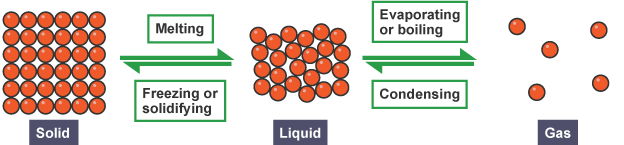
solids liquid and gas
Some substances can change directly from solid to gas without becoming a liquid in between. This is called sublimation. Solid carbon dioxide ('dry ice') and iodine can sublime
Key fact
Evaporation can take place at temperatures lower than the boiling point. It describes the change of state from liquid to gas. For example, when a puddle dries. Boiling is the process in which a liquid evaporates at its maximum rate, at or above the boiling point.
Physical and chemical change
Changes of state are physical changes. The substance remains the same.
It is only during a chemical change that a new substance is formed.
Limitations of the particle model - Higher
The particle model represents particles as inelastic spheres.
It makes a number of simplifications:
it assumes that particles are spheres
it represents spheres as the same size
the gap between atoms in a gas are small enough to make a diagram fit on the page, they should be much larger
differences in the forces of attraction between particles are not fully explained
This means that there is a limit to what the particle model can explain. For example, it cannot explain why substances have different melting points and boiling points.
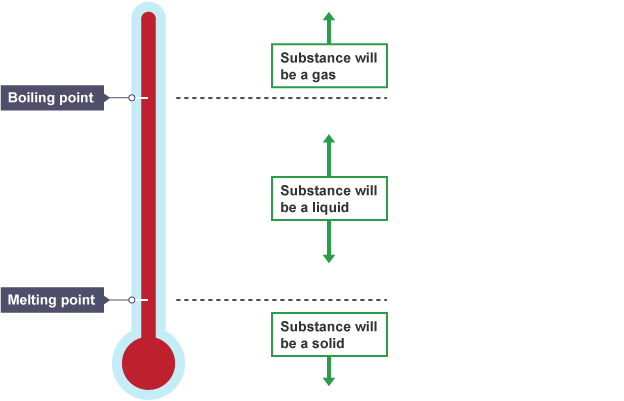
Explaining melting points and boiling points
Melting
Particles in a substance have forces of attraction between them. When a substance in the solid state is heated:
energy is transferred to the particles
the particles move faster and have more kinetic energy
some of the bonds are overcome and particles can slide round each other
Boiling
energy is transferred to the particles
particles eventually gain enough kinetic energy to overcome the forces of attraction
the particles can then move apart
The strength of the forces of attraction between particles varies between substances.
The stronger the forces of attraction between particles, the more energy is needed to overcome them, and so the higher the boiling point and the higher the melting point.
Condensing and freezing
Condensing and freezing are the opposite of boiling and melting. During these processes energy is transferred to the surroundings.