4.1-4.3 Symbols, equations and Formulae
Chemical formulae
- The structural formula describes how the atoms of a given molecule are connected.
- This can be done with either a diagram (shown formula) or a written formula (simplified structural formula), The empirical formula determines the simplest whole-number ratio of each element's atoms in a compound.
- The molecular formula indicates how many atoms of each element are present in one molecule of the compound or element.
- E.g. H2 has two hydrogen atoms, while HCl contains one hydrogen atom and one chlorine atom.

- Structural formula (simplified)
CH3CH2CH2CH3
- Molecular formula
C4H10
- Empirical formula
C2H5
\
Using valency to deduce formulas
- The concept of valency is utilized to deduce compound formulas.
- The valency, or combining power, of an atom indicates how many bonds it can form with another atom. Carbon, for example, belongs to Group IV, which means that a single carbon atom can form four single bonds or two double bonds.
- Each group's constituents have the following valencies:
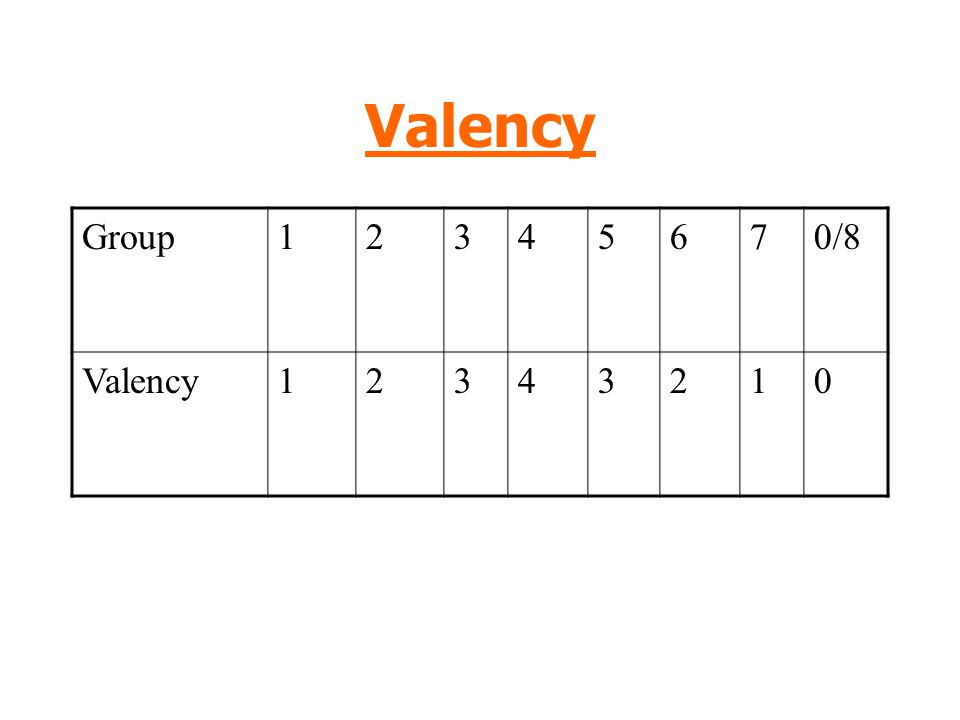
What is the formula of aluminium sulfide?

Ionic Compound Formulae Deduction
- If you know the charge on the ions, you can compute the formulas for these compounds.
- The charges of several common ions are listed below. The table includes a number of common compound ions.
- These ions are referred to as polyatomic ions by certain chemists.
The Charges of Common Ions Table

- The overall sum of the charges of an ionic compound should be 0
- You therefore need to work out the ratio of the ions to ensure this is the case
- When you write the formula of a compound ion it is necessary to use brackets around the compound ion where more than one of that ion is needed in the formula
- For example copper(II)hydroxide is Cu(OH)2
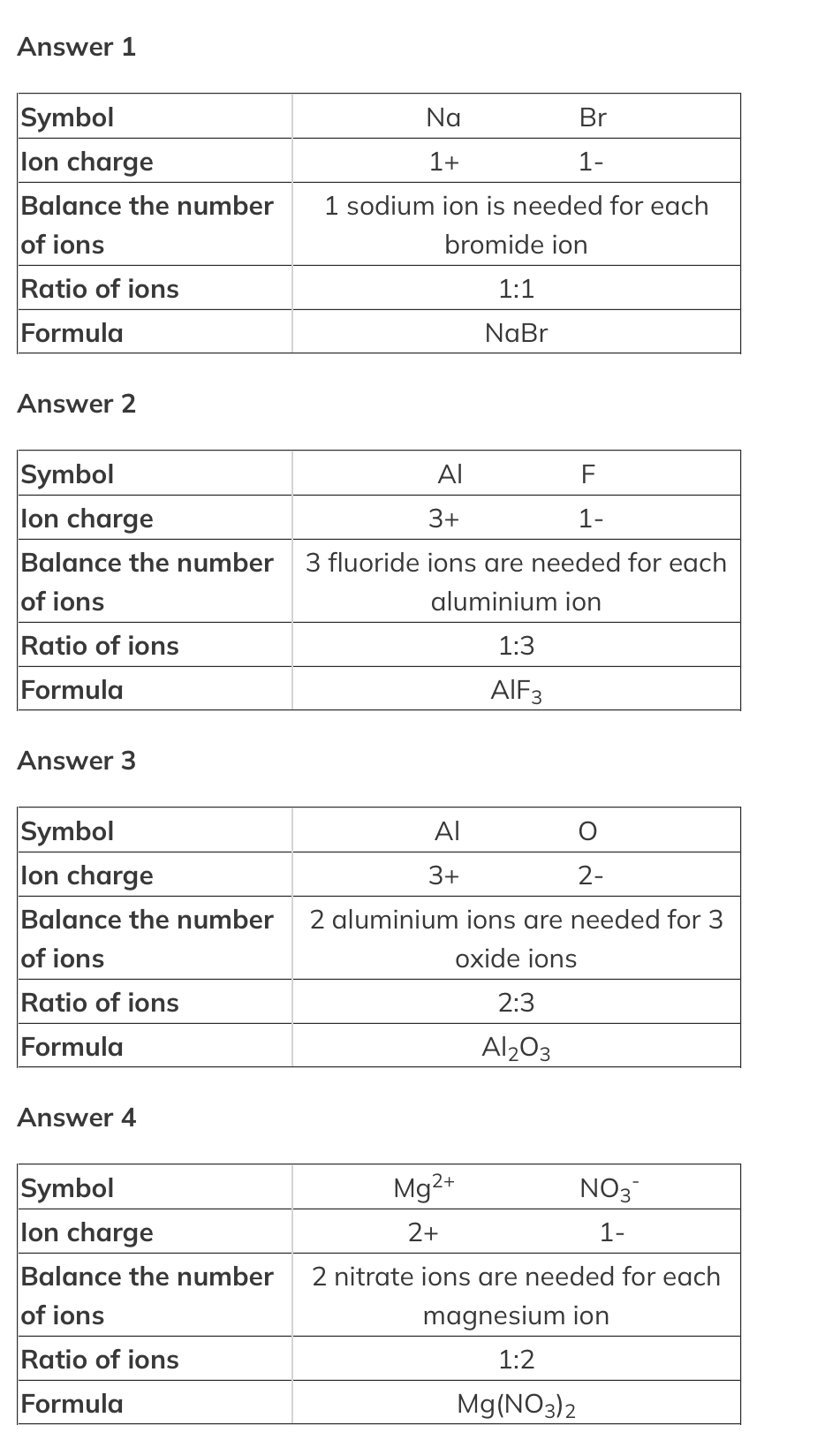
What is the formula of?
- sodium bromide
- aluminium fluoride
- aluminium oxide
- magnesium nitrate
Writing Equations and Balancing
Word Equations
- These show the reactants and products of a chemical reaction using their full chemical names
- The arrow (which is spoken as “goes to” or “produces”) implies the conversion of reactants into products
- Reaction conditions or the name of a catalyst can be written above the arrow
- An example of an word equation for neutralisation is:
sodium hydroxide + hydrochloric acid → sodium chloride + water
- The reactants are sodium hydroxide and hydrochloric acid
The products are sodium chloride and water 💦
Compounds' names
For compounds consisting of 2 atoms:
- If one is a metal and the other a nonmetal, then the name of the metal atom comes first and the ending of the second atom is replaced by adding -ide * E.g NaCl which contains sodium and chlorine thus becomes sodium chloride
- If both atoms are nonmetals and one of those ishydrogen, then hydrogen comes first * E.g. Hydrogen and chlorine combined is called hydrogen chloride
- For other combinations of nonmetals as a general rule, the element that has a lower group number comes first in the name
- E.g. carbon and oxygen combine to form CO2 which is carbon dioxide since carbon is in Group 4 and oxygen in Group 6
- For compounds that contain certain groups of atoms: \n * There are common groups of atoms which occur regularly in chemistry * Examples include the carbonate ion(CO32-), sulfate ion (SO42-), hydroxide ion (OH-) and the nitrate ion (NO3-) * When these ions form a compound with a metal atom, the name of the metal comes first
- E.g. KOH is potassium hydroxide, CaCO3 is calcium carbonate
Writing and balancing equations
- Chemical equations use the chemical symbols of each reactant and product
- When balancing equations, there needs to be the same number of atoms of each element on either side of the equation
- The following nonmetals must be written as molecules: H2, N2, O2, F2, Cl2, Br2 and I2
- Work across the equation from left to right, checking one element after another
- If there is a group of atoms, for example a nitrate group (NO3-) that has not changed from one side to the other, then count the whole group as one entity rather than counting the individual atoms. * Examples of chemical equations: * Acid-base neutralisation reaction: \n NaOH (aq) + HCl (aq) ⟶ NaCl (aq) + H2O (l) * Redox reaction: \n 2Fe2O3 (aq) + 3C (s) ⟶ 4Fe (s) + 3CO2 (g)
- n each equation there are equal numbers of each atom on either side of the reaction arrow so the equations are balanced
- The best approach is to practice lot of examples of balancing equations
- By trial and error change the coefficients (multipliers) in front of the formulae, one by one checking the result on the other side
- Balance elements that appear on their own, last in the process
Example 1 😻
aluminium + copper(II)oxide ⟶ aluminium oxide + copper
Unbalanced symbol equation:
Al + CuO ⟶ Al2O3 + Cu


- Sometimes it can be hard to know what the correct state symbol is and we have to look for clues in the identity of substances in a reaction
- Generally, unless they are in a solution: * Metal compounds will always be solid, although there are a few exceptions * Ionic compounds will usually be solids
- Non-metal compounds could be solids, liquids or gases, so it depends on chemical structure
- Precipitates formed in solution count as solids
- In the worked examples above the final equations with the state symbols would be * 2Al (s) + 3CuO (s) ⟶ Al2O3 (s) + 3Cu (s) * MgO (s) + 2HNO3 (aq) ⟶ Mg(NO3)2 (aq) + H2O (l)
Balancing Ionic Equation

AR and MR
Relative atomic mass
- The symbol for the relative atomic mass is Ar
- The relative atomic mass for each element can be found in the periodic table along with the atomic number
\

- Atoms are too small to accurately weigh but scientists needed a way to compare the masses of atoms
- The carbon-12 is used as the standard atom and has a fixed mass of 12 units
- It is against this atom which the masses of all other atoms are compared
- Relative atomic mass (Ar*)* can therefore be defined as: * the average mass of naturally occurring atoms of an element on a scale where the 12C atom has a mass of exactly 12 units
- The relative atomic mass of carbon is 12 * The relative atomic mass of magnesium is 24 which means that magnesium is twice as heavy as carbon * The relative atomic mass of hydrogen is 1 which means it has one twelfth the mass of one carbon-12 atom
The relative atomic mass of an element can be calculated from the mass number and relative abundances of all the isotopes of a particular element using the following equation:
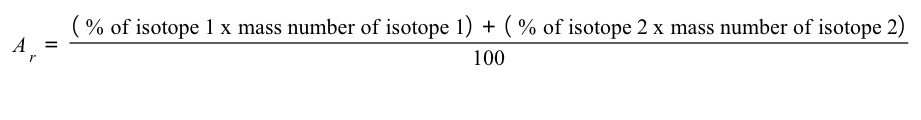
The table shows information about the Isotopes in a sample of rubidium

Relative formula mass Calculation

Calculating Percentage mass
Calculate the percentage of iron in iron(III) oxide, Fe2O3.
RAM (Ar): Fe = 63.5 O = 16

\ \