Conversion Analysis

Answer needs to have the same Sig Figs as the given unit
255x
Density = mass/volume
mass = kg volume - l
Change In Temp Formula
T = Temperature
Matter and Energy
Matter is anything that occupies space and has mass
- Water, Solids, Liquids

Energy unit - joules
When potential energy goes up, kinetic goes down, and vice versa
1 Cals = 1000 cals
SOLIDS
Crystalline Solid - atoms or molecules arranged in geomtric patterns with long range, repeating order
salts, diamond, etc.
geometric shapes reflect the geometric arrangements of their atoms
Amorphous Solid - Atoms or molecules do not have long-range order
glass, rubber, plastic
Solid particles vibrate in place, meaning they are have very low kinetic energy
lots of potential energy
LIQUIDS
atoms and moleculsare close but move around freely, high kinetic energy
have a fixed volume because molecules ar close
assumes the shape of container
not compressible, low potential energy
Boiling point of water - 212F 100C 373K
GASES
compressible
no fixed shape
no fixed volume
very high kinetic energy
low potential energy
Elements
- Pure substance that cannot be broken down into simpler substances
no chem transformation can decompose an element
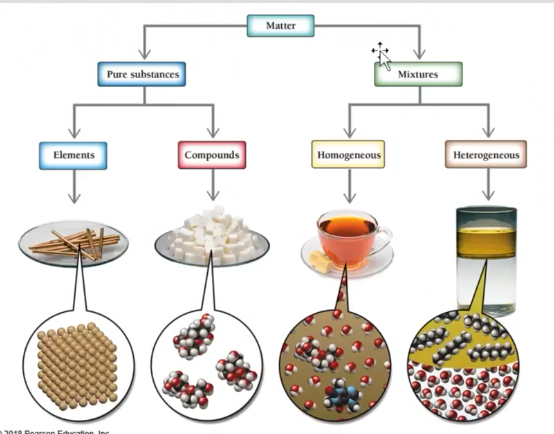
Heterogenous mixture - can see all the different components of a mixture
Homogenous - Uniform in appearance throughout mixture
Physical property - substance displays but doesnt change its composition
chemical property - substance displays only through changing its composition
Reactants always ——→ products
reactants always on left, products always on right
Boiling Points
Every liquid has is its own boiling point