Ap Bio: Unit 3: Enzyme Stuff
Topic 3.1: Enzyme Structure
Enzymes are biological catalysts that speed up chemical reactions by lowering activation energy.
Structure: Enzymes are proteins with a specific 3D shape, including an active site where the substrate binds.
Substrate specificity: The enzyme's active site is complementary in shape to its substrate, fitting like a "lock and key."
Induced fit model: The enzyme slightly changes shape to better fit the substrate when binding occurs.
Topic 3.2: Enzyme Catalysis
Enzyme-substrate complex: The intermediate formed when an enzyme binds to its substrate.
Activation energy: The energy required for a reaction to occur. Enzymes lower the activation energy.
Reaction rate: Measured by the change in product concentration over time.
Effects of inhibitors:
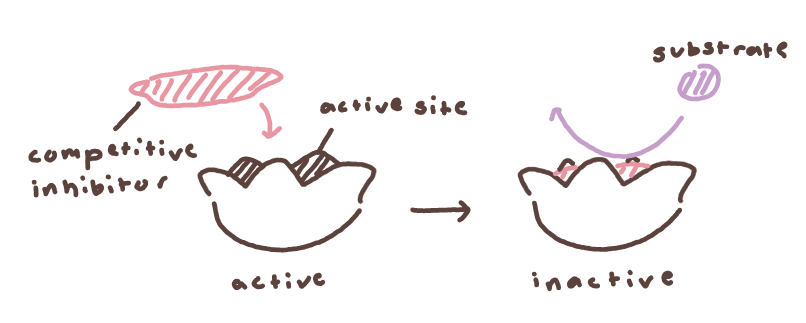
Competitive inhibition: Inhibitor competes with the substrate for the active site. Can be overcome by increasing substrate concentration.
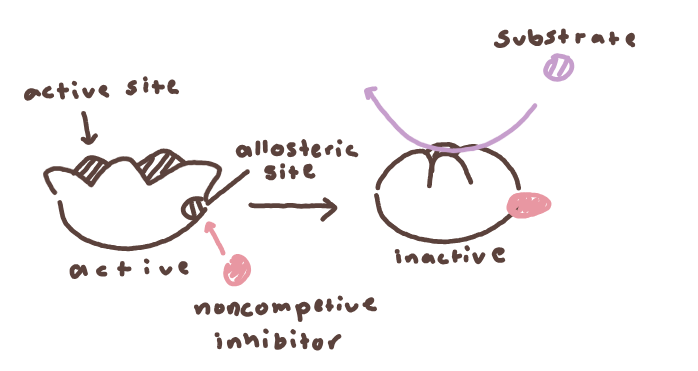
Non-competitive inhibition: Inhibitor binds to an allosteric site (not the active site), changing enzyme shape and decreasing its activity.
Lab Investigation:
Enzyme activity can be studied by measuring how fast substrates are consumed or products are formed (e.g., catalase breaking down hydrogen peroxide into water and oxygen).
Topic 3.3: Environmental Impacts on Enzyme Function
Environmental conditions can alter enzyme shape and function:
Temperature: Extreme temperatures can denature enzymes, while optimal temperatures increase reaction rates.
inc in temp outside optimum range will inc activity, then later denature
dec in temp outside optimum range will slow activity, never denature
pH: Enzymes work best within a specific pH range. Too acidic or basic environments can disrupt bonds in the enzyme, leading to denaturation.
if outside optimum range → dec in enzyme activity, later denatured
Salt concentration (Salinity): Ionic conditions affect enzyme structure. Too much or too little can disrupt bonding in the enzyme's structure.
Enzyme Concentration:
inc → inc activity
dec → dec activity
Substrate Concentration:
inc will inc, and later stay constant to a certain point
Denaturation and Cofactors/Coenzymes
Denaturation is the irreversible process of an enzyme losing its shape and, consequently, its function due to environmental changes (like heat or pH).
Changes in shape cause denaturation
active site can no longer bind to specific substrate
denaturation can be reversible in some cases
Cofactors and coenzymes (non-protein helpers that assist enzyme function)
Cofactors- Inorganic cofactors
Consists of metals
Holoenzyme : An enzyme with the cofactor attatched
Coenzymes- Organic cofactors
ex: vitamins
Topic 3.4: Cellular Energy
Energy in Cells:
Cells use energy for processes like growth, repair, and maintenance.
ATP (Adenosine triphosphate) is the main energy currency of the cell.
ATP stores energy in its phosphate bonds and releases energy when it's hydrolyzed to ADP (adenosine diphosphate) and an inorganic phosphate.
Energy Coupling:
Cells couple exergonic reactions (release energy) with endergonic reactions (require energy).
Exergonic- “Downhill reaction, SPONTANEOUS, releases energy (Catabolic) (Degradation) (AB —> A+B+Energy)
Endergonic- “Uphill” reaction, REQUIRES ACTIVATION ENERGY, absorbs energy (Synthesis/Anabolic) (A+B —> AB)
Metabolism:
Catabolic pathways: Break down molecules and release energy (e.g., cellular respiration).
Anabolic pathways: Build molecules and require energy (e.g., photosynthesis).
Energy
Kinetic Energy- Energy associated with motion
Thermal Energy- Energy associated with the movment of atoms or molecules
Potential Energy- Energy that is stored
Chemical Energy- Potential energy available for release in a chemical reaction
Thermodynamics in Biology:
First Law of Thermodynamics: Energy cannot be created or destroyed, only transformed.
Second Law of Thermodynamics: Every energy transfer increases the entropy (disorder) of the universe.
During energy transfers or transformations, some energy is unusable and often lost as heat