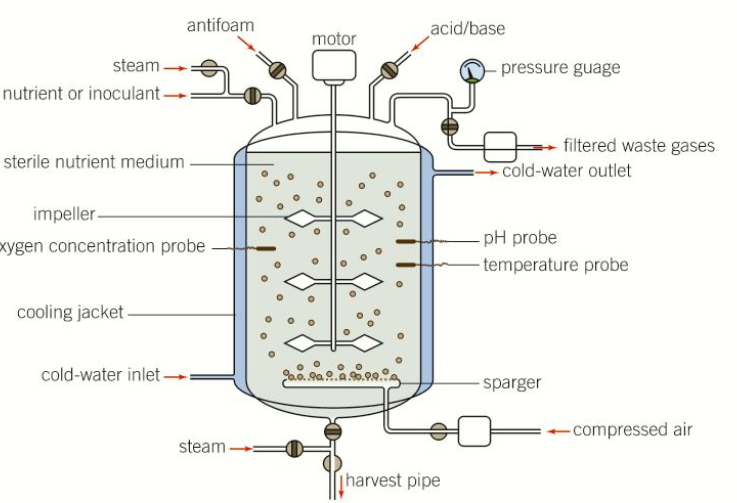
22.7 Culturing microorganisms on an industrial scale
In any bioprocess, the microorganism involved must be able to synthesis or break down the chemical required, work reasonably fast, give a good yield of the product, use relatively cheap nutrients, and not require extreme (expensive) conditions. It must not produce any poisons that contaminate the product or mutate easily into non-functioning forms.
Primary and Secondary metabolites
Primary metabolites - substances that are wanted which are formed as an essential part of the normal functioning of a microorganism e.g ethanol (product of anaerobic respiration in yeast), ethanoic acid and a range of amino acids and enzymes.
Secondary metabolites - organisms produce substances which are not essential for normal growth but are still used by cells e.g pigments and the toxic chemicals plants produce to protect themselves against attack by herbivores. The organism wouldn’t suffer in the short term without them (often required in the product of bioprocess)
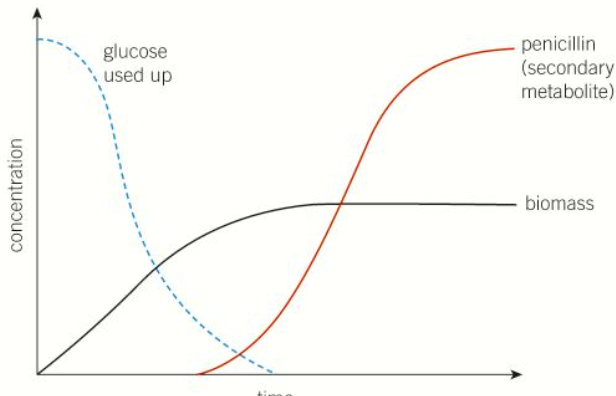 Primary metabolites are usually formed in the period of active growth, whilst secondary metabolites tend to be formed during the stationary phase of the life of the culture once the cell mass has reached it’s maximum.
Primary metabolites are usually formed in the period of active growth, whilst secondary metabolites tend to be formed during the stationary phase of the life of the culture once the cell mass has reached it’s maximum.
Types of bioprocess
Once a microorganism has been chose, ideal size and shape of the bioreactor (reaction vessel) decided, the organisation of the commercial production then has to be decided, there are two main ways, batch fermentation and continuous fermentation.
Batch fermentation
The microorganisms are inoculated into a fixed volume of the medium.
As growth occurs, nutrients are used up and both new biomass and waste products build up.
As the culture reaches the stationary phase, overall growth ceases but during this phase the microorganisms often carry out biochemical changes to form the desired end products (like antibiotics and enzymes)
The process is stopped before the death phase and the products harvested. The whole system is then cleaned and sterilised and a new batch culture is started up.
Continuous culture
Microorganisms are inoculated into sterile nutrient medium and start to grow
Sterile nutrient medium is added continually to the culture once it reaches the exponential point of growth.
Culture broth is continually removed - the medium, waste products, microorganism and the product - keeping the culture volume in the bioreactor constant.
Continuous culture enables continuous balanced growth, with level of nutrients, pH and metabolic products kept more or less constant.
Both methods of operation a bioreactor can be adjusted to ensure either the maximum production of biomass or the maximum production of the primary or secondary metabolites.
The majority of industrial processes use batch or semi-continuous cultivation.
Continuous cultivation is largely used for the production of single-celled protein and in some wastewater treatment processes.
All bioreactors produce a mixture of unused nutrient broth, microorganisms, primary metabolites and possibly secondary metabolites and waste products. → the useful part of the mixture has to be separated out by downstream processing, this is one of the most expensive parts of the whole bioprocess.
Cost can vary from 15-40% of the whole process.
Controlling bioreactors
Temperature
If too low - they will not grow quickly enough
If too high - they will denature, inhibited or destroyed.
Bioreactors often have a heating and or cooling system linked to temperature sensors and a negative feedback system to maintain optimum conditions.
Nutrients and oxygen
Can be added in controlled amounts to broth when probes or sample tests indicate that the levels are dropping.
Mixing things
Inside there are large volumes of liquid, simple diffusion is not enough to ensure all microorganisms receive enough food and oxygen or it is kept at the right temperature, therefore often have a mixing mechanism to stir continuously.
Asepsis
Most bioreactors are sealed, aseptic units.
If this process involves genetically engineered organisms then it is a legal requirement that they should be contained within the bioreactor and not released into the environment.