Hydrocarbons reactions
Alkane to alkene remove hydrogen (2)
Alkene to alkane add hydrogen (2)
Reactions of alkanes
Subsitution
combustion -Burn an alkane in oxygen to make carbon dioxide and water
CnH2n+2 + O2 à nCO2 + (n+1)H2O
halogenation - substitution one hydrogen for one halogen atom (F, CL, Br, I) - UV light catalyse reaction
CnH2n+2 + O2 à nCO2 + (n+1)H2O
dehydrogenation - removing (2) hydrogen → an alkene
Uses: oil refining industry to manipulate moecules
heat as catalyst
CnH2n+2 à CnH2n + H2
Dehydrogenation of propane produces propane
CH3-CH2-CH3 à CH3-CH=CH2 + H2
Reaction to Alkenes
Combustion - burn in oxygen gas produce carbo dioxide and water
Addition - most useful and common reactions of alkenes
Break double bond and use those electrons to bond with other atoms
Reduction - the loss or gain of hydrogen atoms
catalytic reduction of alkene is also called hydrogenation
alkene + H2 (g) à alkane
needs a metal catalyst like Pd, Al, Pt or Ni.
oxidation increase oxygen content of molecule
permanganate goes from purple to brown
Dichromate goes from orange to green
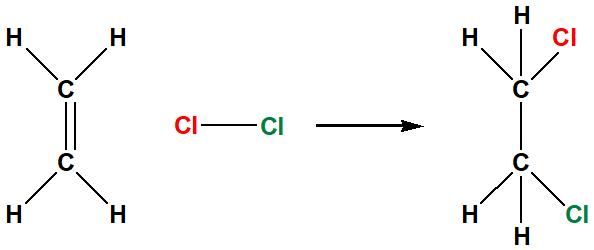
halogenation Cl2 and Br2 only
Break the alkene double bond and break halogen to halgon bond
Hydrohalogenation
rate of reaction increases with acidity
When HX adds t symmetric alkene - doesn’t matter which carbon of the alkene group gets bonded to H
When HX adds to an asymmetric alkene
Markovnikov rule: H will add to carbon of the double bond that already has the most H bonded directly to it
Hydration - adding water to alkene, acid catalyst to start e.g Hcl , H2SO4
Polymerisation - joining together of monomer units without loss of atoms
Reactions of aromatic Hydrocarbons
Combustion - produces CO2 + H2O
Subsitution - 1/+ of the hydrogens around beneze ring can be replaced by another atom/group
halogenation
add -Cl, -Br, -I to aromatic ring
nitration
add -NO2 group
sulfonation
add -SO3H group
alkylation
add -R group where R = CH3, CH2CH3 etc
acylation
add -COR group, where C is double-bonded to O