Fractional Distillation of Crude Oil
Crude Oil
Mixture of saturated hydrocarbons with different chain lengths extracted from underground
Varying lengths of the alkane result in a broad range of boiling points. This allows for the separation of crude oil by fractional distillation
Fractional Distillation
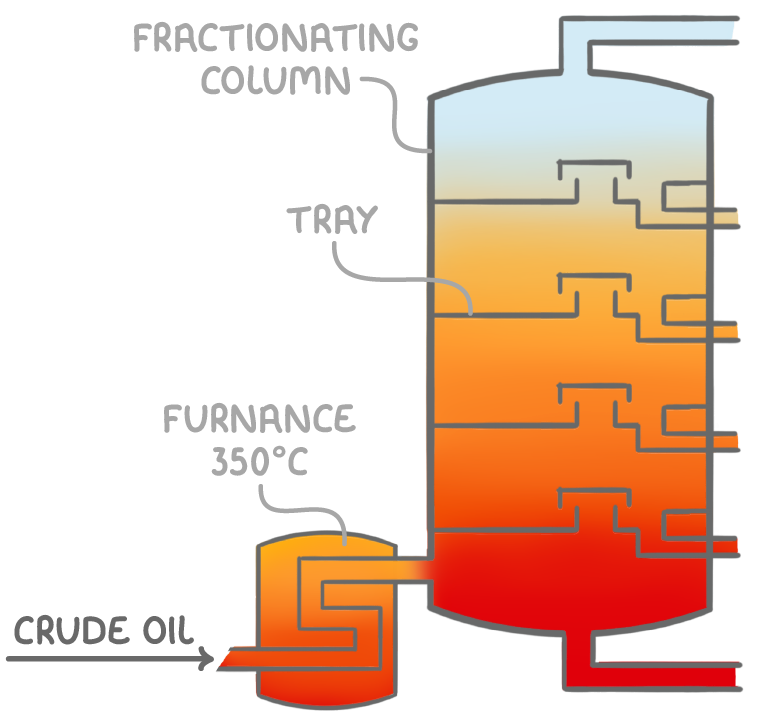
Crude oil is heated to 350°C in a furnace to vaporise the hydrocarbon mixture
These vapours enter a fractionating column that has a higher temperature at bottom (350°C) and lower temperature at the top (40°C)
As the hot vapours rise, they cool. When the vapour temperature drops below the boiling point of the hydrocarbon in the mixture, it condenses from gas to liquid on the tray surface
The condensed hydrocarbon liquid that accumulates on each tray are drawn off at specific intervals as fractions (mixture of hydrocarbons with similar boiling points)
Fractions
Boiling Point
Boiling point increases progressively. down the column as the alkane chain length increases in each successive fraction. Lightest gases boil at below 0°C while undistillable residue requires temperature of about 500°C
Viscosity
Steadily increases as you move down the column. Very light distillates like gasoline or jet fuel, flow freely. Heavier fratctions at the bottom have high viscosities
Flammability
Decreases down the column, light gases and short-chain hydrocarbons at the top ignite easily making them excellent fuels. Heavy, long chain fractions at the bottom are less flammable and do not make as good fuels