Physical Science Final Study Guide
Final Exam Study Guide (Spring Semester)
**Remember, this is meant to GUIDE your STUDY. Therefore, while this gives you direction in what to study for the final, it is not an exhaustive list. Follow the instructions below to make the best use of this study guide.
Instructions: For each item listed below, you need to look over your notes, textbook, assignments, previous study guides, and previous tests to fill in the following things:
- Follow the instructions given (Draw, compare, explain, etc.)
- Describe what each item is (definition or description)
- Give examples that apply
- Explain how each item connects to other items
- Explain how the item fits into the unit overall
Chemical Reactions:
Unit 5: Chemical Reactions Notes, Chemical Reactions Study Guide-Unit 5 - Science
Looking at a chemical equation, be able to identify:
- Reactants-starting compound
- Products-ending compound
Be able to Balance Chemical Equations
Understand and explain the Law Conservation of mass and how it connects to balancing equations
- states that matter cannot be created or destroyed
Types of Chemical Equations and be able to identify chemical equations:
- Synthesis-compounds add together
- Decomposition-compound breaks down
- Single Replacement-single atom switches with an atom in the pair
- Double Replacement-atoms from each pair trade places; everyone has a partner
5 ways to change reaction rates (TedEd Video on Google Classroom)
- increase surface space(break up clumps)
- increase temperature
- decrease volume (smaller container)
- add a catalyst
- increase concentration (more particles, more collisions)
Identify characteristics and examples of Acids and Bases
- Acids:
- Substances with H+ ions (the more H+, the more acidic the substance)
- Properties of acids:
- Sour taste
- Conducts electricity
- Corrosive
- Can react strongly with metals
- Examples:
- Acetic Acid: Vinegar
- Citric Acid: lemons, limes, & oranges
- Bases:
- Substances with OH- ions (the more OH-, the more basic the substance)
- Properties of bases:
- Bitter taste
- Slippery feel
- Conducts electricity
- Corrosive
- Will not react with metals
- Examples:
- Cleaning products
- Chalk
Know how the pH scale is organized and ways to determine pH:
Values for Acids
- Less than 7
- Values for Bases
- More than 7 up to 14
- Value for Neutral
- 7
- Red and Blue litmus paper
- Acid:
- Blue turns red
- Base
- Red turns blue
- Neutral
- Neither changes
- Indicator (like red/purple cabbage juice)
pH paper
- What color it changes to indicates how acidic or basic the solution is
Identify differences between Exothermic vs endothermic
- Endothermic:
- A chemical reaction that requires excess energy (heat) to be absorbed before a reaction can occur
- Exothermic:
- A chemical reaction that releases excess energy after new chemical bonds are formed
Limiting and Excess Reactants
- Limiting reactant: starting compound that limits the amount of product that can be made
- Excess reactant: the reactant that could keep reacting if the other had not been consumed
Nuclear Chemistry:
Nuclear Processes (Unit 6) Updated 19-20.pptx, Nuclear Chemistry Unit Study Guide - Science
Universal Forces
- Gravitational
- The force of attraction of objects due to their masses
- The amount of gravity between objects depends on their masses and the distance between them
- Gravity is the weakest of the forces in the atom
- Electromagnetic
- The force that results from the repulsion of like forces and the attraction of opposites
- The force that holds electrons around the nucleus
- Strong
- The force that holds the atomic nucleus(like charges) together
- The force that counteracts the electromagnetic force
- Weak
- This force plays a key role in the possible change of subatomic particles
- For example, a neutron can change into a proton and an electron
Radioactive Decay:
- Causes: Unstable nuclei expel energy and subatomic particles to get to a more stable state
- Radioactivity: Process in which an unstable atomic nucleus emits charged particles and/or energy to become stable. Can become a different element
- Radioisotope: Any atom containing an unstable isotope (unstable amount of neutrons)
- Alpha Decay: A way to create a stable nucleus is to emit 2 protons and 2 neutrons from the nucleus
- Alpha Particle:
- The particle emitted is called an alpha particle.
- It is positively charged.
- It is the same as a helium particle
- Beta Decay: In order to create a stable number of neutrons in the nucleus, a neutron can split into a proton (+) and an electron (-)
- Beta Particle:
- The electron created is called a beta particle
- The beta particle is emitted from an unstable nucleus
- In beta decay, the product isotope has one more proton and one less neutron than the reactant isotope
- Gamma Decay: The emission of electromagnetic radiation of an extremely high frequency i.e. very high energy, giving out excess energy in order to stabilize the unstable nucleus
- Gamma Rays/Radiation
- Gamma Ray: a penetrating ray of energy emitted by an unstable nucleus
- Gamma rays/radiation have no mass or charge
- During gamma decay, the atomic number and mass don’t change, but the energy of the nucleus decreases
- Half Life:
- HalfLife – time required for one-half of a radioactive sample to decay.
- HalfLife or Rate of Nuclear Decay varies from element to element
- If you have a larger sample of radioactive material, it will decay at a constant rate, however, more radiation (Alpha, Beta, or Gamma) is given off
- Uses for Nuclear Decay and Half-Life:
- Radioactive Dating
- The age of an object is determined by comparing the object’s carbon14 levels with the carbon 14 levels in the atmosphere
- Energy source
E=mc2
- E=energy released (J)
- M=mass difference (kg)
- C=speed of light in a vacuum (3 x 108 m/s)
- A small amount of mass can release a large amount of energy
Nuclear Fission:
- A large nucleus is split into two smaller ones
- During nuclear fission, neutrons are released
- There are two types of nuclear fission that can occur: Spontaneous and Induced
- Causes: adding a neutron to a large particle, forcing it to excite and split into two smaller atoms
- Chain Reactions: a process in which neutrons released in fission produce an additional fission reaction in at least one further nucleus. This nucleus in turn produces neutrons, and the process repeats.
- Uses for Fission:
- Uncontrolled chain reactions can be used as nuclear weapons
- Controlled chain reactions can be used as nuclear power
Nuclear Fusion:
- Two small nuclei collide to form a larger nucleus
- The reactants’ total mass is less than the product’s mass, so the remaining mass is converted into energy.
- Causes: Extreme pressure and temperature
- Uses for Fusion:
- Generate electricity with fusion reactors
Law of Conservation of Mass, Law of Conservation of Energy, and how it connects to Nuclear Decay, Fission, and Fusion:
- Law of Conservation of Mass
- Mass is neither created nor destroyed in chemical reactions
- Law of Conservation of Energy
- Energy can neither be created nor destroyed; rather, it can only be transformed or transferred from one form to another.
- Nuclear Decay:
- An equal amount of particles/mass/energy before and after decay
- Fission:
- An equal amount of particles/mass/energy before and after the reaction
- Fusion:
- An equal amount of particles/mass/energy before and after the reaction
Physics: Motion and Forces:
Unit 7: Motion and Forces Study Guide - ScienceForces (Spring 18-19) ppt.ppt
Motion:
- Distance vs Displacement:
- Distance: the measure of “how much ground an object has covered during its motion”
- Displacement: refers to the measure of “how far out of place is an object.”
- Speed vs. Velocity:
- Speed: the time rate at which an object is moving along a path
- Velocity: the rate and direction of an object's movement
- Speed Calculations: S = D / T
- Acceleration Calculations: A = (Vf - Vi) / T
- Graphs:
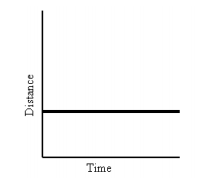
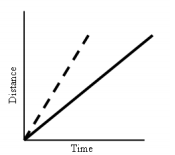
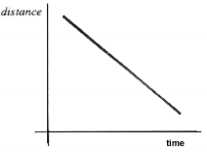
- Position vs. Time
- Slope means: object is moving at a constant rate in a positive or negative direction
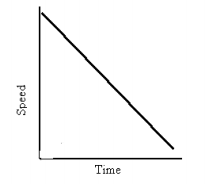
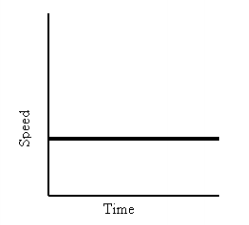
- Velocity vs. Time
- Slope means: object is slowing down or speeding up
- Interpreting Graphs:
- Drawing Graphs:
- Position vs. Time Graph and Velocity vs. Time Graphs that say the same thing:
Forces: a push or pull acting on an object
- Newton’s Laws of Motion:
- Law One: objects at rest remain at rest, and objects in motion remain in motion with the same velocity, unless acted upon by an unbalanced force
- Inertia: an object will continue its current motion until some force causes its speed or direction to change
- Examples:
- When the driver brakes or turns, people in the car continue moving forward or sideways until their seatbelt stops them
- Computer on desk
- Law Two: Force causes acceleration while mass resists acceleration
- F = m x a: force equals mass times acceleration
- Fw = m x g: the force of weight equals mass times the acceleration of gravity
- Calculations:
- Examples:
- heavier objects require more force to get moving than lighter objects.
- Knowing the mass of the car and its acceleration, we can determine the force of impact
- A baseball will go further than a bowling ball when hit with the same force with a bat because the bowling ball has more mass
- Law Three: states that every time one object exerts a force on another object, the second object exerts a force that is equal in size and opposite in direction back on the first object
- Examples:
- when a car strikes the wall, the wall gives an equal and opposite force on the car
- swimming
- Net Force Examples/Calculations
- the sum of all the forces acting on an object
- Equilibrium:
- State of equality
- No net force
- No acceleration
- Constant speed
- An object at rest (not always true)
Momentum:
Calculating Momentum: p = m x v
- Law of Conservation of Momentum:
- The conservation of momentum states that, within some problem domain, the amount of momentum remains constant; momentum is neither created nor destroyed, but only changed through the action of forces as described by Newton's laws of motion.
- Transfer of Momentum:
- Elastic Collisions (Bounce off each other)
- Momentum is conserved
- They bounce off of each other-go opposite directions
- Inelastic Collisions (Stick together)
- Momentum is conserved
- They stop and stick together
- Examples
- Inelastic:
- Head-on car crash
- Dropped ball does not return to its original height
- Elastic:
- Billiard table balls
- bowling ball
- Comparing Momentum of different objects
\n
Unit 8: Energy
Energy and Mechanical Waves (Spring 18-19)Copy of Work Energy and Power.ppt Electricity ppt.pptxhttps://quizlet.com/699149749/unit-8-energy-review-flash-cards/
Energy Unit:
Work:
- When a force acts on an object in the direction the object moves
- For a force to qualify it must
1. Displacement MUST happen
2. Force MUST cause the displacement
- Examples:
- a horse pulling a plow through the fields
- a father pushing a grocery cart down the aisle of a grocery store
- a freshman lifting a backpack full of books upon her shoulder
- a weightlifter lifting a barbell above her head
- Work = Force x Distance
- Measured in Joules
- 1 Joule = 1 Newton*1 meter
Power
- Power is the rate of doing work
- Doing work at a faster rate requires more power.
- Power = Work / Time
- 1 watt = 1 joule / second
- 1 horsepower = 746 watts
Simple Machines
Energy
- Ability to do work
- When work is done to an object, energy is transferred to it.
- Work is the transfer of energy
- Measured in Joules (same as work)
- Type of Energy
- Potential Energy:
- Energy of position
- relates to the relative position of objects in the system.
- rubber band- position (distance) between the two ends
- Stored energy occurs if something is stretched or compressed (elastic)
- clock spring
- bungee cord
- Kinetic Energy:
- Energy that an object has when it is in motion = kinetic energy
- KE = ½ mv2
- Person walking
- A thrown baseball
- Motion and Energy Types
- Subcategories of Energy:
- Elastic
- PE stored in stretching or compressing shape of object and ability to regain original form (springs)
- Nuclear
- PE stored in the nucleus of an atom. (Nuclear Fission and Fusion)
- Chemical
- PE stored in bonds (gasoline)
- Electrical
- KE, the movement of electrons. (Lightning)
- Thermal
- KE (Heat)
- Light
- KE that travels through space through waves that is visible to the human eye
- Law of Conservation of Energy
- The Law of Conservation of Energy states that energy cannot be created or destroyed.
- Energy can be transferred from object to object
- Energy can be transferred into different forms
Elasticity
- Elastic Potential Energy
- potential energy stored in stretching or compressing an object and it being able to regain its original form
- Elastic Deformation
- An object's ability to be stretched or compressed, then regain its original position or shape as force is applied
- Plastic Deformation
- As a force is applied to an object, that object is unable to regain its shape; it remains stretched or compressed
Waves
- Longitudinal
- A wave in which the vibration of the medium is parallel to the direction the wave travels
- Parts of wave
- Compression: The part of a longitudinal wave where the particles of the medium are close together
- Rarefaction: The part of a longitudinal wave where the particles of the medium are far apart
- Wavelength: The distance between two corresponding parts of a wave (like high point to high point)
- Sound Waves
- a longitudinal wave consisting of compressions and rarefactions, which travels through a medium and is detectable by the human ear.
- Transverse
- A wave that moves the medium in a direction perpendicular to the direction in which the wave travels
- Parts of wave
- Crest: The highest point of a wave
- Trough: The lowest point of a wave
- Equilibrium: Halfway between the highest and lowest points of the wave
- Wavelength: The distance between two corresponding parts of a wave (like high point to high point)
- Amplitude: The height of a wave
- Electromagnetic Spectrum
- Different wavelengths of energy traveling in a transverse wave that include Radiowaves, Microwaves, Visible Light, Xrays, and Gamma Rays
Light and Color
- Primary Colors:
- Red
- Blue
- Green
- Secondary Colors:
- Magenta
- Yellow
- Cyan
- All colors mixed:
- White Light
Electricity
- Series circuits
- A series circuit connects the components one after the other
- A break in any part of a series circuit stops the flow of current in the whole circuit
- Parallel circuits
- A parallel circuit divides into two or more branches.
- If a component breaks or is removed, the other components remain on