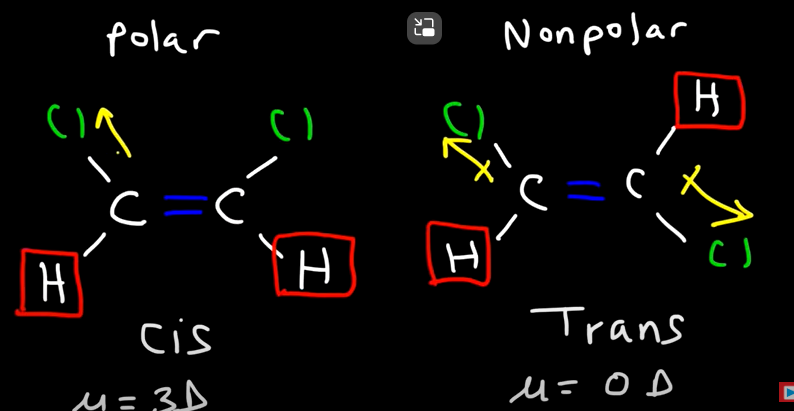
Difference between Cis and Trans isomers
Cis: atoms are on the same side Ex: hydrogen
Trans: atoms are on different side ex: hydrogen
*Isomer can affect the polarity of a molecule
-affect boiling point
If carbons are attached to two different groups, then it can have trans/cis isomers
*look at where there is a double bond, if the ends are different: goanna have cis/trans face