Reactions of Aldehydes & Ketones
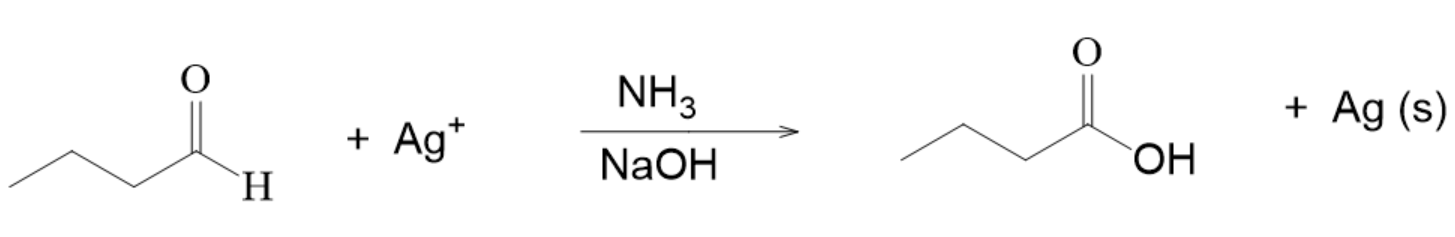
Oxidation of Aldehydes
- aldehydes can be oxidized to carboxylic acids by
- chromic acid
- is made of silver ion, ammonia, and hydroxide ion
- this reaction can also be called the or
Example with Chromic Acid
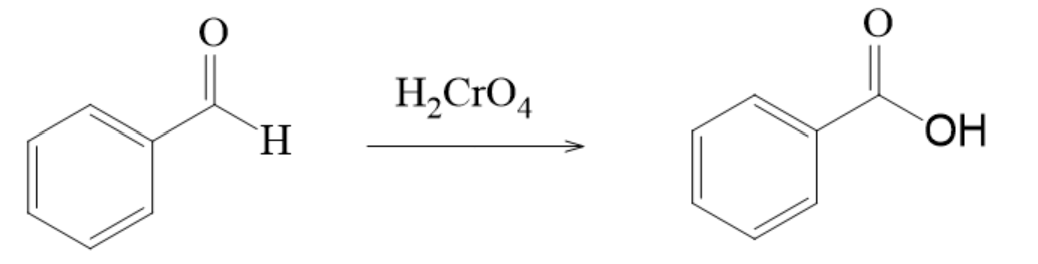
Example with Tollen’s Reagent
Formation of Hemiacetals & Acetals with Alcohols
Formation of Hemiacetals
- aldehydes/ketones and hemiacetals exist in equilibrium when dissolved in an alcohol
- the reaction to form a hemiacetal is an example of an addition reaction
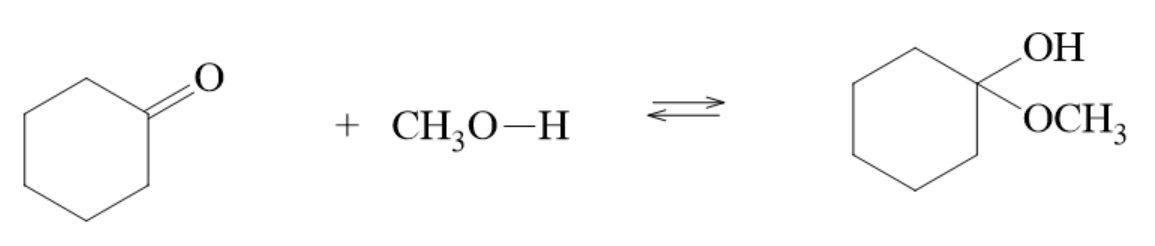
Formation of Acetals
- aldehydes/ketones can be formed into acetals in the presence of
- an acid catalyst
- excess of alcohol
- this reaction results in the loss of a water molecule
- the aldehyde/ketone actually reacts with 2 molecules of the alcohol
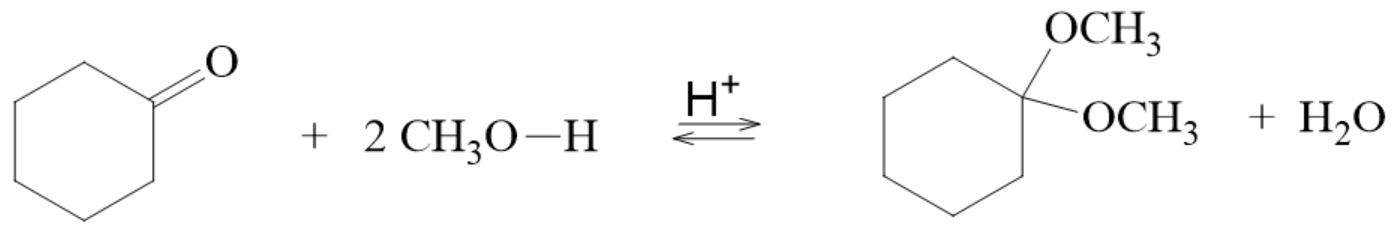
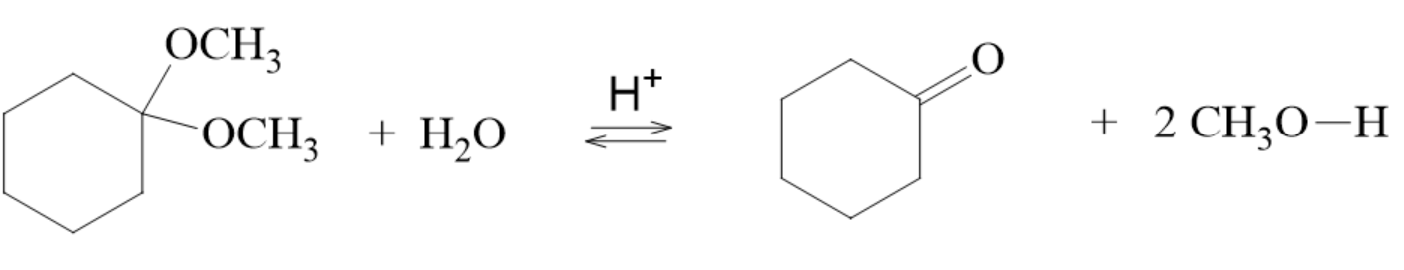
Hydrolysis of Acetals
- : a reaction where water breaks the bonds in a compound
- The hydrolysis of an acetal is the reverse reaction of acetal formation
- This is a reversible reaction and is controlled by the amounts of reactants used
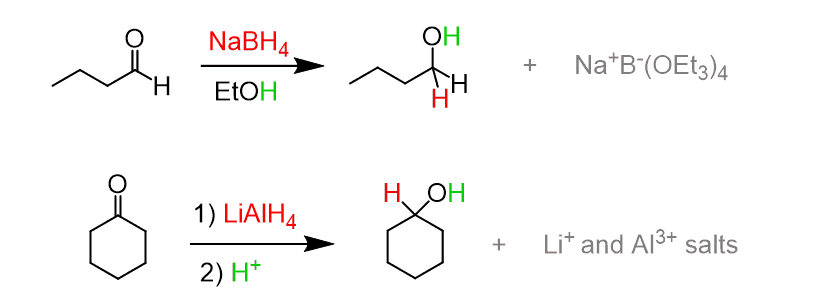
Reduction of Aldehydes to Alcohols
- This reaction can be done using one of two agents
- Sodium borohydride
- Lithium aluminum hydride (LAH)
- also reduces carboxylic acids, esters, and amides