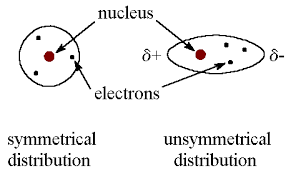
Intermolecular Forces
The forces that bond atoms together within a molecule are called intramolecular forces
The forces between atoms are intermolecular forces. They are weak attractions compared to the attraction between atoms and are easily broken by adding energy.
Intermolecular forces:
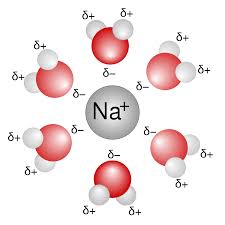
Ion dipole.
ionic with polar, cation with an anion
ionic substances dissolve easily in water (polar) by dissociating into ions
the force of attraction between ion and polar end is enough to overcome the force of attraction holding the ionic compound together
“ like dissolves like “ “ charged dissolves charged”
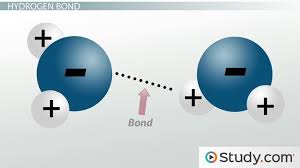
Dipole dipole
polar with polar
there is electrostatic attraction between oppositely charged ends of polar molecules
these forces usually occur in liquids
polar molecules will orient themselves so that the oppositely charged ends of the molecules are near to one another
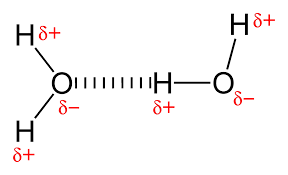
a type of dipole dipole
hydrogen bonding
hydrogen in a molecule with O, N, or F - highly electronegative atoms
stronger than regular dipole dipole forces, but still weaker than ion dipole
the positive nucleus of hydrogen is attracted to the negative dipole of the neighboring molecule
induced dipole forces
dipole dipole
caused by the charge on one molcules causing a change in the distribution of electrons
inducing a dipole is a temporary polariziation
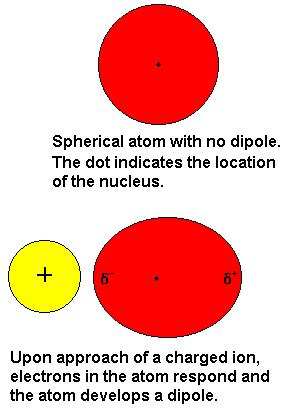
ion induced dipole forces
ion and non polar
results when an ion in close proximity to a nonpolar mlecule distorts the electron density of the non polar molecule
the molecule becomes momentarily polirized and the two molecules will be attracted
to one another
dipole induced dipole forces
polar and non polar
the charge of a polar molecule is responsible for inducing the charge (by distortion of the electron cloud) on the nonpolar molecule
the molecule becomes momentarily polarized and the two molecules will be attracted to one anotheer
london dispersion forces
nonpolar and non polar
act between any particilies, but is the only force that acts between nonpolar molecules
occur for only an instant due to the molecules bonds vibrating and having a mometary, uneven distribution
molecules continue to do this on a random, ongoing basis
very short lived and extremely weak