4: Organisms and their adaptations to the environment
Adaptations to the Environment
Goals
Understand environmental controls on organisms
Obtain a general appreciation for adaptations
Discuss applications: invasive species, environmental assessment
Reading Guide
Chapter 3: The challenges of living in Aquatic Environments
Chapter 4: The challenges of living in Terrestrial Environments
Importance
The environment sets the stage, organisms have to adapt or move to other locations.
Understanding responses to the environment allows us to:
Understand adaptations
Determine habitat use
Draw species distributions
Manage invasive species
an example: red devils
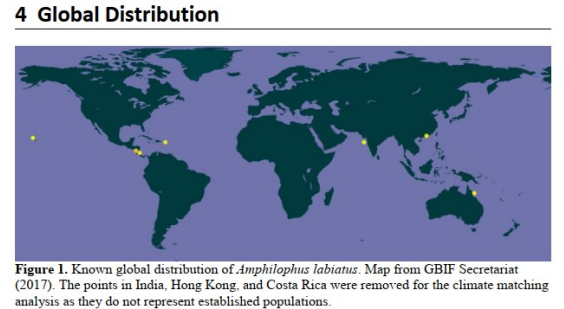
Key adaptations
Low flow
High solutes
Reproduces in cavities, under rocks
Bottom feeder, omnivorous
Understanding the how organisms respond and adapt to their environments help us understand their current distribution and response to change.
Aquatic environments
Water properties
Water is key for life.
Thermal properties
Density
Viscosity
Solvent
Hydrogen and pH
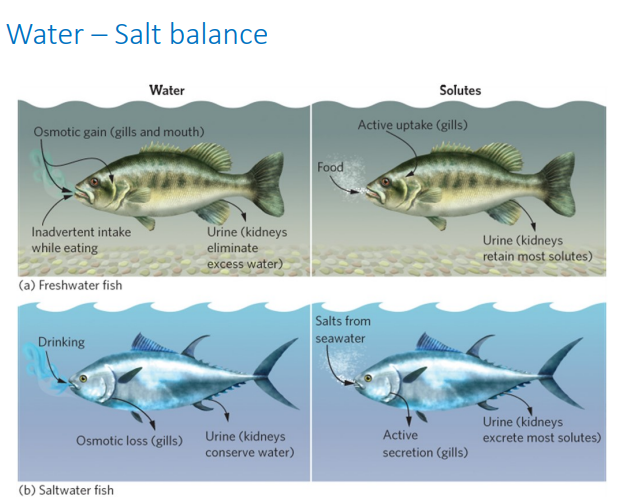
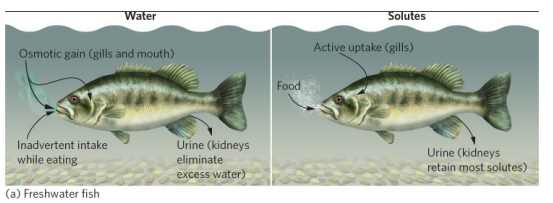
Osmoregulation: Freshwater
Hyperosmotic
High solutes in tissue than in water
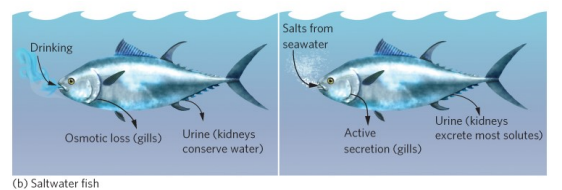
Osmoregulation: Saltwater
Hyposmotic
High solutes in water
Aquatic Environments
Saltwater
Food - Proteins - Nitrogen
Ammonia
simple way to excrete excess N
toxic
Urea
more complex
saves water
Urea as adaptation
sharks and rays
balance salts in body vs environment
require further adaptations
trimethylamine
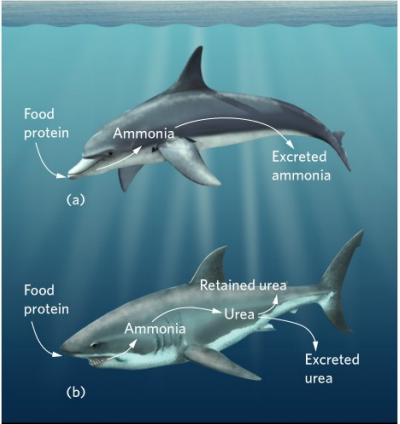
Saltwater - energy cost
landlocked lakes
hypersaline


Osmotic balance
road salt impacts
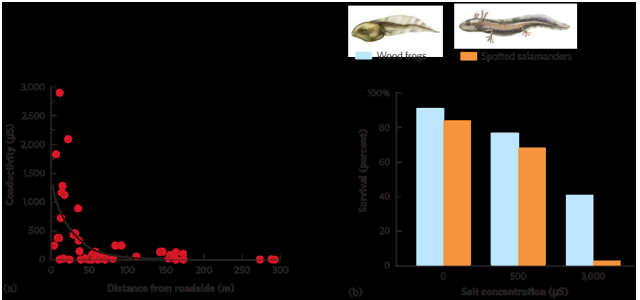
Gas exchange
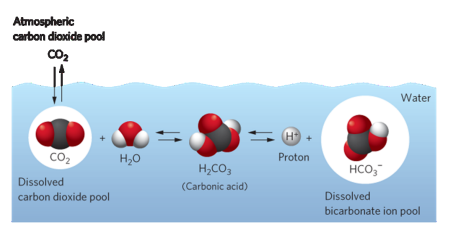
carbon dioxide
not as abundant in water
CO2 and HCO3- stable molecules
CO2 abundant in low pH environments
moves slowly in water
CO2 smaller than HCO3-
Boundary layer
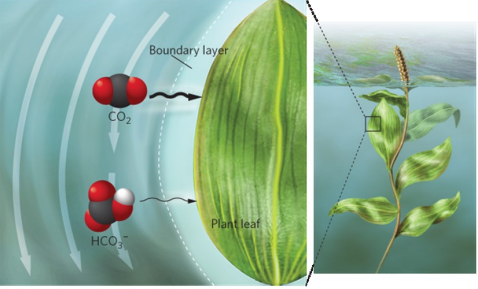
Gases
Oxygen exchange
not as abundant in water
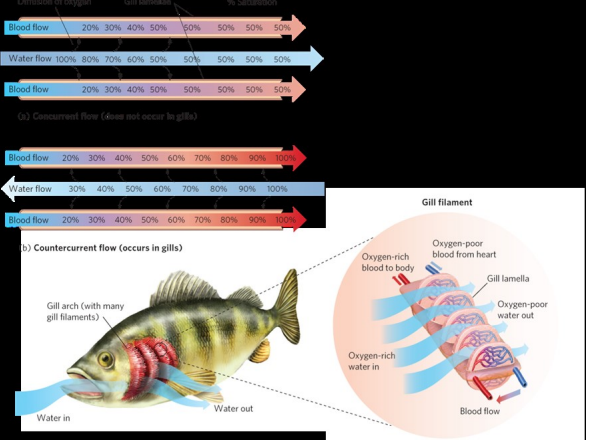
concurrent vs countercurrent circulation
Water pH
CO_2 - H2O balance
pH = -log (H+ concentration)
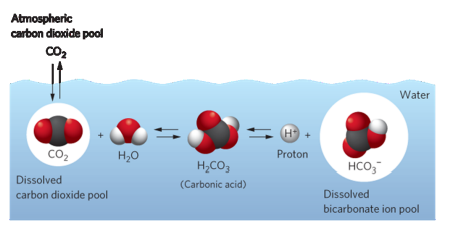
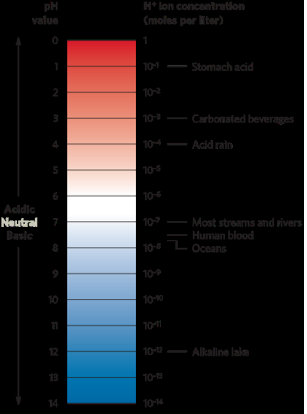
Episodic acidification
La Selva biological Station, Costa Rica

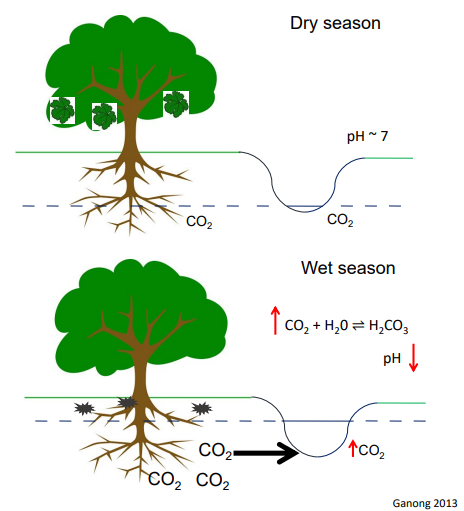
Aquatic organisms appear to be well - adapted to episodic events
Vulnerable only to extreme declines in pH
There is an energy cost!

Adaptations to the Environment
Summary
Adaptations to aquatic environments
Importance
Ion balance
Gas exchange
pH