Acids and Alkalis Study Notes
CHAPTER 14: INTRODUCTION TO ACIDS AND ALKALIS
A. Notes
Focuses of this chapter:
Reactions of typical dilute acids (with metals / bases / carbonates / hydrogencarbonates)
Ionization of acids and alkalis in water
Basicity of acids
Bases and alkalis
Reactions of alkalis (with acids / non-metal oxides / ammonium compounds / metal ions)
Corrosive nature of concentrated acids and alkalis
1. ACIDS
1.1. Examples of Acids
Common laboratory acids include:
Hydrochloric acid: HCl(aq)
Sulphuric acid: H₂SO₄(aq)
Nitric acid: HNO₃(aq)
Organic and Inorganic Acids:
Organic Acids: Contain carbon and are produced by living organisms.
Examples:
Ethanoic acid: CH₃COOH
Oxalic acid: (COOH)₂
Citric acid: C₆H₈O₇
Inorganic Acids: Known as mineral acids.
Examples:
Hydrochloric acid: HCl
Sulphuric acid: H₂SO₄
Nitric acid: HNO₃
Phosphoric acid: H₃PO₄
Carbonic acid: H₂CO₃
1.2. Properties of Acids
At room temperature, pure acids can be:
Colourless gases (e.g., hydrogen chloride).
Colourless liquids (e.g., sulphuric acid, nitric acid, ethanoic acid).
White crystalline solids (e.g., citric acid, oxalic acid, tartaric acid).
Taste: All acids have a sour taste (NEVER taste in laboratory!).
Electrolytes: Aqueous solutions are electrolytes and can conduct electricity.
pH: All acids have a pH value < 7.0 (at room temperature) and can change the color of acid-base indicators:
Litmus: Red (acidic), Purple (neutral), Blue (alkaline)
Phenolphthalein: Colourless (acidic & neutral), Purple (alkaline)
Methyl Orange: Red (acidic), Yellow (neutral & alkaline)
1.3. Ionization of Acids
1.3.1. Ionization of Acids
When hydrogen chloride gas dissolves in water:
Reaction: HCl(g) → H⁺(aq) + Cl⁻(aq)
Process of forming ions is called ionization.
Definition: An acid is a hydrogen-containing molecular compound that gives hydrogen ions as the only cations when dissolved in water.
General equation of ionization:
HA(aq)\rightarrow H^{+}(aq)+A^{-}(aq)
(where A⁻(aq) is anion specific to the acid).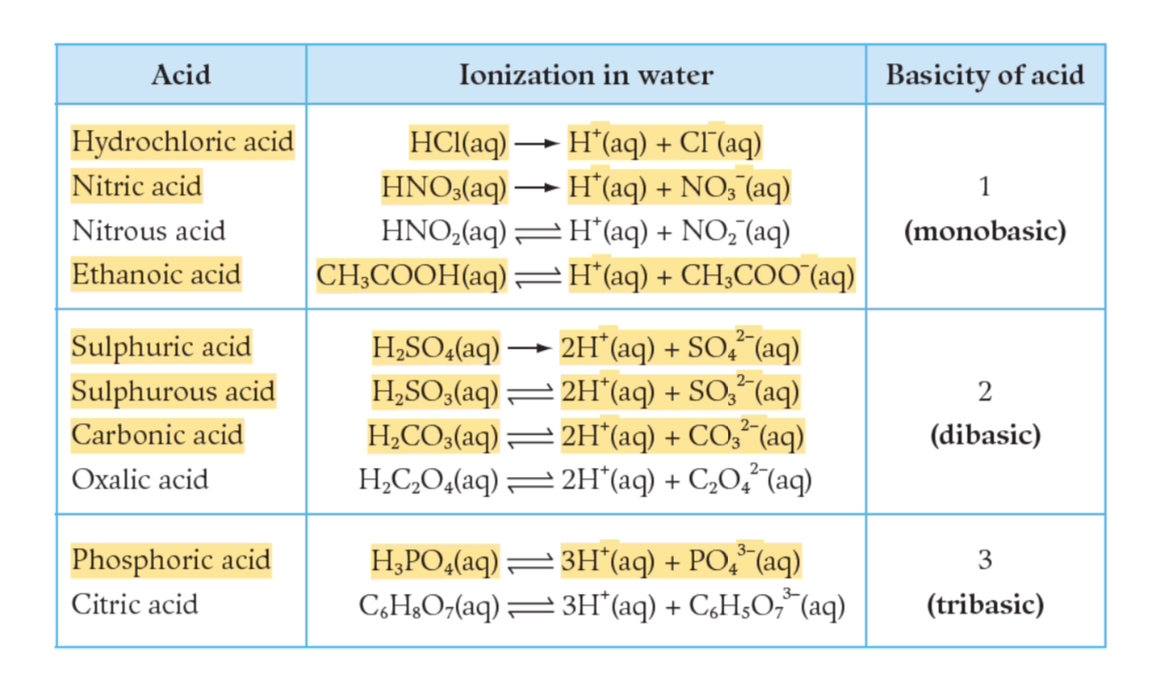
Reversible reactions
ethanoic acid ionization:
CH₃COOH(aq) ⇌ CH₃COO⁻(aq) + H⁺(aq)
The double arrow (⇌) indicates that the reaction is reversible.
Forward and backward reactions occur simultaneously.
Other examples of reversible reactions:
Production of ammonia (Haber Process): \displaylines{N_2(g)+3H_2(g)\rightleftharpoons2NH_3(g)}
Reaction between ammonia and water: \displaylines{NH_3(aq)+H_2O(l)\rightleftharpoons NH_4^{+}(aq)+OH^{-}(aq)}
Irreversible Reactions
Reactions that only proceed in one direction are referred to as irreversible reactions.
Single arrows (\rightarrow) are used in their chemical equations
1.3.2. Basicity
Definition: The basicity of an acid is the maximum number of hydrogen ions that can be produced by a molecule of the acid when ionized in water.
Example of an acid’s basicity:
Hydrochloric Acid (HCl): 1 (Monobasic)
Sulphuric Acid (H₂SO₄): 2 (Dibasic)
Phosphoric Acid (H₃PO₄): 3 (Tribasic)
Basicity may not equal total number of hydrogen atoms in the molecule (e.g., Ethanoic Acid: CH₃COOH).
1.4. Acid Reactions
1.4.1. With Metals
General equation:
metal + saltPredicted reactivity: Dilute hydrochloric and sulphuric acids react with metals above copper in the reactivity series.
Example 1: Zinc + Dilute hydrochloric acid
Chemical equation: Zn(s) + 2HCl(aq)
ightarrow ZnCl₂(aq) + H₂(g)Ionic equation: Zn(s) + 2H^+(aq)
ightarrow Zn^{2+}(aq) + H₂(g)Observations:
Colourless gas bubbles evolve.
Solution becomes warm.
Example 2: Lead + Dilute sulphuric acid
Chemical equation: Pb(s) + H₂SO₄(aq)
ightarrow PbSO₄(s) + H₂(g)Observation: Colourless gas bubbles evolved, but reaction stops when lead(II) sulphate forms, blocking further contact.
1.4.2. Neutralization Reactions
General equation:
ext{Acid} + ext{Base}
ightarrow ext{Salt} + ext{Water}Example: Sodium hydroxide + Dilute hydrochloric acid:
Chemical equation: HCl(aq) + NaOH(aq)
ightarrow NaCl(aq) + H₂O(l)Ionic equation: H^+(aq) + OH^−(aq)
ightarrow H₂O(l)