Aromatic chemistry recap
Aromatic = benzene ring
Development of structure from Kekulé model
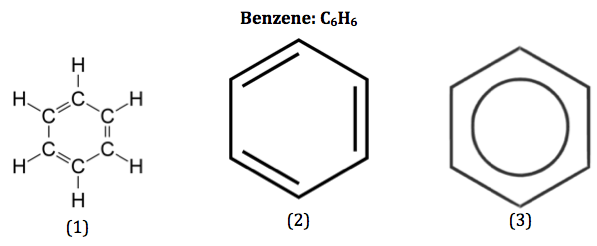
Current model = the delocalised model with a ring of electron density above and below:
Bond | Length / nm |
C - C | 0.154 |
C - C (Benzene) | 0.140 |
C = C | 0.134 |
C ≡ C | 0.120 |
Overall, each carbon-carbon bond is intermediate between a single and a double bond. The delocalised system is very important in the chemistry of benzene and its derivatives. It makes benzene unusually stable. This is sometimes called aromatic stability.
Uses electrophilic substitution reactions, not electrophilic addition
nitration
acylation
Nitration of benzene:
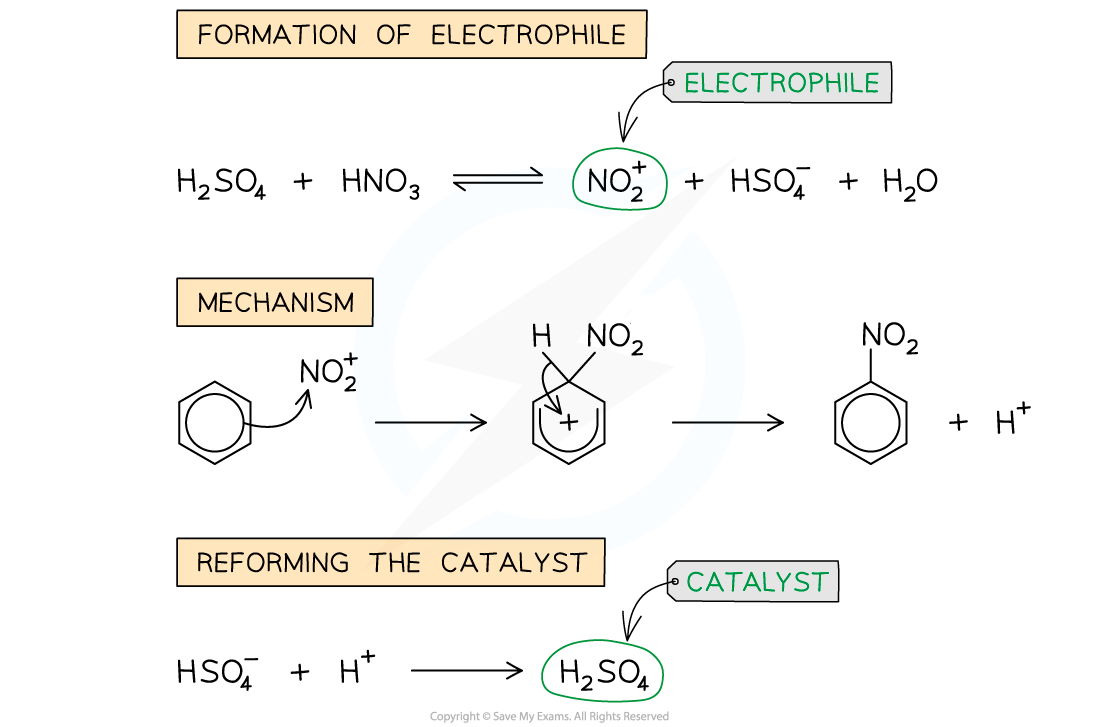
Curly arrow = movement of a pair of electrons, direction of arrow shows direction that they move in.
Second part of mechanism step is a temporary loss of delocalisation where the NO2 is bonded.
The delocalised system of the aromatic ring has a high electron density that attracts electrophiles. At the same time the electrons are attracted towards the electrophile.
A bond forms between one of the carbon atoms and the electrophile. But, to do this, the carbon must use electrons from the delocalised system. This destroys the aromatic system, the carbon loses an H+ ion with the electron in the C - H bond returning to the delocalised system. The sum of these reactions is the substitution of H+ by the electron.
Friedel-Crafts acylation:
The acyl chloride can be anything e.g. ethanoyl chloride
These reactions use aluminium chloride as a catalyst
RCO substituting for a hydrogen on the aromatic ring
Reaction takes place because the aluminium atom in aluminium chloride has only six electrons in its outer main level and readily accepts a lone pair from the chlorine atom of RCOCl.
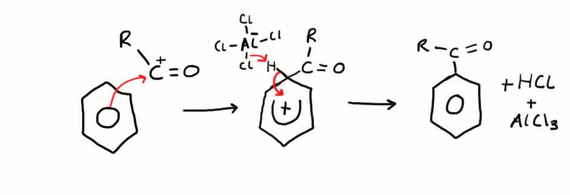

Regeneration of AlCl3:
H+ + AlCl4- —> AlCl3 + HCl