L6: Aluminium Alloys
Learning Objectives:
Apply the theory of precipitation and growth to the real-life example, Al-Cu.
How would we design a heat treatment to induce fine, well dispersed precipitates
Solution heat treat and age at low temperature
Discuss the transition from metastable precipitates towards equilibrium

Discuss the influence of precipitate size evolution on material properties
– hardness is higher with more Cu (lever rule)
– hardness first increases and then decreases,(cutting→bowing)
– peak ageing at low temperatures takes longer
– peak higher at lower temperature (↓solute solubility)
Be aware of precipitation hardening in a wide range of alloys
Be prepared to apply the principles learned to other alloying systems
(1) How to Design a Heat Treatment to Induce Fine, Well-Dispersed Precipitates:
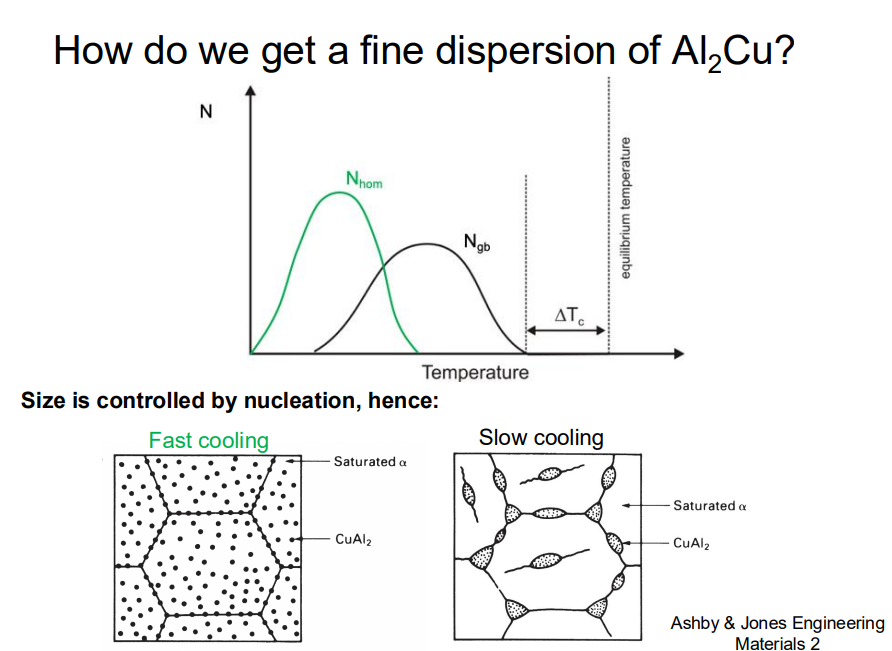
To achieve fine and well-dispersed precipitates in Al-Cu alloys, the following heat treatment steps are essential:
Solution Treatment: The alloy is heated to a temperature within the α-phase region to dissolve all copper into the α-phase, ensuring a homogeneous distribution of copper.
Quenching: The alloy is rapidly cooled (water quenched) to freeze the homogeneous copper distribution in the matrix. Rapid cooling prevents the formation of large precipitates, which would weaken the material.
Ageing: The alloy is aged at low to moderate temperatures to allow the formation of fine precipitates. The ageing temperature and time are critical to control the size and spacing of the precipitates. Lower ageing temperatures favour the formation of smaller, more numerous precipitates, while higher temperatures lead to coarser precipitates.
The key is to balance the cooling rate and ageing conditions to maximize the number of fine precipitates, which enhance strength by hindering dislocation motion.
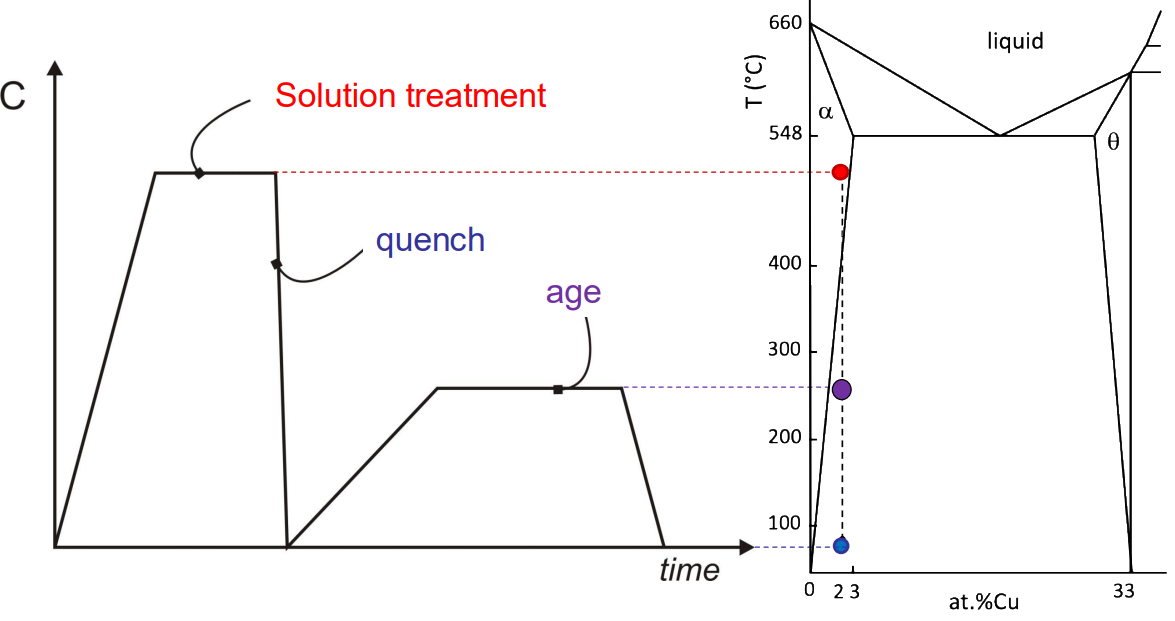
(2) Transition from Metastable Precipitates Towards Equilibrium:
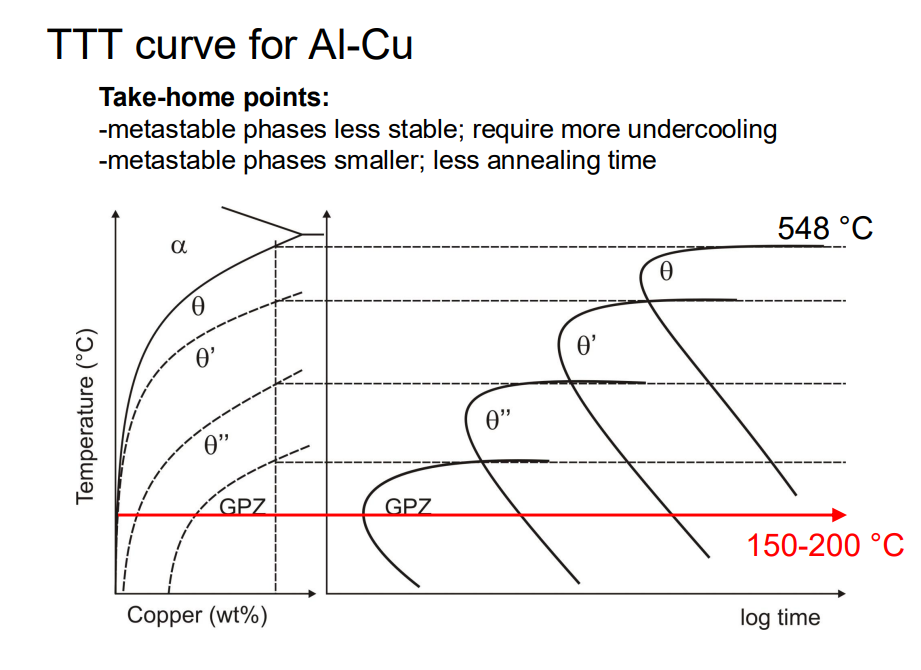
The precipitation sequence in Al-Cu alloys involves several metastable phases before reaching the equilibrium θ phase (Al₂Cu). The sequence is as follows:
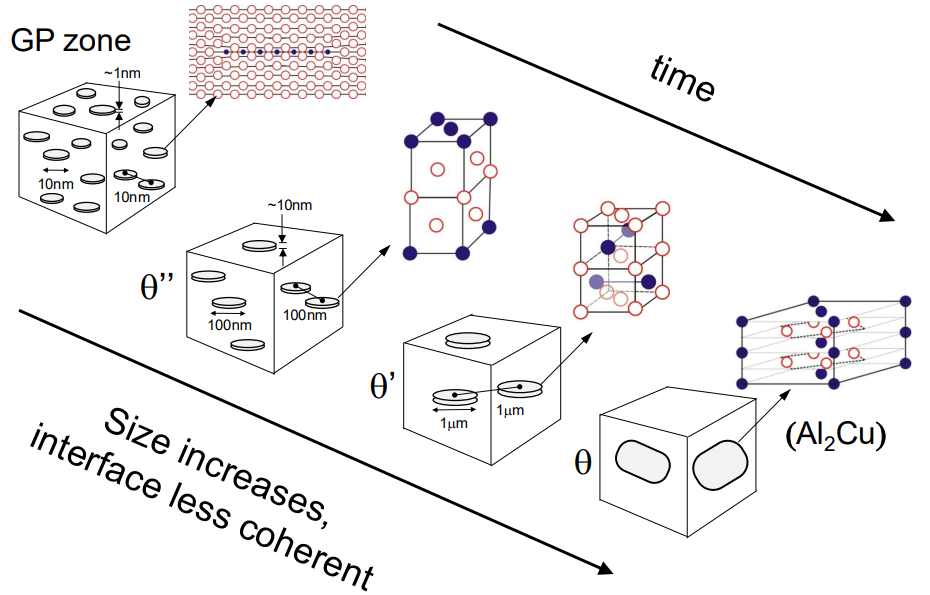
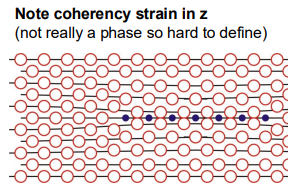
GP Zones (Guinier-Preston Zones): These are fully coherent, small regions of high copper concentration in the aluminium matrix. They form first due to their low nucleation energy barrier.
θ'' Phase: This phase is also fully coherent but has a slightly higher nucleation barrier than GP zones.
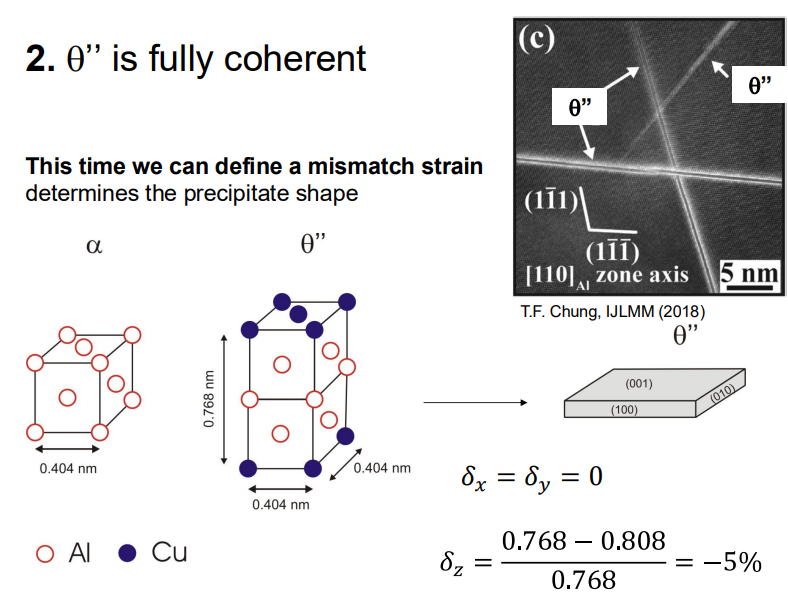
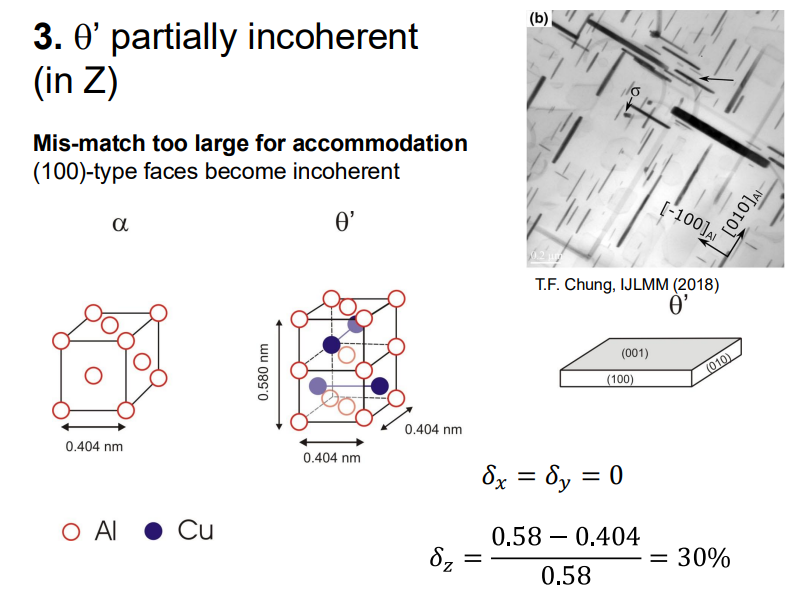
θ' Phase: This phase is partially coherent, with some interfaces becoming incoherent due to increased lattice mismatch.
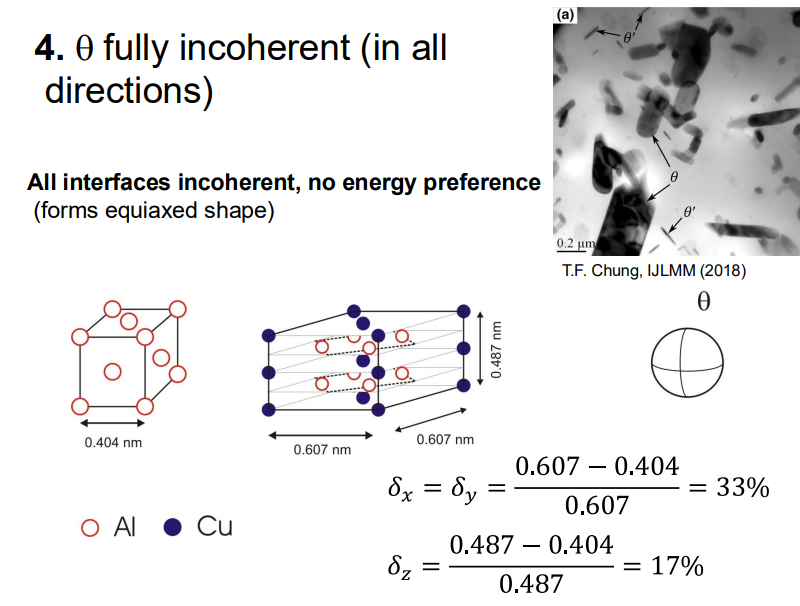
θ Phase (Equilibrium Phase): This phase is fully incoherent with the matrix and forms last. It has the highest nucleation energy barrier but is the most thermodynamically stable.
The transition from metastable to equilibrium phases is driven by kinetics, with the system evolving towards lower energy states over time. The presence of metastable phases is a result of their lower nucleation barriers, allowing them to form more readily than the equilibrium phase.
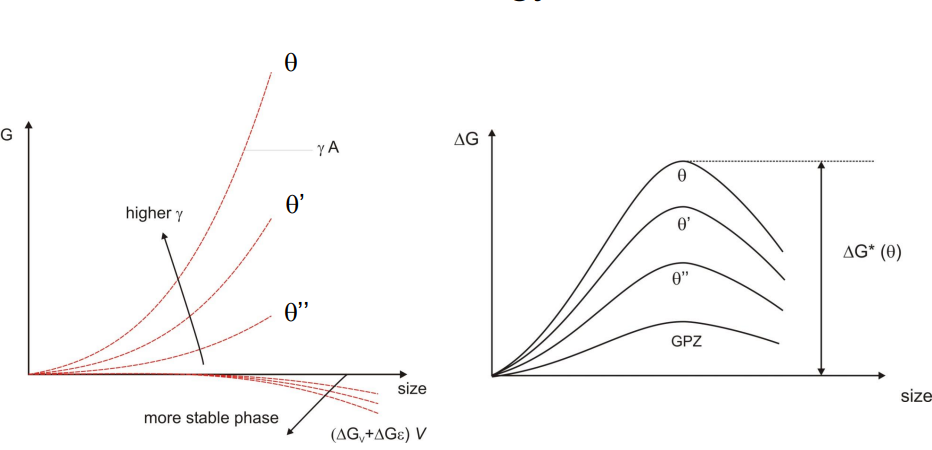
(3) Influence of Precipitate Size Evolution on Material Properties:
The evolution of precipitate size and spacing significantly affects the mechanical properties of the alloy, particularly its hardness and strength:
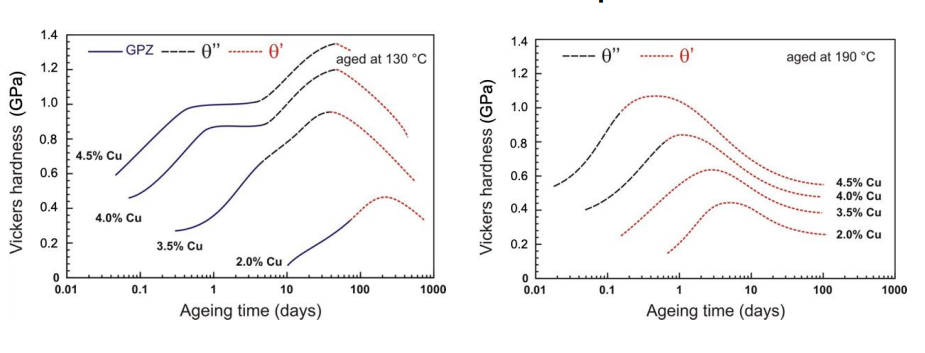
Initial Ageing (GP Zones and θ''): At the start of ageing, the formation of GP zones and θ'' precipitates increases hardness due to coherency strain and cutting stress. Dislocations must cut through these small, coherent precipitates, which requires extra stress.
Peak Ageing (θ' and θ''): The maximum hardness is achieved when a mixture of θ'' and θ' precipitates is present. The spacing between precipitates is optimal, and dislocations must bow around them, increasing the stress required for deformation.
Over-Ageing (θ Phase): As ageing continues, the precipitates coarsen, and their spacing increases. Dislocations can now bow more easily between the larger precipitates, reducing the stress required for deformation. This leads to a decrease in hardness and strength.
The cutting stress mechanism dominates when precipitates are small and closely spaced, while bowing stress becomes more significant as precipitates grow and their spacing increases. The larger the spacing, the easier it is for dislocations to bow around the precipitates, reducing strength.
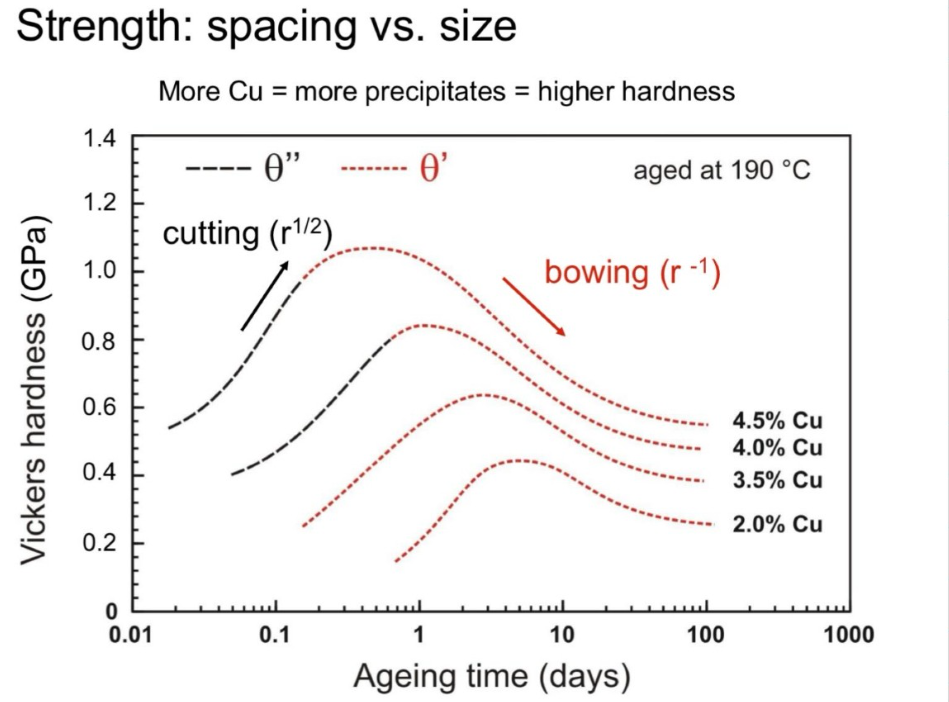
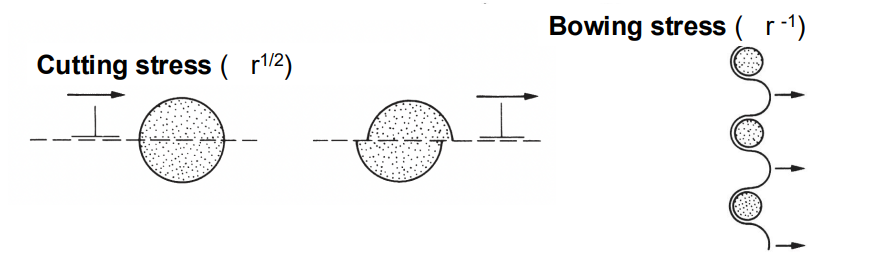
(4) Precipitation Hardening in a Wide Range of Alloys:
Precipitation hardening is not limited to Al-Cu alloys; it is a common strengthening mechanism in many alloy systems. Some examples include:
Al-Zn-Mg Alloys (7xxx series): Used in aerospace applications for their high strength-to-weight ratio.
Al-Mg-Si Alloys (6xxx series): Commonly used in automotive bodies due to their good formability and strength.
Cu-Be Alloys: Used in applications requiring high strength and electrical conductivity.
Ni-Cr-Ti-Al Alloys: Used in high-temperature applications, such as turbine blades, due to their ability to form γ' precipitates.
In all these systems, the principles of precipitation hardening—controlling nucleation, growth, and coarsening of precipitates—are applied to optimize material properties. The key is to design heat treatments that maximize the number of fine, well-dispersed precipitates to enhance strength while avoiding over-ageing, which leads to coarser precipitates and reduced strength.
Key Takeaways:
Heat Treatment Design: Solution treatment, quenching, and controlled ageing are essential for fine precipitate formation.
Metastable to Equilibrium Transition: The system evolves through metastable phases (GP zones, θ'', θ') before reaching the equilibrium θ phase.
Property Evolution: Hardness and strength are influenced by precipitate size and spacing, with cutting stress dominating at small sizes and bowing stress at larger sizes.
Wide Applicability: Precipitation hardening is a versatile strengthening mechanism used in various alloy systems, including Al-Cu, Al-Zn-Mg, and Ni-based superalloys.