Atmosphere
Isotope fractionation in the hydrosphere
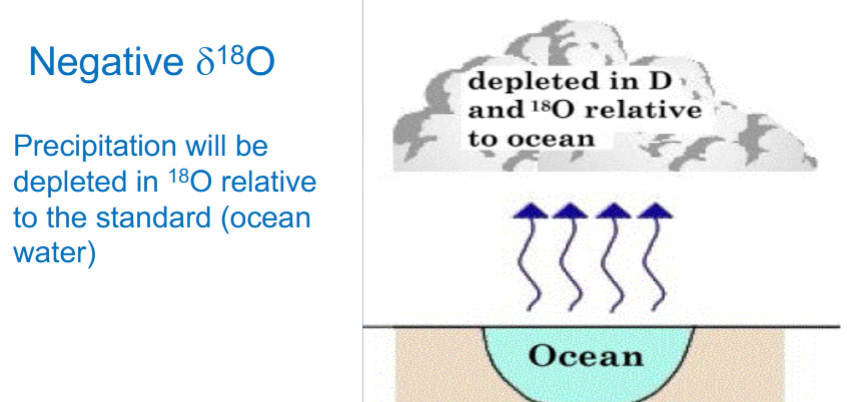
Evaporation of surface water in equatorial regions - air masses of H2O reduced in 18O and D compared to seawater (so means that the amount of vapor depends on the temperature)
Air is forced in more cooler air in the northern atmosphere - water condenses (enriched in 18O and D compared to vapor remaining)
Remaining vapor = Isotopic composition of first precipitation (18O and D are removed from vapor, vapor reduces)
18O and D become increasingly negative with geographic latitude and altitude


Intergla
cial conditions:
18O water is heavier (harder to evaporate) — glaciers ate poor in 18O — oceans rich in 18O
Glacial conditions:
Less heat, less energy for evaporation, 18O even harder to evaporate — glaciers get even less 18O — oceans get even richer in 18O
Indicators in oceans, higher 18O:16O = colder climates
Indicators on land glaciers, lower 18O:16O ratios = colder climates
The terrestrial mass-dependent fractionation line is characterized by a distinct slope that reflects the isotopic composition changes associated with different environmental conditions, particularly during glacial and interglacial periods. Also, their fractionation depends on their mass difference (proportional). The slope is 0.52, and its intercept is 0.
We can also find evidence of solar gas isotopes gain on Earth
D/H graph:
So yes, the D/H ratio tells us that Earth's water came from early solar system material, but probably not directly from the Sun — rather from bodies that inherited or fractionated those isotopes.
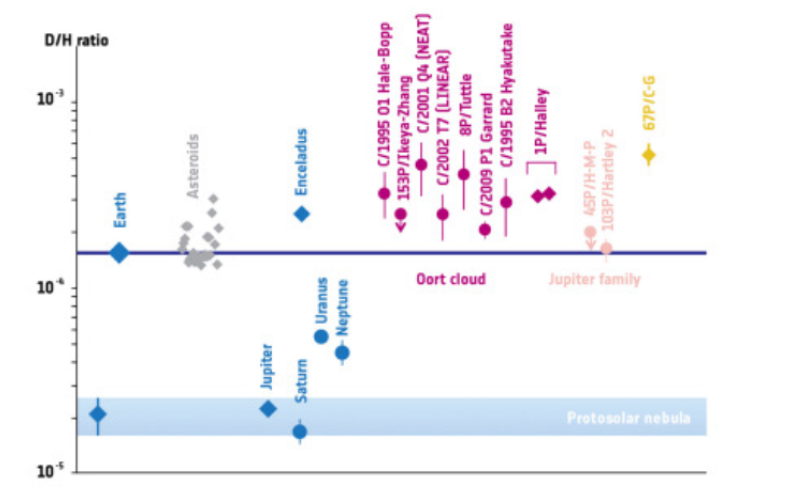
Higher D/H means the material came from colder regions (like comets or interstellar space) and heavier isotopes.
Lower D/H means it formed in hotter regions (like near the Sun).
In Summary: The Big Picture
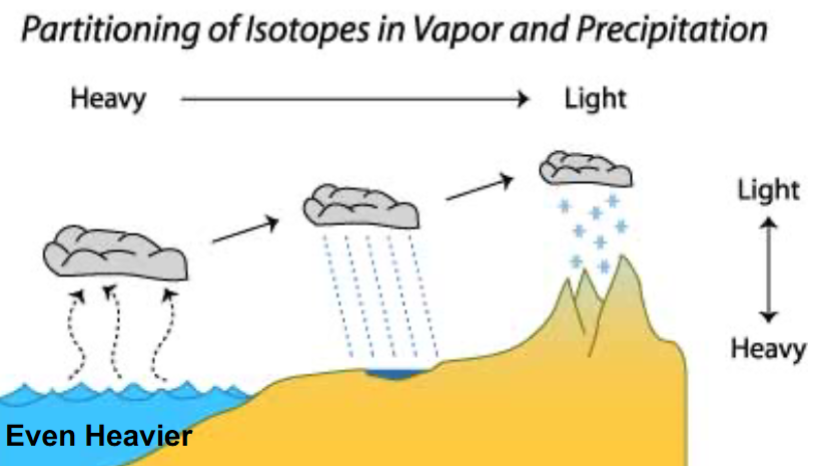
Evaporation and Precipitation: The lightest water molecules (those with less 18O and D) are more likely to evaporate and leave the ocean, and as air cools, heavier molecules (18O and D) condense out, enriching precipitation with heavier isotopes.
Climate and Isotopes: Warmer climates (interglacial periods) have water that’s richer in 18O, while colder climates (glacial periods) have water that’s poorer in 18O. This pattern helps scientists figure out past climates.
D/H Ratio: The D/H ratio helps scientists understand where Earth's water came from. Higher D/H means it came from colder places, while lower D/H means it came from hotter places near the Sun.
All of these processes show how water’s isotopes can act like a "time capsule," revealing information about Earth’s past, its climate, and even where our water originally came from in the solar system.