Introduction To Biological Science / Chemistry composition of living organism
Principles of Science
Causality – Every event has a cause that leads to an effect.
Uniformity – Natural laws are consistent and apply universally.
Scientific Reasoning
Inductive Reasoning: Drawing general conclusions from specific examples.
Deductive Reasoning: Applying observation and experimentation to reach specific conclusions
The Scientific Method is a systematic process used to investigate phenomena, acquire new knowledge, or correct and integrate previous knowledge. It typically involves:
Observation: Identifying a problem or question.
Hypothesis: Forming a testable explanation.
Experimentation: Conducting experiments to test the hypothesis.
Data: Interpreting the data.
Conclusion: Drawing conclusions based on results repeating the experiment to verify findings
Scientific Attitude
1. Problems have solutions
2. Respect for the power of theoretical structure
3. Thirst for knowledge
4. Ability to separate fundamental concepts from irrelevant
5. Ability to suspend judgment
6. Appreciation of probability and statistics
7. Automatic preference for scientific explanation
8. Understanding the tolerance limits
9. Awareness of assumptions
10. Determinism
11. Empathy for human conditions
12. Empiricism
13. Loyalty to reality
14. Parsimony
15. Precision
16.Respect for quantification and appreciation of
mathematics
17. Respect for scientific paradigms
18. Scientific manipulation
19. Skepticism
20. Willingness to change position
Limitations of Science -Science cannot:
1. Answer questions about value
2. Answer questions of morality
3. Deal with unique
4. Answer the questions about Supernatural
As Life biology -The existence of an individual human being or animal
Characteristics of a Living Things - Cellular Organization
• Metabolism
• Movement or Motility
• Irritability or Responsiveness
• Reproduction
• Growth and development
• Living things evolve
• Adaptiveness
Theories of Life
Divine Creation Theory: This idea says that a god or supernatural being created life. ( all based on bible)
Abiogenesis Theory: This theory suggests that life started from non-living things like simple chemicals, ( you are dust u shall be return )
Biogenesis Theory: life comes from other living things. In other words, living organisms reproduce to create new life.
Theory of Evolution: This theory, developed by Charles Darwin, says that living things change over time to adapt to their surroundings. Through many generations, these changes led to the variety of life forms we see today.
a. Chevalier de Lamarck’s
b. Charles Darwin’s Natural Selection Theory
Theory of Evolution: Lamarck’s concept explains that the descent with modifications and changes in the environment caused changes in the organism. ( by means of adaptation )
1. Theory of Needs
2. Theory of Use and Disuse
Theory of Evolution: Darwin’s Natural Selection Theory - survival of the fittest overproduction, struggles for existence, survival of the fittest. variation of the characters and transmissions of variation of characteristic
Theories of Mutation - cause of environmental mutagens, radiation, alcoholism, drugs, pills, genetic modification, or GMOs they inhibit mitotic stages
Chemistry composition of a living organism
Why chemistry matters - everything we do is affected by chemical reaction (we as individual are made of molecules )
Differents between
Elements - pure substances consist of only oneidentical atom
Compound - is substance formed by two or more different types of elements
Atom - Smallest unit of matter every element is made up of one kind of atom
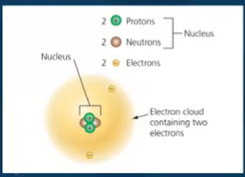
protons - positive electric charge
electron - negative electric charge
neutron electrically neutral ( no charge )
Structure of atoms
chemical bonding and molecules
two types of chemical bonds:
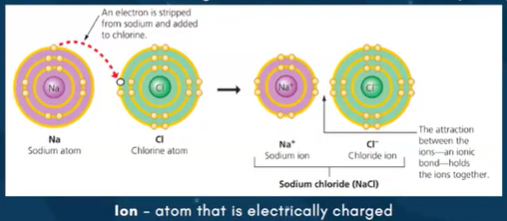
Ionic bond - transferring of electrons
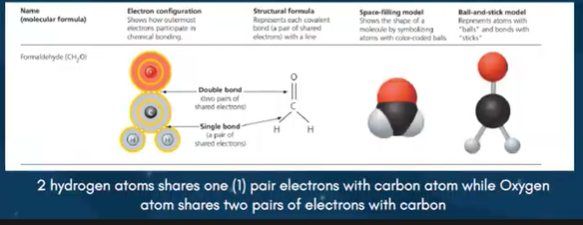
Covalent - bond Sharing between 2 or more atoms ( strongest bond )
IONIC BOND - transferring electrons EZ. table salt (NaCL)
COVALENT BOND - shared electrons ( ex. formaldehyde )
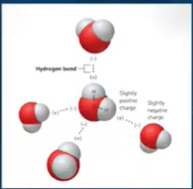
HYDROGEN BOND - a special Type of Dipole-dipole bond hydrogen atom bonded to strongly electronegative atom ( commonly for water )
organic - usually found living organisms most like made carbon-based molecule
inorganic - usually lack carbon and abiotic nature
large biological molecules -
carbohydrates
lipids
protein
nucleic acids
Carbohydrates -Often referred to as "carbs", a Class of molecules that includes sugars and polymers of sugars, Primary source of energy (animals); building material (plants)
Different classes of carbohydrates:
Monosaccharides - come from the Greek word mono and sacchar or sugar and its monomer’s building block ( cannot be broken down ) glucose, fructose and galactose
Disaccharides
Polysaccharides
glucose and fructose are called isomers
galactose - bran sugar and commonly found in dairy products
lactose - ( glucose and galactose ) milk sugar common in dairy products
maltose - ( glucose and glucose ) less sweet and commonly use in beverages
sucrose - ( glucose and fructose ) most common carbohydrates know as table sugar
POLYSACCHARIDES • Complex carbohydrates (long chains of sugars) polymers of monosaccharides
Example: • Starch • glycogen • Cellulose
Lipids - Hydrophobic (water-fearing) Do not mix with water Glycerol and fatty acids
Example: Phospholipids, Triglycerides, Waxes, Fats and Steroids
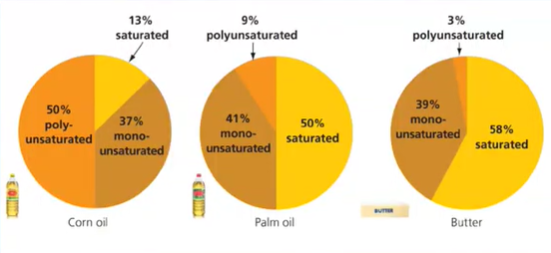
relative amounts of different fats
Steroids - Many famous athletes have admitted using chemically modified ("designer") performance- enhancing anabolic steroids
Proteins - Polymer of amino acids • Building block: Amino acid (AA) - 20 kinds of AA • Accounts for 50% of the dry weight of most cells "Worker bees"
Nucleic acid - Store information and provide the instructions for building proteins. Nucleotides - building blocks Two types of nucleic acids:
• DNA - Deoxyribose nucleic acids
• RNA - Ribonucleic acids DNA - double-stranded RNA - single-stranded