Chapter 2: Experimental Techniques
Elements, Compounds, and Mixtures
Element: Simplest substance, cannot be broken down, made of identical atoms/molecules.
Atom: Basic unit of matter, defines chemical properties of an element.
Molecule: Two or more atoms chemically bonded.
Ion: Electrically charged atom/group of atoms (anion: negative, cation: positive).
Compound: Two or more elements chemically combined in specific proportions.
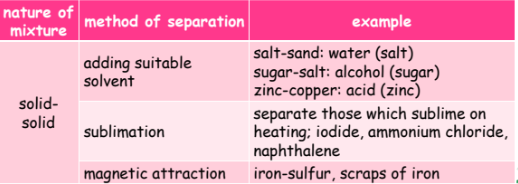
Mixture: Substance composed of multiple components, easily separated physically.
Heterogeneous: unevenly mixed, easily separated (filtration, magnet).
Homogeneous: evenly mixed, requires processes like distillation or chromatography for separation.
Measurement
Units: Uses International System of Units (SI units).
Time:
Tool: Digital stopwatch/timer.
Unit: Seconds (s).
Temperature:
Tool: Thermometer.
Unit: Kelvin (K), Celsius (°C).
Mass:
Tool: Digital balance.
Unit: Kilogram (kg), gram (g).
Volume (Liquid):
Tool: Measuring cylinder, burette, volumetric pipette.
Unit: cm .
Volume (Gas):
Tool: Gas syringe.
Unit: m , cm .
Scientific Methods
Process: Problem → Hypothesis → Experiment → Data Collection → Data Analysis → Conclusion.
Advantages: High control level, cause-effect determination.
Disadvantages: Subjectivity, can be costly and time-consuming.
Solutions and Solubility
Solute: Substance that dissolves.
Solvent: Substance that dissolves the solute.
Solution: Mixture of solute and solvent.
Aqueous Solution: Water as the solvent.
Saturated Solution: Maximum solute dissolved at a specific temperature.
Solubility increases with temperature.
Separation and Purification Techniques
Filtration: Separates solid-liquid mixtures.
Crystallization: Purifies solids by forming crystals.
Simple Distillation: Separates liquids with different boiling points.
Fractional Distillation: Separates miscible liquids with close boiling points.
Chromatography: Separates solutes in a solution.
Locating agents are used for colorless solutes.
RfRf = {distance travelled by substance}/ {distance travelled by solvent}


Purity
Essential for food and drugs to prevent side effects.
Paper chromatography can identify purity.
Pure substances have sharp melting/boiling points, while impure substances have a broader range due to the disruption of the crystal lattice..