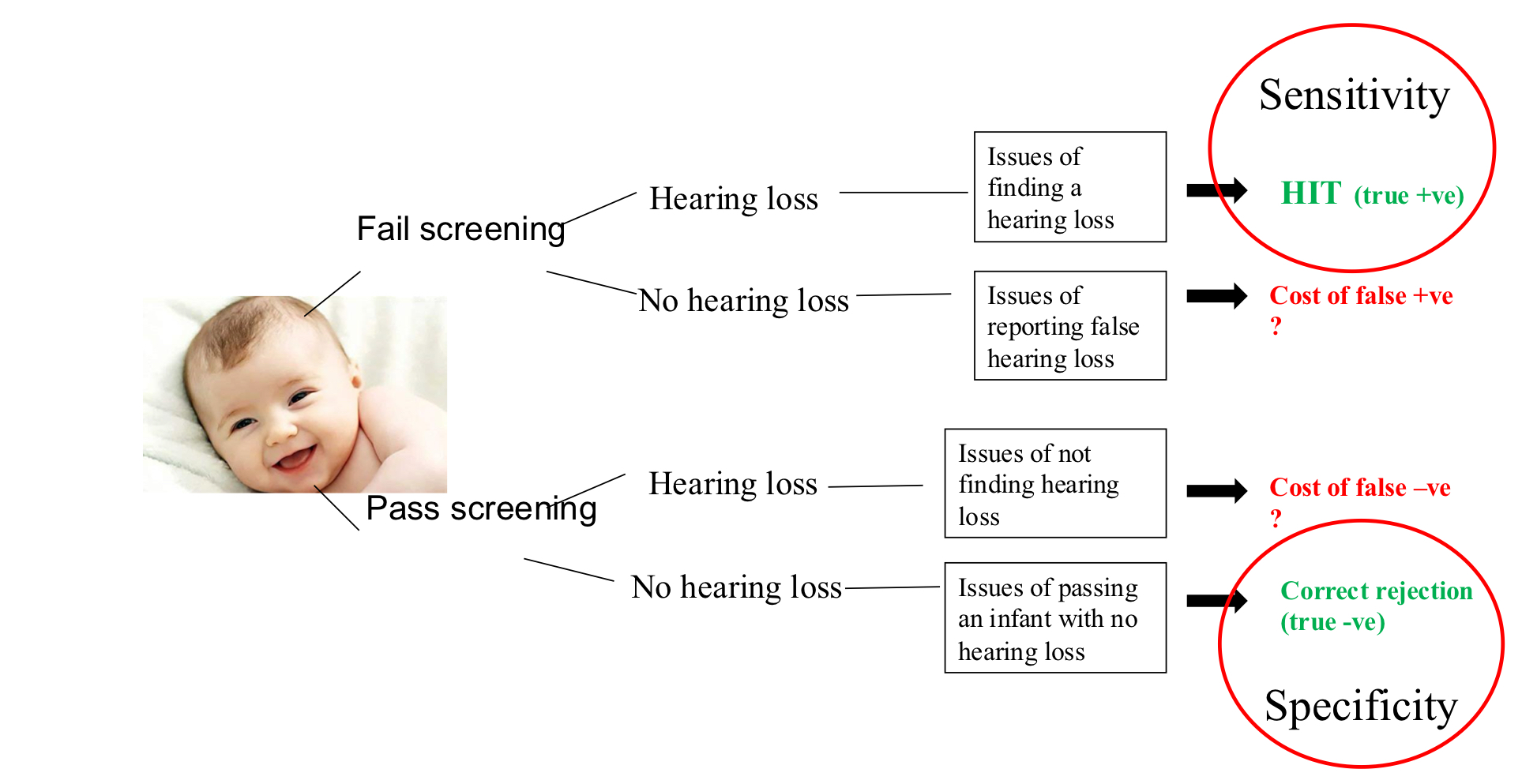
Sensitivity & Specificity → accuracy of screen
Sensitivity: a percentage of the test picking up target condition (detect it)
Specificity: a percentage of the test rejecting target condition (identify it’s not here)
Ideal screening would be 100% sensitive and 100% specific:
only fail screening if HL is present (100% sensitive)
only pass screening if no HL is present (100% specific)
Finding the balance
Ideally maximise true positives (i.e., high sensitivity to pick up all hearing-impaired babies)
however more sensitive a test is, false positives will also increase
consider the cost of missing a HL compared to referring lots of babies for hearing assessment that do not need it
Examples: fire alarm - prioritise sensitivity because cost (life & property) is so high
Newborn hearing screening (UNHS): overview and components
Target condition for universal newborn hearing screening (UNHS) generally defined as bilateral moderate or greater sensorineural hearing loss (SNHL).
Two main technologies:
Otoacoustic emissions (OAEs): fast, easy, cheap; measures outer hair cell function; does not detect auditory neuropathy; higher referral rate; quick screening.
Automated Auditory Brainstem Response (AABR or ABR): more expensive and slower but provides information about neural pathways up to the brainstem; can detect auditory neuropathy in some contexts; used as the primary screening tool in Victoria (AABR is automated and not waveform-interpreted by screeners).
CHIRP stimulus (time-delayed chirp) vs broadband click:
CHIRP presents low frequencies first and high frequencies later; results in more synchronous basilar membrane response and shorter screening times without changing intensity: is maintained.
CHIRP may miss some auditory neuropathy cases because synchrony is less critical for those patterns; some programs are adapting to avoid missing neuropathy.
Stimulus and target used in Victoria (Australia):
Stimulus intensity:
Target condition for screening: bilateral moderate or greater SNHL; not all hearing losses are targeted by the screen.
Two-stage ABR process (ABR1 and ABR2):
ABR1: first screening attempt; most babies pass or proceed to ABR2.
ABR2: second screening attempt for those who did not pass ABR1.
If ABR2 still not a pass in both ears, the baby is referred to audiology for further testing.
A “flip-flop” phenomenon: passing in one ear on ABR1 and failing on ABR2 cannot be summed across ears to declare normal hearing.
Flow and outcomes:
Green path: clear response (pass) in ABR1 or ABR2; move to community surveillance.
Brown path: refer to audiology if ABR is not clearly pass; may still result in a diagnosis that is different from the target condition.
Key terms:
“Pass” = screen indicates hearing is adequate for early speech and language development; does not prove normal hearing.
“Refer” = screen indicates risk; requires diagnostic audiology to determine presence/severity and management.
“Target condition” = condition the screening aims to identify (here, bilateral moderate or greater SNHL).
Two primary screening options and outcomes:
OAEs approach: faster, cheaper; higher referral rate; less likely to detect auditory neuropathy.
ABR approach: slower and more costly but provides more information; used in Victoria as the primary newborn screen (AABR).
Epidemiology and rollout:
The VicNIC trial demonstrated that the technology could balance sensitivity and specificity; rollout began in 2005; system evolved to aim for universal coverage with pre-discharge screening.
Population coverage around 99.7% (in Victoria in 2019), with some gaps (refusals or births outside hospital).
General population prevalence approx. for bilateral moderate or greater SNHL; NICU population has higher risk (~).
Population-level outcomes and costs (2019 data):
Target condition among those referred for diagnostic audiology around
Unilateral hearing loss of any degree comprised about 25 ext{%} of diagnostic results.
Permanent bilateral mild hearing loss about 12 ext{%}.
Practical implications:
Newborn hearing screening has dramatically reduced median age of diagnosis from over a year (historical with risk-factor screening) to around 1 month with UNHS.
Early identification enables earlier intervention and potentially better language outcomes, though benefits vary by degree of hearing loss and other factors (as shown by later data like the LONG-TERM LOCKY study).
Historical development in Victoria (to illustrate program evolution):
Pre-1992: no universal screening; risk-factor screening only.
1992–2005: risk-factor referral plus maternal/child health nursing behavioral screening (BOA-like approaches) with high resource use and poor reliability.
VICNIC trial (early 2000s) validated ABR/OAEs approach; 2005 rollout began, expanding to universal coverage.
2005 onward: pre-discharge ABR-based screening and statewide expansion; ongoing monitoring and data collection by ViSP.
Guthrie card and metabolic screening context in newborns:
Guthrie card: heel-prick blood spot collected at birth; used for screening for metabolic disorders with very high sensitivity and specificity; simple yes/no results.
Distinction: metabolic screening (Guthrie card) is a laboratory test measuring a defined disease state; there is a clear “has/has not” result.
In contrast, newborn hearing screening detects a risk for a sensory/retrocochlear issue and is not a definitive diagnostic test.
Two screening modalities and target conditions beyond newborns
Absence of a perfect test for all hearing loss levels means screening will identify some non-target conditions or milder losses; this is acceptable so long as follow-up and management pathways are efficient and accurate.
Retrocochlear pathology considerations (ABR as screening tool; MRI as confirmatory diagnostic tool):
ABR is less sensitive than MRI for detecting retrocochlear pathology (e.g., acoustic neuroma) but is cheaper, faster, and useful for triage.
Estimated false positive rate for ABR screening of about in some Danish studies; MRI false positive rate is under for acoustic neuroma.
The ABR-guided pathway: abnormal ABR → refer to ENT for MRI; normal ABR → still monitor and consider ENT if symptoms persist; not all GPs will refer, so ABR can strengthen referral rationale.
Cost and access considerations in triage:
MRI is more accurate but more expensive; ABR helps triage who should get MRI, reducing unnecessary MRI requests while not delaying care for those who need it.
In some settings, MRI cost equivalence to ABR changes the calculus; in others, ABR remains a cost-effective triage step.
Retroc Cochlear pathology: acoustic neuroma/vestibular schwannoma
Anatomy and presentation:
Acoustic neuroma (vestibular schwannoma) is a benign tumor at the cerebellopontine angle (CPA) where pons, medulla, and cerebellum meet.
Common presentation: unilateral or asymmetric SNHL, tinnitus, balance disturbances; facial weakness or other cranial nerve symptoms can appear as the tumor grows.
Diagnostic pathway and management decisions:
Diagnosis: usually MRI; ENT involvement for treatment planning and monitoring.
Audiological indicators that raise suspicion: unilateral SNHL, asymmetrical SNHL (sensorineural asymmetry), absent reflexes, and speech rollover.
Speech rollover in testing:
Definition: Speech score decreases when presenting at higher levels, indicating possible retrocochlear pathology.
Practical threshold: a drop of 20 percentage points or more from the maximum score is considered significant rollover.
Example: maximum score 75%; at higher level score 50% → rollover significant (
75% − 50% = 25% drop).Important caveats: rollover alone is not diagnostic; used to strengthen the case for further retrocochlear investigation.
Sensorineural asymmetry criteria for retrocochlear concern:
A significant sensorineural asymmetry is a difference in air- or bone-conduction thresholds of at least across adjacent frequencies.
Change from octave-only to include non-octave (interpolated) frequencies increases sensitivity but also false positives; current practice uses adjacent/interpolated frequencies (not just octaves).
If an asymmetry is detected, ABR and ENT/MRI referrals are considered; asymmetries are considered sensorineural in origin (bone conduction used for comparison).
Reflex patterns and signs:
Absent or abnormal acoustic reflexes (tone burst; tone decay) can indicate retrocochlear pathology when combined with other signs.
Management considerations for amplification:
In retrocochlear cases, amplification (hearing aids) must be used cautiously due to potential distortion and rollover; rewards depend on location and severity of pathology.
Referral pathway and monitoring:
Suspected retrocochlear pathology → referral to ENT with ABR as part of the audiological triage; MRI for confirmation; ongoing monitoring (e.g., yearly) if pathology is slow-growing.
ABR in screening vs retrocochlear diagnosis; practical considerations
ABR screening advantages:
Provides information beyond a pure cochlear check; helps identify issues along the auditory pathway up to the brainstem.
Two-stage ABR improves accuracy and reduces missed cases while keeping turnaround time manageable.
ABR limitations and triage role:
ABR is less sensitive than MRI for detecting retrocochlear pathology; false positives exist (e.g., ~ in some reports).
MRI is more accurate but costly; ABR can triage who should proceed to MRI, balancing sensitivity, specificity, cost, and access.
Diagnostic pathway after ABR:
Abnormal ABR: report to GP, recommend ENT referral for MRI; provide patient-facing language about the test and the need for specialist follow-up.
Normal ABR: still may warrant ENT follow-up in some cases; ABR does not completely rule out retrocochlear pathology.
Considerations for tele-audiology and access:
Digital and remote screening/tracking can impact costs and timeliness; planning for access in remote populations is part of optimizing the system.
CMV screening and emerging screening approaches
CMV (cytomegalovirus) as a contributor to congenital SNHL:
CMV infection can cause sensorineural hearing loss and other neurodevelopmental issues if untreated.
Screening approaches:
Guthrie card (birth blood spot) has poor sensitivity for CMV; retrospective testing is possible but unreliable for early intervention.
Saliva or urine PCR testing within the first 21 days of life has higher sensitivity and specificity for CMV, enabling earlier antiviral interventions when needed.
Guidelines suggest offering salivary CMV testing when a child fails the newborn hearing screen within the first 21 days to enable timely management.
Practical implication:
Incorporating CMV screening with newborn hearing screening could improve outcomes by enabling earlier antiviral treatment and better developmental trajectories if CMV is the underlying cause.
Population-level benchmarks and key performance indicators (KPIs)
Joint Committee on Infant Hearing (JCIH) benchmarks for resource-rich environments:
Population coverage for UNHS: high (near universal access).
Referral rate should be low enough to not overwhelm diagnostic services while retaining sensitivity.
Follow-up rate to diagnostic testing after referral should be high.
Diagnostic accuracy and enrollment in early intervention programs should be achieved by defined ages: aim for screening by 1 month, diagnostic assessment by 3 months, and early intervention by 6 months (the 1-3-6 rule).
Evolution of KPIs and timelines:
In the Victoria program, the aim has been to achieve rapid progression through the pathway with a pre-discharge (in-hospital) ABR screening and subsequent diagnostic processes.
2019 ViSP data show breakdowns by what the diagnostic audiology findings were (Target Condition, unilateral loss, permanent bilateral mild loss, etc.).
Practical data and examples from the Victorian program
Population coverage and workflow (2019 ViSP data):
Coverage: around population coverage; slight gaps due to refusals or out-of-hospital births.
Diagnostic follow-through: strong capacity for diagnostic audiology services in the state; differences exist across states (e.g., NSW) in cross-border patient flows.
Diagnostic results by screening: about had the target condition; unilateral loss of any degree ~; permanent bilateral mild loss ~.
Testing intensity and pacing:
The stimulus used in screening: ; the CHIRP approach reduces time to screen but must balance sensitivity for neuropathies.
The proportion of infants exiting after ABR screening to the surveillance process is substantial, illustrating the efficiency of the two-stage ABR model.
Turnaround times and milestones:
Time from screen to diagnostic audiology and then to hearing aid fitting (where applicable) is tracked with green boxes showing median days; the system aims to minimize time to diagnosis and intervention.
The LOCKY study (Longitudinal Outcomes of Children with Hearing Impairment):
A multi-state study comparing early versus late identification and amplification.
Used BE4FA (Better Ear Four-Frequency Average: average of thresholds at 500, 1000, 2000, 4000 Hz) to assess language outcomes at age 5 across three severity bands (30 dB HL, 50 dB HL, 70 dB HL).
Findings: Early identification and amplification yield greater language outcomes for more severe hearing losses (70 dB); for mild losses, the difference is smaller; early amplification may not always translate into large gains for mild losses due to several interacting factors.
Clinical interpretation from LOCKY data:
Early detection and amplification are most clearly beneficial for more significant losses; policy and clinical decisions about screening emphasis should consider this differential impact.
Practical implications for clinical decision-making and patient care
The clinical decision analysis framework informs which tests to perform in order to maximize patient benefit while minimizing costs and patient burden.
Screenings are designed to efficiently triage who needs full diagnostic workups; the cost-benefit balance is central to program design and ongoing policy decisions.
In newborn screening, high catchment and adherence to follow-up are essential for real-world effectiveness; trust and communication with families are crucial to ensure adherence to referral pathways.
For retrocochlear pathology, ABR serves as a screening/triage tool rather than a definitive diagnosis; MRI remains the gold standard for confirmation but is more resource-intensive.
Ethical and practical implications:
False positives can cause parental distress and unnecessary medical testing; false negatives can delay critical early intervention.
System design should minimize harm from false results while ensuring high sensitivity for treatable conditions.
Access and equity considerations are central: remote populations, tele-audiology, and cross-border service access all affect outcomes.
Next steps in education and practice:
Understand the differences between OAEs and ABR, including suitability for different target conditions and the implications for referral rates.
Be comfortable with the ABR screening workflow, including two-stage ABR, CHIRP vs broadband click stimuli, and interpretation caveats (e.g., flip-flop, rollover).
Be able to discuss with families what a referral means and what steps will occur next in the diagnostic pathway.
Keep in mind the evolving guidelines (JCIH 1-3-6; later pushes toward even earlier intervention) and how they apply to local health service realities.
Quick reference: key terms and formulas (LaTeX)
Target condition for UNHS (typical): bilateral moderate or greater SNHL.
Two-by-two outcomes (screening): TP, FP, TN, FN.
Sensitivity and Specificity:
BE4FA (Better Ear Four-Frequency Average):
Targeted hearing loss definitions (illustrative):
Bilateral moderate or greater SNHL: thresholds in both ears at or above the moderate level across key frequencies.
CHIRP stimulus and intensity in UNHS:
(intensity used in many ABR screenings)
Time targets in JCIH 1-3-6 framework (common baseline):
Screen by age
Diagnostic assessment by age
Early intervention by age
Population and prevalence (illustrative Victorian data):
Population coverage: (99.7%)
General prevalence: for bilateral moderate or greater SNHL
NICU prevalence:
ABR false positives (in some reports):
ABR screening intensity and outcomes:
ABR1 pass rate and ABR2 pass rate determine the majority going to green path (surveillance) vs refer-to-audiology paths.
Clinical Decision Analysis Framework
Purpose
To balance:
Diagnostic accuracy and sensitivity,
Time and cost efficiency, and
Impact on patient management and outcomes.
Key Considerations
Time (clinician + patient)
Cost (financial + emotional + societal)
Test sensitivity and specificity
Availability of equipment/personnel
Whether the test result will change patient management
“If an extra test won’t alter management, perhaps it shouldn’t be done.”
Example
Reflex testing at 4 kHz: technically possible, but time-consuming, uncomfortable, and rarely changes management → typically unnecessary.
3. Types of Costs in Healthcare
Category | Description / Example |
|---|---|
Financial | Out-of-pocket costs, healthcare system burden, consumables, sustainability. |
Productivity | Time off work, reduced income, caretaker costs. |
Emotional | Stress, uncertainty (e.g., parents during newborn screening). |
Time | Duration of appointments, follow-ups, travel. |
Cost of inaction | Untreated hearing loss → greater long-term rehabilitation costs. |
4. Screening vs. Diagnostic Testing
Aspect | Screening | Diagnostic Test |
|---|---|---|
Purpose | Identify people at risk | Confirm presence / quantify severity |
Cost | Low | High |
Coverage | Large populations | Targeted |
Outcome | “Pass” = low risk, “Refer” = needs further testing | Quantitative data for management |
Examples | Newborn hearing, bowel, breast, metabolic disease screening | Audiometry, full ABR |
Clinical Decision Analysis Goal
Maximise benefit (catch true cases early)
Minimise cost (limit unnecessary diagnostics)
Screenings identify those likely to have the condition, then refer for diagnostics.
5. Screening Outcome Matrix
True Condition | Screen Result Positive (Refer) | Screen Result Negative (Pass) |
|---|---|---|
Condition Present | True Positive (Hit) → correct referral | False Negative (Miss) → undetected condition |
Condition Absent | False Positive (False Alarm) → unnecessary referral | True Negative (Correct Reassurance) |
Cost of Errors
False Positive: emotional distress, wasted time, loss of trust, extra costs.
False Negative: more severe → missed diagnosis, delayed speech/language development, missed cochlear implant window, lifelong educational and economic consequences.
6. Sensitivity & Specificity
Term | Definition | In Newborn Screening Context |
|---|---|---|
Sensitivity | Ability of test to correctly detect those with the condition | % of babies with hearing loss that the screening correctly refers |
Specificity | Ability to correctly identify those without the condition | % of babies with normal hearing who pass the screening |
High sensitivity → fewer missed cases but more false alarms.
High specificity → fewer false alarms but risk missing true cases.
Must balance both depending on cost of errors.
Analogy
Fire alarm: must detect every real fire (high sensitivity), even if it occasionally rings for burnt toast (false positives).
7. ABR as a Screening Tool
Two Major Uses
Newborn Hearing Screening
Retrocochlear Pathology Management (adults)
8. Screening Example – Newborn Hearing Screening
Laboratory vs. Functional Screening
Metabolic (e.g., Guthrie card): clear yes/no lab result.
Hearing: subjective threshold definition; no single “blood test”.
Cost Justification
Late-identified profound loss ≈ $1 million educational support per child.
~70–80 babies/year in Victoria with bilateral, ≥ moderate SNHL → $70–80 million impact vs. far lower screening cost.
9. History of Victorian Newborn Hearing Screening
Period | Milestone |
|---|---|
Pre-1992 | No systematic screening. |
1992–2005 | Risk-factor questionnaires + behavioural screening by maternal-child-health nurses (BOA-like, unreliable). |
2005 | VICNIC NHS trial proved AABR tech validity → rollout begins. |
2005–2012 | Gradual expansion from tertiary hospitals → state-wide programme (Victorian Infant Hearing Screening Programme – VIHP/VISP). |
NICU risk: 1 in 100 babies with ≥ moderate SNHL (vs 1 in 1000 general).
Screening before discharge ensures maximum coverage.
10. AABR Technology
AABR = Automated Auditory Brainstem Response.
Uses algorithmic waveform template → screener doesn’t interpret waveforms.
Detects presence/absence of neural response to sound.
OAE | AABR |
|---|---|
Tests outer hair cell function | Tests neural synchrony (8th nerve + brainstem) |
Faster, cheaper | Slower, costlier |
Misses auditory neuropathy | Detects auditory neuropathy |
High false-refer rate (10–20%) | Low false-refer rate |
In Victoria, AABR is used exclusively to avoid missing auditory neuropathy.
11. Stimulus and Procedure Details
Stimulus type: initially click, now CE-CHIRP (since 2021)
Low → high frequency sequencing improves synchrony.
Faster test time; still 35 dB nHL intensity.
Target condition: Bilateral, ≥ moderate permanent SNHL.
CHIRP may slightly reduce detection of auditory neuropathy (fewer failures due to better synchrony) → clinical monitoring under revision.
12. Newborn Hearing Screening Pathway
Terminology
Pass = auditory nerve responding at 35 dB nHL → adequate for speech/language development.
Refer = needs diagnostic follow-up (“ticket to audiology”).
Not “fail” → family-friendly language.
Two-Stage AABR Protocol
AABR 1: Initial screen.
Must obtain clear response in both ears in one session.
If not → AABR 2.
AABR 2: Repeat screen (separate day).
If still not clear both ears → referral to diagnostic audiology.
“Right-ear AABR1 pass + left-ear AABR2 pass ≠ overall pass.” (called flip-flop).
Flow Outcomes
Most babies → AABR 1 pass → discharge.
Some → AABR 2 pass → community surveillance.
Few → refer → diagnostic ABR.
13. JCIH Benchmarks (“1-3-6” & “1-2-3” Rules)
Stage | Target Age | Updated Goal (2019) |
|---|---|---|
Screen completed | 1 month | (same) |
Diagnosis confirmed | ≤ 3 months | ≤ 2 months (123) |
Early intervention enrolled | ≤ 6 months | ≤ 3 months |
Australia meets/exceeds benchmarks due to strong infrastructure.
Emphasis: earlier = better → stronger speech/language outcomes.
14. Victorian Performance (2019)
Population coverage: 99.7%.
Median timeline:
Diagnostic completion ≈ < 60 days.
HA fitting ≈ < 70 days.
Follow-up: High (due to strong public network).
Outcomes: Among diagnostic cases:
~30% = target condition.
~25% = unilateral losses.
~12% = bilateral mild loss.
Confirms expected false-refer rate (intentional safety margin).
15. Impact of Universal Screening
Median Age of Diagnosis
Era | Median Diagnosis Age |
|---|---|
Pre-1989 (no screening) | ~24 months |
Risk-factor only (1992–2005) | ~12–15 months |
UNHS (since 2005) | ~1 month |
→ Enables timely fitting and improved outcomes.
16. The LOCHI Study (Longitudinal Outcomes of Children with Hearing Impairment)
Conducted by NAL (Hearing Australia).
Compared states with vs. without newborn screening.
Design
Children with bilateral ≥ moderate SNHL.
Compared age at first fitting (months) vs global language score at 5 years.
Findings
Loss Severity (BE4FA) | Impact of Early Fitting |
|---|---|
Severe (~70 dB HL) | Major benefit → early fitting → higher language scores. |
Moderate (~50 dB HL) | Moderate benefit. |
Mild (~30 dB HL) | Minimal long-term difference. |
Early detection is most critical for greater losses; less measurable benefit for mild losses, though still eligible for intervention.
17. Transition to Retrocochlear Pathology Section
Definition
Retrocochlear = beyond the cochlea (8th nerve → brainstem → temporal lobe).
Most cases → vestibular schwannoma / acoustic neuroma.
Pathophysiology
Benign (non-cancerous) tumour in cerebellopontine angle (CPA).
Compresses adjacent nerves → auditory + vestibular symptoms.
Common Presentation
Middle-aged adults.
Unilateral or asymmetrical:
Hearing loss,
Tinnitus (differs between ears),
Aural fullness,
Distortion,
Mild imbalance.
Severe/late → facial weakness, swallowing difficulty, headaches.
Diagnosis
MRI (gold standard).
Audiologists → identify red flags → refer for medical imaging.
18. Audiological Red Flags / Findings
Test | Possible Retrocochlear Signs |
|---|---|
Audiogram | Asymmetric SNHL (but may be normal). |
Acoustic Reflex | Absent or tone-decay pattern. |
Speech Audiometry | Rollover > 20 % decrease in score when level ↑. |
Speech Rollover
Normally, louder = better clarity.
In retrocochlear loss, louder = poorer clarity (distortion).
Criterion: ≥ 20 % absolute drop from max score.
Suggests 8th-nerve pathology → supports referral for ABR / MRI.
19. ABR in Retrocochlear Screening
Detects delayed or absent Wave V on affected side.
Compared interaural Wave V latency (> 0.4 ms difference = abnormal).
Used as triage tool before MRI.
Limited ABR access in private clinics may delay referral; clinical protocols vary.
20. Key Clinical Reasoning Principles
Concept | Application |
|---|---|
Triaging | Use screening/ABR to prioritise high-risk patients for MRI. |
Resource awareness | Tests should alter management decisions. |
Ethical balance | Consider emotional, temporal, and financial costs. |
Communication | Replace “fail” with “refer” for sensitivity and clarity. |
✅ Summary Table
Topic | Essential Figures & Facts |
|---|---|
AABR intensity | 35 dB nHL |
Stimulus type | CE-CHIRP (low → high freq) |
Target condition | Bilateral ≥ moderate SNHL |
Screen type | 2-stage AABR |
Benchmarks | 1-2-3 (screen ≤ 1 mo; diagnose ≤ 2 mo; intervene ≤ 3 mo) |
False negatives > false positives | Missing hearing loss = highest cost |
Retrocochlear indicators | Asymmetry, rollover ≥ 20 %, reflex abnormalities |
Diagnosis confirmation | MRI |