5. Nuclear physics
Structure of an Atom
Atoms are the building blocks of matter, composed of subatomic particles: protons, neutrons (which make up the nucleus), and electrons (which orbit the nucleus).
Protons have a charge of +1, electrons have a charge of -1, and neutrons are neutral but contain equal amounts of positive and negative charge.
The mass of a proton and neutron is considered as one atomic mass unit (u), while the electron's mass is approximately 1/2000 of a proton.
In a neutral atom, the number of protons equals the number of electrons.
Isotopes and Radioactive Decay
Isotopes are variants of an element that have the same number of protons but a different number of neutrons.
For example, helium-4 has two protons and two neutrons (mass number 4), while an unstable isotope of helium might have three neutrons.
Radioactive decay is the process wherein unstable nuclei emit radiation to become stable, involving alpha, beta, and gamma emissions.
Types of Radioactive Emissions
Alpha particles are made of two protons and two neutrons, carrying a mass number of 4 and a charge of +2.
Beta particles are electrons emitted from a neutron, which transforms into a proton and an electron during decay, represented by a mass number of 0 and a charge of -1.
Gamma rays are high-energy electromagnetic waves with no mass and no charge and travel at the speed of light.
Properties of Radiation
The strength of ionization varies among the three types of radiation: alpha particles are the most ionizing, beta particles are moderately ionizing, and gamma rays are the least ionizing.
Penetration abilities also differ: alpha particles can be stopped by paper, beta particles require aluminum, and gamma rays need thick lead to be stopped.
lonization can be harmful; for example, ionized living tissue can lead to cell damage or cancer.
Measuring Radiation and Background Radiation
Background radiation, typically low-level, comes from cosmic rays, natural sources in rocks, and elements like radon gas in the air.
Geiger-Müller (GM) tubes measure radiation by detecting ionization events in air; readings should account for background radiation for accuracy.
Background radiation levels usually measure around 20 counts per second, which is significantly lower than the rate from most radioactive sources.
Half-Life
The half-life of a radioactive substance is the time required for the quantity of unstable nuclei to reduce by half.
The decay process is random and spontaneous, varying in time but consistently following a pattern for different isotopes.
Knowing how to calculate remaining radiation levels based on half-life is essential, although memorizing specific half-lives is not required.
The Alpha Scattering Experiment
Ernest Rutherford's alpha scattering experiment involved directing alpha particles at a thin sheet of gold foil.
Observations included most particles passing through unobstructed, some being deflected, and a very small fraction bouncing back, leading to significant discoveries regarding atomic structure.
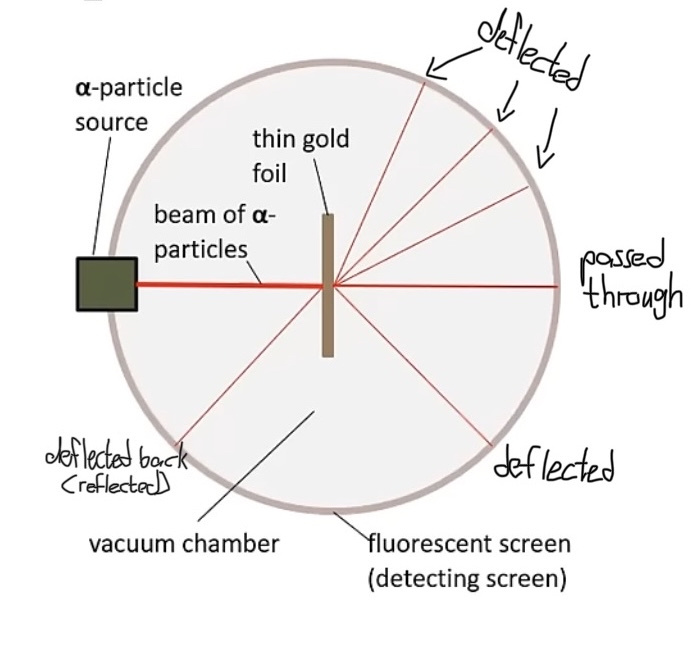
Atomic Structure and Observations
The atom is primarily composed of empty space. This indicates that there is significant separation between the nucleus and electrons in an atom.
The nucleus of an atom is dense and contains a high concentration of mass and positive charge.
This leads to the conclusion that alpha particles, which are also positively charged, will be repelled by a similarly charged nucleus.When alpha particles pass closely to the nucleus, they typically either pass through with only minor deflections or bounce back if they collide directly with it. This illustrates that the nucleus is relatively small compared to the overall size of the atom.
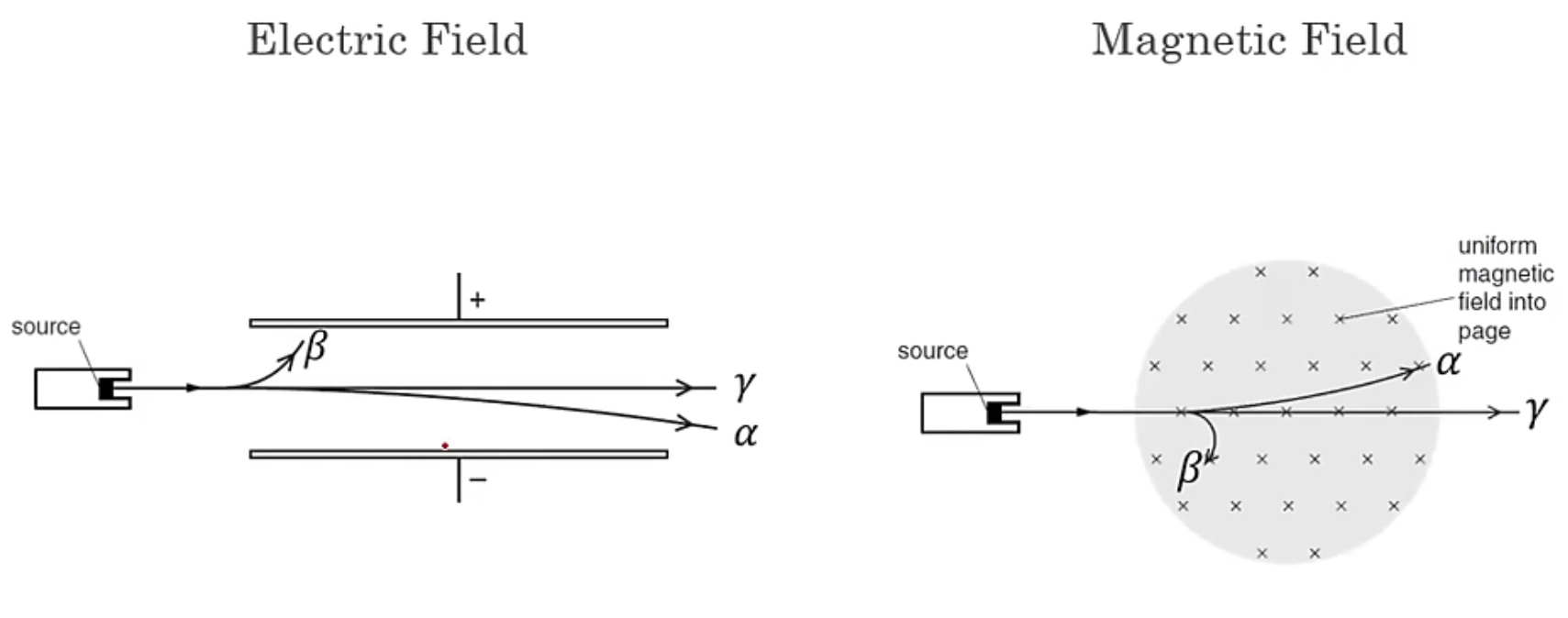
Concept of Deflection
Deflection occurs when charged particles, such as alpha and beta particles, interact with charged fields. For example, alpha particles are attracted to negatively charged areas, while beta particles are drawn to positive areas.
Fleming's left hand rule is applicable when determining the direction of forces acting on charged particles in magnetic fields. As charged particles move, they can be considered as a flow of current, affecting their movement.
The mass of an alpha particle is substantially greater than that of a beta particle, approximately 8,000 times heavier, which is why alpha particles exhibit less deflection compared to beta particles.
Applications of Radiation
Smoke Detectors: These devices employ radioactive isotopes to detect smoke. When smoke interrupts the flow of radiation from the isotope to a sensor, it triggers an alarm.
Sterilization of Medical Equipment: Medical instruments can be sterilized using devices that emit radiation rather than microwaves, effectively killing harmful pathogens without the need for disposability.
Irradiation of Food: Radiation can be used to ensure food safety by eliminating pathogens without adding radioactive substances directly to the food.
Cancer Treatment: Gamma radiation is utilized in radiotherapy to target and treat cancerous cells.
Industrial Measurement: In manufacturing, radiation sources can be used to measure the thickness of materials, such as aluminum sheets. By detecting variations in radiation passing through, the machinery can adjust thickness in real-time.
Safety Precautions for Radiation Exposure
Store radioactive materials in lead-lined boxes equipped with locks and secure them in locked cupboards to prevent unauthorized access.
Reduce exposure time when working with radioactive materials, as even brief contact can be harmful.
Increase distance from the radiation source when possible; staying further away provides greater safety from exposure.
Utilize shielding, primarily through lead materials, to effectively block most types of radiation and enhance safety during handling and storage.