Changes of state and the particle model
Density of materials
Density tells us how packed matter(mass) is in a specific space(volume).
Density=mass/volume
Density=Kg/m3 g/cm3
Mass=Kg/g
Volume=m3 /cm3
Solids: Solids have a fixed volume and they cannot be squashed. They also have a fixed shape which cannot be changed and they do not flow so they cannot be poured. Solids also have a high density as the particles are closely packed together.
Liquids: Liquids cannot be squashed and they have a volume which is fixed. They differ from solids due to being able to flow easily Although liquids are dense, they have a lower density than solids.
Gases: Gases are easy to squash. The particles are very far apart which is why gases are less dense than liquids.
*An object will float if its density if its lower than the density of the fluid is is placed in.
Changes of state
Changes of state are physical changes which differ from chemical changes because the material recovers its original properties if the change is reversed.
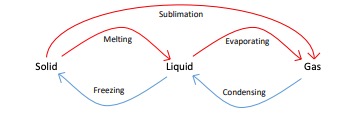
Mass is conserved during a state of change.