Reactions of benzene
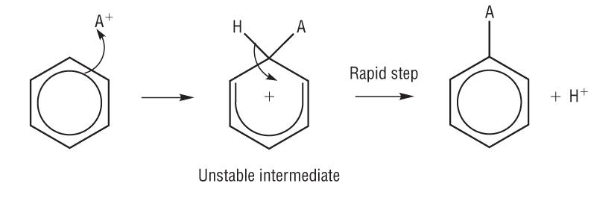
Electrophilic substitution of benzene
A+ = any electrophile
be really careful not to draw the delocalised ring too long
do not extend past the two neighbouring carbons (correct shown in step 2)
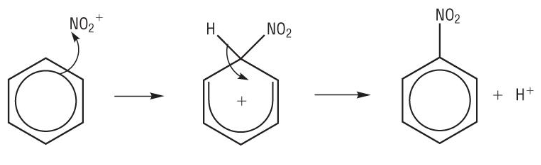
Nitration of benzene
mechanism
Formation of the electrophile
sulphuric acid is acting as acid
nitric acid is acting as base
also means sulphuric acid acts as a catalyst
regains H+ ion from reaction so never used up
hence sits above arrow in full equation
done at 50C
monosubstitution

overall equation
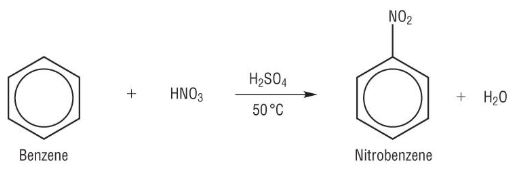
Nitration summarised
electron ring attracts electrophiles
a bond forms using delocalised electrons
the aromatic ring is lost briefly before electrons from the hydrogen bond restore the delocalised system
sulphuric acid acts as a catalyst and is restored by the addition of the hydrogen ion formed
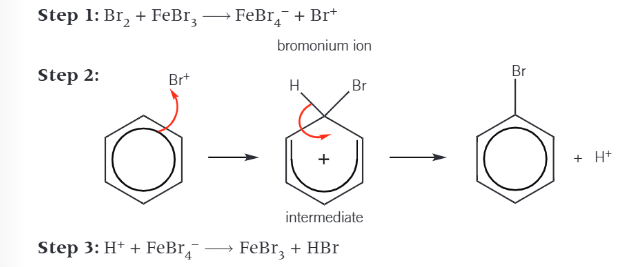
Halogenation of benzene
formation of the electrophile uses a halogen carrier
induces polarisation and produces a halogen electrophile
FeBr3 is regenerated in step 3 thus acts as a catalyst
Cl2 and AlCl3 can also be used in the same way
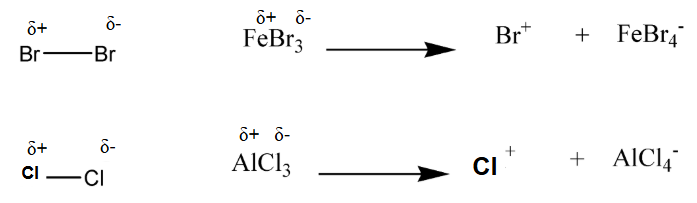
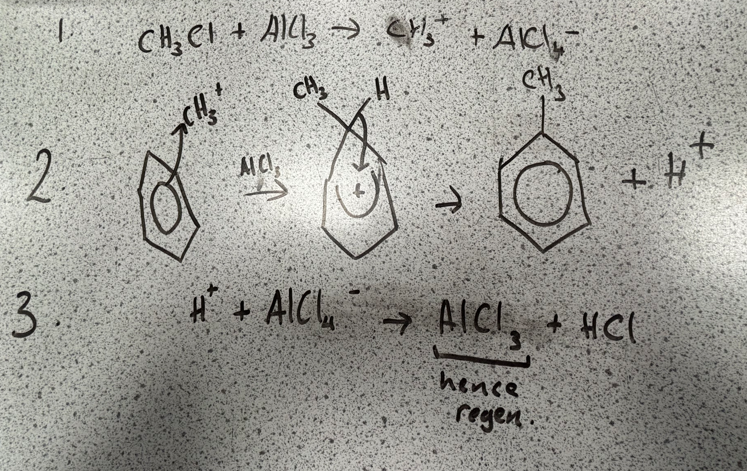
Alkylation
reagents and conditions
alkyl chloride, RCl
halogen carrier catalyst eg. AlCl3
anhydrous conditions
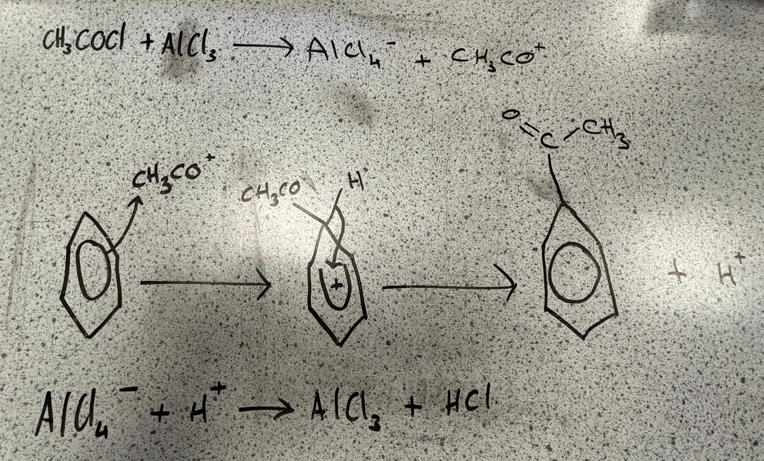
Acylation
reagents and conditions
acyl chloride, RCOCl
halogen carrier catalyst eg. AlCl3
anhydrous conditions