Inhalation Anesthetics (Volatile Gases)
Definition: Inhalation anesthetics are inorganic gases (N2O) or volatile liquids converted into gas (sevo, iso, des) that are administered via the lungs to produce general anesthesia.
Benefit over IV agents:
Ability to monitor endtital anesthetic concentration (because of gas analysis called mass spec), which gives direct feedback on the concentration in lungs (and remember, what is in the lungs is directly representing what is in the BRAIN). Remember, though, that the % you dial a vaporizer at does not necessarily mean that is the amount that is in the body; you should titrate based off the output value! Inspiration will be higher than expiration until equilibrium is reached.
breath to breath adjustments by turning a vaporizer dial
Potent bronchodilators
Meyer-Overton Hypothesis: suggests an old school way of thinking how the mechanism of action of volatile anesthetics works:
States that lipid solubility of anesthetics is directly proportional to their potency as inhaled anesthetics.
Stronger potency = lower MAC
Suggests that depth of anesthesia is determined by the number of anesthetic molecules dissolved in the brain rather than depending on the specific anesthetic agent used.
New school way of thinking how the mechanism of action of volatile anesthetics works: General anesthesia is produced via many different interactions with membrane-bound proteins in the brain and spinal cord.
Enhancement of inhibitory ion channels: Agonizes GABA, Glycine, and potassium channels. Hyperpolarization occurs.
Blocking excitatory channels: Antagonizes NMDA/glutamate, nicotinic, and sodium channels. Depolarization is blocked.
Locations for volatile anesthetics:
GABA-A agonism (to support inhibition) is the primary site for effects in the brain and increases Chloride influx, which then hyperpolarizes neurons (so they are unable to depolarize). They most likely prolong the duration that chloride channels stay open.
The brain has the following primary action sites: glycine receptor agonism (to support inhibition), NMDA antagonism (to create inhibition), sodium channel inhibition (to create inhibition); NOT GABA. Volatile anesthetics induce immobility at the ventral/anterior horn.
N2O (inorganic gases) MOA: NMDA antagonism and stimulation of potassium 2P channels. NO stimulates GABA-A receptors!!
Target regions:
Unconsciousness (higher order cerebral functions): Cerebral cortex, thalamus, reticular activating system (RAS)
Amnesia (memory formation and response to pain): Amygdala, hippocampus.
Immobility (upper/lower motor neuron synapse): Ventral/anterior horn.
Analgesia (nociceptive signals along the ascending pathway): Spinothalamic tract
Autonomic modulation: pons and medulla.
Vapor Pressures
Contemporary volatile anesthetics exist in liquid form below 20°C. If independent of volume or atmospheric pressure, a volatile liquid in a closed container will have molecules escape from the liquid phase to the vapor phase.
vice-versa boiling occurs in an open container
When the number of molecules in the vapor phase are in equilibrium with the molecules in the liquid phase, this creates a saturated vapor pressure.
Tip: think of hot boiling water in a kettle. As it gets hotter, more gas starts coming up and trying to get the lid off. This means that there are more molecules in the vapor.
Vapor pressure is directly proportional to temperature; increased temperature results in increased vapor pressure BUT also decreased solubility (Henry’s law). Think of des.
Decreased solubility (aka, the Blood:gas coefficient) means more stays in the alveoli (as it is harder to “dissolve” it), and likewise the brain; less is therefore in the blood/circulation.
Interpretation: decreased solubility = faster onset/offset.
This works for offset too because the gas doesn't "want" to be in the blood, so it leaves the tissues and blood quickly to enter the alveoli to be exhaled (remember there is still a concentration gradient and redistribution effect to pull it away from the brain). There is no large "reservoir" of gas dissolved in the blood to slow down the process.
Special note: low CO = faster onset/offset and high CO = slower onset/offset; even though this might sound counterintuitive, having more blood flow also means that more volatile anesthetic is carried away from the alveoli.
This affects more soluble agents (especially iso) the most. With a right to left cardiac shunt though, des/N2O are more impacted by the dilutional effect (more on this below).
Partition coefficients are the anesthetic solubility in blood and tissues; the affinity of an anesthetic for one particular tissue versus another.
How easily a solute will dissolve into a solvent depends on the physiochemical characteristics of both compounds; aka, like likes like! OR, another way to say this is it is a measure of how a gas distributes between two compartments at equilibrium.
1) Blood:gas partition coefficient — this is 1 unit of anesthetic in the blood:x unit of anesthetic in the alveoli. Remember that the concentration in alveoli = concentration in the brain, so you want it to STAY in the alveoli. Therefore, a smaller number means that the speed of onset/offset will be faster.
2) Oil:gas partition coefficient — remember that the anesthetic needs to cross the BBB to actually reach the brain. Therefore, a larger number means higher lipid solubility, better efficiency to access the brain, and overall increased potency.
In modern anesthetics, a lower blood:gas partition coefficient is preferred (aka, a faster onset/offset) instead of increased potency.
Partial Pressures
Dalton’s Law: Total gas pressure is equal to the sum of partial pressures in a container (Pt = P1 + P2 + etc)
Another important calculation: partial pressure = (atm pressure) x (volume %)
Importance: Depth of anesthesia depends on partial pressure of the anesthetic in the brain, not the volume percent on dial.
Remember that there are two different types of vaporizers: sevo and iso use a concentration-calibrated variable bypass vaporizer (an “open system” that automatically adjusts to the atmospheric level), while des requires a special electronic/vaporized “Tec 6” vaporizer (a “closed" system” that is based off the % dialed and NOT the atmospheric pressure).
Thus, a Tec 6 vaporizer may lead to underdosing, as higher altitude = decreased partial pressure (which would decrease the concentration in the brain). You would have to increase your dial to compensate for this. You can think of O2 to help remember this — no matter your altitude, there is always 21% O2 in the air; but their is altitude sickness because the partial pressure of O2 decreases and it is harder to get O2 into your lungs.
EX calculations:
6% Desflurane at sea level results in a delivered partial pressure of 45.6 mmHg (0.06 X 760 mmHg = 45.6 mmHg)
6% Desflurane in Denver (1 mile above sea level) results in a delivered partial pressure of 37.2 mmHg. (0.06 X 620 mmHg = 37.2 mmHg)
Based on a similar principles, administering des in a vaporizer intended for sevo, iso, or halothane would be catastrophic (a massive delivery of des with possible hypoxia); while a halothane vaporizer could theoretically be used to deliver iso (as the partial pressure are nearly identical; also similar to sevo and enflurane, an even older agent than halothane)
HLH Rule: Placing an agent with a high vapor pressure into a vaporizer designed for an agent with a low vapor pressure will result in too high of a dose/output compared to what you set the dial at.
LHL Rule: Placing an agent with a low vapor pressure into a vaporizer designed for an agent with a high vapor pressure will result in too low of a dose/output compared to what you set the dial at.
Minimum Alveolar Concentration (MAC)
1 MAC definition: when 50% patients will not respond to a NORMAL painful stimuli; Measure of potency, akin to ED50 (which is used for IV drugs)
higher MAC = less potent drug (inverse relationship)
Site of Action: MAC is mediated principally by the spinal cord, not the brain
Remember, MAC is a reference point, not a prescription; each patient’s needs will vary on a spectrum. It allows for comparison of potency between different volatile agents.
MAC exhibits additivity: 0.5 MAC Isoflurane + 0.5 MAC N2O = 1 MAC.
Variations:
MAC Awake: when 50% patients will respond to a verbal command to open their eyes (approx. 1/3 MAC). This is also the same MAC level where recall/awareness is lost. However, MAC-memory is much lower than MAC-Awake.
MAC BAR: when 50% patients will have blocked adrenergic response to an INCISION, e.g., tachycardia or HTN (approx. 1.5x MAC).
Factors affecting MAC:
Decreases (aka, you need less of an anesthetic): Elderly (decreases by 6% per decade after 40 y.o.), hypothermia, hyponatremia, acute ethanol use, metabolic acidosis, decreased CNS neurotransmitters (benzodiazepines, barbiturates, ketamine, opioids, lithium, lidocaine, hydroxyzine, alpha2 agonists, N2O, IV anesthetics), hypotension, hypoxia, anemia, CABG, pregnancy to 24-72 hours postpartem
Increases (aka, you need more of an anesthetic): Youth (peaked for infants 1-6 months, although sevo is the same for neonates vs infants), red hair (due to pheomelanin production), hyperthermia, smoking, hypernatremia, chronic ethanol use (think of tolerance), increased CNS neurotransmitters (acute amphetamine intoxication, acute cocaine intoxication, levodopa, ephedrine, MAOIs)
No effect: HTN, gender, obesity (Obesity does not slow down the rate of onset, but it can cause delayed emergence due to storage of the inhaled anesthetic in adipose tissue), potassium/magnesium abnormalities, thyroid dysfunction
Even though thyroid function ITSELF does not affect MAC, profound hypothyroidism can depress CO, which then leads to faster induction (or vice versa)
Children have faster onset/emergence rate at the same anesthetic gas concentrations as adults because:
higher minute ventilation per smaller area (the “ventilation effect”); the FRC is a smaller size compared to adults, meaning more “fresh” gas exchange can occur.
There is also a preferential distribution of their INCREASED (compared to adults) CO to vessel-rich groups (like the brain), which overrides the "dilution" effect of the high cardiac output.
Temperature is based on Henry’s Law, which states that (Gas solubility) = (partial pressure of the gas) x (gas constant). Thus, temperature has an inverse relationship to solubility (opposite of how typically material dissolves into liquids)
FA/FI Curves
Focus on achieving anesthesia by raising partial pressure in the brain/spinal cord; this can be done by focusing on two locations — what is leaving the vaporizer (Fi, vapor concentration) and what is inside the alveoli (Fa, exhaled vapor concentration).
Remember that alveolar concentration = brain concentration; we cannot measure brain concentration, so we use alveolar concentration as a surrogate value
Pathway: Fi —> Fa —> buildup of anesthetic partial pressure inside the alveoli is being opposed by continuous uptake of agent in blood —> CO distributes the anesthetic agent throughout the body to tissues
Referring back to what has already been said above:
low solubility = less uptake into the blood = increased rate of rise = faster equilibration of Fa/Fi = faster onset
high solubility = more uptake into the blood = decreased rate of rise = slower equilibration of Fa/Fi = slower onset
Always remember that the partial pressure of an anesthetic agent in the blood is what equates to a greater effect, not the amount of anesthetic actually dissolved in the blood. SUMMARY: Fa/Fi basically equates to speed of onset.
Understanding the chart:
From top to bottom, this would be: N2O > des > sevo > iso. Uptake is the most important factor in the rate of rise of Fa:Fi, NOT distribution or overpressurization.
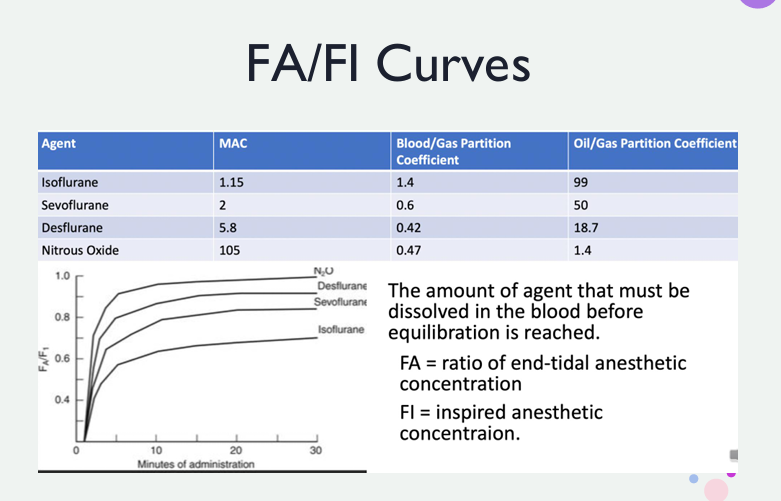
FA factors:
FI, aka rate of delivery to the alveoli (“washin”): fresh gas flow, setting of the vaporizer, time constant of the delivery system, anatomic dead space, alveolar ventilation, FRC
Rate of removal from alveoli (uptake): anesthetic solubility, CO, partial pressure gradient between the alveolar gas and mixed venous blood
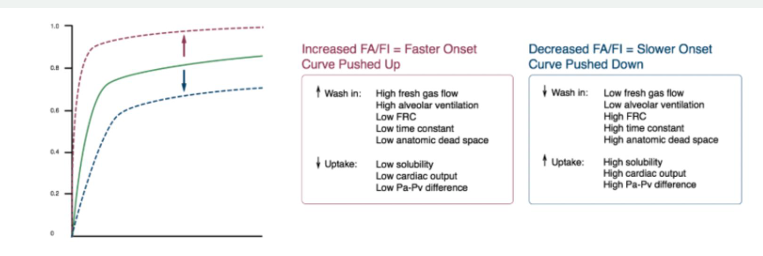
1) For the Fa/Fi to increase (curve pushed up), there must be greater wash in (high FGF, high alveolar ventilation, low FRC [think about wanting to maximize the effective lung space], low time constant, low anatomic dead space) or reduced uptake (low solubility, low CO, low Pa-Pv difference) to keep everything in lungs as much as possible
2) For the Fa/Fi to decrease (curve pushed down), there must be reduced wash in (low FGF, low alveolar ventilation or spontaneous ventilation, high FRC, high time constant, high anatomic dead space) or increased uptake (high solubility, high CO, high Pa-Pv difference)
Tip: As the patient becomes more anesthetized, alveolar ventilation decreases, which decreases the amount of anesthetic they inhale. The use of mechanical ventilation bypasses this feedback loop.
Cardiac Shunts and FA/FI
Right to left shunt: because some of the deoxygenated blood leaving the right heart bypasses the lungs, it does not pick up the inhalation agent or oxygen. This leads to dilution when it mixes in the left heart. PaO2 and partial pressure of anesthetic in arterial blood is diluted. SUMMARY: It essentially creates a bypass where a portion of the blood never "sees" the anesthetic gas in the lungs
EX of right to left shunt: tetralogy of fallot, foramen ovale, Eisenmenger’s syndrome, tricuspid atresia, Ebstein’s anomaly
Agents with lower solubility are more influenced due to undergoing very little uptake by the blood and the effect of cardiac dilution is unchecked when grabbed directly from the lungs. EX: des is affected the most by right to left cardiac shunts.
More soluble agents experience a greater degree of uptake by the blood, which partially offsets the dilution effect. EX: iso is affected the least by right to left cardiac shunts.
Left to right shunt: Minimal effect on anesthetic uptake or induction time.
In the context of anesthesia, endobronchial intubation (accidentally intubating the right or left mainstem bronchus) acts physiologically as an intrapulmonary right-to-left shunt. This means it also slows onset/offset.
**If you are giving an IV drug (like Propofol), a right-to-left shunt actually speeds up its onset of action for the brain. Also, a left-to-right shunt for IV drugs would SLOW DOWN its speed.
Distribution of Inhaled Anesthetics
After entering the blood, anesthetics distribute to nearly all the tissues in the body; based on blood flow, solubility, and partial pressure gradients.
Determinants of tissue uptake: tissue:blood partition coefficient, tissue blood flow, partial pressure difference between arterial blood and tissue
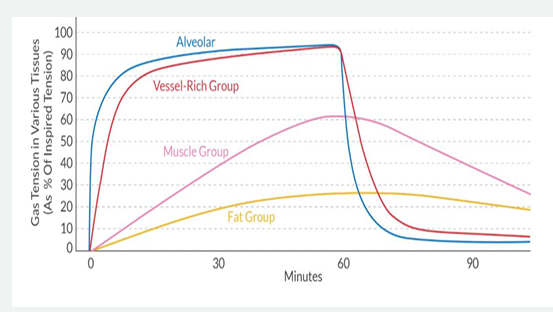
Tissue:Blood partition coefficient: determines the time of equilibration of inhaled anesthetic between the tissue and alveoli. The body is divided into 4 Tissue Groups, based on how much CO it receives relative to its percentage in body weight.
Order of what tissue groups equilibrate first to last with Fa: Vessel-rich group —> muscle group —> fat group —> vessel-poor group.
Vessel-rich group (brain, heart, kidney, liver): 75% CO, 10% body mass
Complete equilibration between the alveoli and brain may be achieved as quickly as 6-10 minutes
Muscle group: 20% CO, 50% body mass
Fat group: 5% CO, 20% body mass
Tip: think of the vessel-poor group as a high capacity sink capable of storing large amounts of agent
Vessel-poor group (bones, tendons, cartilage): <1% CO, 20% body mass
A higher tissue:blood partition coefficient = higher tissue solubility (and faster onset/offset; think of it bringing the gas in the blood into tissues faster, so it spends less time in the blood)
overpressurization: the higher the concentration of anesthetic delivered to the alveolus, the faster the onset.
Example: If you want a patient to reach 1 MAC of Sevoflurane (roughly 2%), you might set the vaporizer to 4% or 6% initially. This "overpressurizes" the circuit to overcome the initial uptake by the blood and vessel-rich group, reaching the target FA much faster.
Other Effects Specific to N2O
Concentration Effect: Only clinically relevant to N2O and on induction (AKA, why you have to do just turn of the anesthetic gas and turn up FGF to speed up emergence). This explains why, even though N2O has a higher blood:gas solubility, it is actually faster onset/offset (on the topmost part of the Fa:Fi curve), instead of des; there is a faster equilibration with the Fa. It refers to Nitrous oxide ITSELF.
Step 1) ConcentratING effect: when a patient is breathing at room air, nitrogen is the primary gas in the alveolus. N2O, however, is 34x more soluble in the blood than nitrogen. Thus, when N2O is introduced into the lung, the volume of N2O going from the alveolus to the blood is much higher than the amount of nitrogen moving in the opposite effect. This then causes the alveolus to shrink in size, creating a relative increase (“concentrating”) in the Fa.
Step 2) Augmented gas flow: As the concentrating effect temporarily reduces alveolar volume, this creates a temporary negative pressure (vacuum), which physically pulls more fresh gas from the breathing circuit into the lungs. This increases alveolar ventilation, augmenting Fa; this is only a temporary phenomenon, though, as alveolar volume is restored quickly.
Second Gas Effect: when N2O and a second gas are introduced into the alveolus, the rapid uptake of N2O will cause the alveolus to temporarily shrink. This reduction in alveolar volume and augmented tracheal inflow cause a relative increase in concentration of the second gas. The partial pressure of oxygen also increases when the alveolus shrink (transient effect). End result: concentration of the other gases is higher than if they were administered alone to make faster onset/offset.
The Second Gas Effect is essentially the Concentration Effect applied to a second volatile agent (like Sevoflurane or Isoflurane) that is being administered simultaneously with N2O
It is a stronger effect for gases with high blood:gas solubility
Diffusion hypoxia: gas containing areas of the body can absorb up to 30 L of N2O within 2 hours; most of it is eliminated within 5 minutes after N2O is discontinued. As this tremendous amount of N2O is transferred back into the alveolus, it dilutes alveolar oxygen/CO2 and causes temporary diffusion hypoxia and hypocarbia.
Prevention: Administer 100% Fio2 for 3-5 minutes after N2O has been discontinued. New evidence suggest a lower Fio2 during emergences may reduce absorption atelectasis and improve postop gas exchange.
Gas Metabolism
Elimination Mechanisms:
Primary mechanism: alveolar elimination.
Secondary: hepatic biotransformation.
Rule of 2’s:
Des: 0.02% metabolites created
Because it is such a low hepatic metabolism, it has the greatest lung elimination
Iso: 0.2% metabolites created
Sevo: 2-5% metabolites created
Halothane: 20% metabolites created
**N2O is basically 0% (0.004%)
Minimal (not clinically significant): percutaneous loss.
Halogenated anesthetics undergo metabolism by the P450 system, primarily the CYP2E1 enzyme (think of counting backwards during a traditional mask intubation where you count backwards but just switch the 3 and 2). During induction, metabolism does not significantly counter the rise in Fa; it plays a more important role during recovery.
A big reason for why halothane is not used anymore is the 20% hepatic biotransformation that creates trifluoroacetic acid (TFA) metabolites and is the primary mechanism for immune-mediated (IgG) halothane hepatitis.
Des and iso may produce a minimal amount of TFA and fluoride ions. More of a possible concern in patients with previous TFA exposure (e.g., history of halothane sensitivity). Thus, if someone previously had a history of halothane hepatitis, it is best to give them sevo.
Sevo is not metabolized into TFA, but releases a larger amount of fluoride ions that theoretically cause high output renal failure, which is unresponsive to vaso.
Soda Lime Contribution to Gas Breakdown:
Desiccated (dried out) soda lime + des or iso can produce carbon monoxide. Des > iso if using baralyme (a CO2 absorbent not made in the US, which contains a strong base)
Desiccated OR hydrated soda lime + sevo can produce Compound A. In rat studies, Compound A has been shown to be nephrotoxic. This is more likely when sevo is administered with low flow anesthesia, also putting the patient at risk for acute kidney damage. Prevention: if using baralyme (a CO2 absorbent not made in the US, which contains a strong base), then FGF must be >2 L/min.
The FDA recommends a minimum FGF of 1 L/min for up to 2 MAC hours and 2 L/min after 2 MAC hours.
Tip: remember that Compound A and CO are produced OUTSIDE of the body, while TFA/free fluorides are produced IN the body
CNS Effects of Inhaled Anesthetics
CMRO2 (which is normally 60% dependent on electric activity and 40% on cellular hemostasis) is decreased in a dose dependent manner, but only to the extent that volatile anesthetics reduce the electrical activity. Once the brain is isoelectric, volatile anesthetics cannot reduce CMRO2 further.
1.5-2 MAC is required to produce an isoelectric state; if enough volatile gas is given to someone to get to this point, CMRO2 decreases by 60%
Only N2O cannot produce an isoelectric state
Sevo in high concentrations (2 MAC) can produce seizure activity, especially more common in pediatrics and when rapidly increased titration. Further exacerbated by hypocapnia.
CBF: the brain matches its blood flow with its metabolic requirement; when the metabolic demand increases, blood vessels dilate (aka, cerebral vascular resistance decreases), while when the metabolic demand decreases, blood vessels constrict (aka, cerebral vascular resistance increases). This is known as coupling, or cerebral autoregulation.
Volatile anesthetics are cerebral vasodilators. This leads to two competing factors: vasoconstriction from the reduction in CMRO2 and direct vasodilation from the anesthetic agent (known as decoupling, or decreased cerebral autoregulation)
At concentrations > 0.5 MAC, these gases uncouple CMRO2 and CBF (aka, increase CBF even though they decrease CMRO2). Although this creates a favorable cerebral oxygen supply-demand ratio, it also increases ICP. Mild hyperventilation and/or concurrent administration of propofol, opioids, or barbiturates will partially offset the vasodilator effects.
N2O is different as it increases both CMRO2 and CBF.
Cerebral Autoregulation: CBF becomes increasingly dependent on systemic blood pressure as the concentration of volatile anesthetic is increased.
Normally, cerebral vasculature continuously adjusts vessel diameter to maintain a constant CBF between cerebral perfusion pressures of 50-150 mmHg. This is done by vasoconstriction in hypocapnic conditions (alkalosis) and vasodilation in hypercapnic conditions (acidosis).
While anesthetics disrupt this autoregulation, very mild hyperventilation can effectively prevent the increase in CBF.
CSF Volume: Although volatile anesthetics affect CSF dynamics (e.g., increasing/decreasing CSF production/absorption), it is greatly overshadowed by their ability to increase CBF
Evoked potential monitoring: evoked potentials are used to monitor the integrity of the neural pathway. The 4 types of evoked potentials:
1) Somatosensory (SSEP): Monitor the integrity of the dorsal column, specifically the medial lemniscus. This region is perfused by the posterior spinal arteries.
2) Motor (MEP): Monitors the integrity of the corticospinal tract. This region is perfused by the anterior spinal arteries.
3) Visual (VEP)
4) Brainstem auditory (BAEP)
The most important compartments of the evoked potential waveform is the amplitude (strength of the nerve response) and latency (speed of nerve conduction)
Losing the evoked potential signal or having a diminished response suggests ischemia to the pathway being monitored. Dx: When amplitude decreases by 50% or greater and/or latency increases by 10% or greater
Volatile agents automatically decrease amplitude and increase latency; this is further compounded by combining the volatile gas with N2O, hypoxia, hypercarbia, and hypothermia.
Interpretation: The best technique to do when evoked potentials are monitored is TIVA without N2O. If you do use a volatile anesthetic, use <0.5 MAC and supplement with propofol or opioids.
Do not use muscle relaxants with MEPS outside of a small induction dose for intubation (you want to assess the motor pathways, so you do not want to block them)
Ketamine enhances the signal.
CV Effects of Inhaled Anesthetics
Modulated nitric oxide release
inhibited Ach-induced vasodilation
impaired sodium/calcium pump (SO DECREASED LEVELS OF SODIUM AND CALCIUM, NO DEPOLARIZATION OCCURS)
Primary cause of decreased MAP: decreased calcium in vascular smooth muscle = systemic vasodilation = decreased SVR/venous return
Secondary cause of decreased MAP: decreased calcium in the myocyte = myocardial depression = decreased inotropy
N2O actually increases blood pressure due to SNS activation; N2O + a volatile anesthetic causes less blood pressure reduction when compared to the MAC equivalent of the volatile agent alone
Volatile agents directly affect cardiac conduction:
decreased SA node automaticity
Decreased conduction velocity through the AV node, His-Purkinje system, and ventricular pathways
Increased duration of myocardial repolarization by impairing the outward potassium current; this increases the AP duration, which prolongs the QT interval (an easier way to think about this is the decreased calcium levels = prolonged QT interval)
Altered baroreceptor function
**Rapid dose increases in des, and to a lesser degree iso, can actually cause tachycardia by 5-10%. This is actually due to pulmonary irritation causing SNS activation.
Pulmonary irritation —> SNS activation —> norepinephrine release —> beta1 stimulation
Tachycardia may be minimized but not abolished through addition of opioids, alpha2 agonists, or beta1 antagonists
N2O actually stimulates the SNS and increases HR
There is a small decrease in baseline contractility, however the myocardium remains preload responsive, with myocardial depression being dose dependent. Some overall decrease in CO?
Coronary vascular resistance: there is greater increase in coronary blood flow compared to myocardial oxygen demand. This is done by preferentially dilating the small cardiac blood vessels that are 20-30 micrometers in diameter
Potency of coronary artery vasodilation (greatest to least): iso —> des —> sevo
As iso is the most potent vasodilator of these, there are possible concerns of coronary steal. Normally, as myocardial oxygen demand increases, healthy vessels dilate. REMEMBER: the heart’s oxygen extraction rate is 75%; since it cannot significantly increase its extraction rate anymore, it increases its own blood flow to satisfy its oxygen requirement. Severely stenotic vessels are maximally dilated beyond the point of stenosis, though; this means that when myocardial oxygen demand increases, the already dilated vessels will not be able to dilate further. Thus, coronary blood flow would only be directed to the healthy tissue; basically, at the expense of the poor tissue.
This is also known as “Reverse robin hood” effect, as the blood takes the path of least resistance
The "cardioprotective" reputation of volatile gases comes from Ischemic Preconditioning; by triggering molecular pathways that "prime" the myocardium to survive periods of ischemia through the activation of adenosine receptors and ATP-sensitive potassium channels. This keeps the channels closed during stress, reducing cell death and protecting the heart during an MI or cross-clamping in cardiac surgery
Iso is the best for cardioprotection, due to also having a unique mild beta-adrenergic agonist profile that helps maintain CO by increasing heart rate and decreasing SVR
Sevo has the least effects on heart rate
Pulmonary Effects of Inhaled Anesthetics
the central chemoreceptor in the medulla maintains tight control of PaCO2; every 1 mmHg increase in PaCO2 above baseline should increase minute ventilation by 3 L/min. However, volatile agents cause a dose dependent depression of the central chemoreceptor and the respiratory muscles, causing hypercarbia.
Remember: Normal minute ventilation is 5-8 L/min
MV = Vt x RR —> Vt is around 500 ml (6-8 mL/kg), while RR is 12-16 breaths/minute (every 4-6 seconds)
MOA:
1) Altering the respiratory pattern: decreased tidal volume with partial compensation through increased resp rate, although not enough to prevent a rise in PaCO2. Overall effect: rapid, shallow breathing. This also then causes increased dead space ventilation.
2) Impairing the response to CO2: The CO2 response curve represent the sensitivity of the entire respiratory system to PaCO2.
Rightward and down shift: decreased response to CO2 (aka, you have an increased apneic threshold, which means you need a higher PaCO2 level before your lungs are stimulated to want to breath). Summary: there is a decreased ventilatory response to changes in paCO2. This creates acidosis; minute ventilation would be less than predicted.
Causes: general anesthetics, opioids
Leftward shift: increased response to CO2. Creates alkalosis; minute ventilation would be more than predicted.
*These acid base disturbances are effects, not causes of the shift in the curves! Instead, these graphs represent effects of medications.
Additional resp effects:
Impaired motor neuron output and muscle tone to upper airways: impaired airway dilator muscles (e.g., genioglossus and tensor palatine) lead to upper airway obstruction, while impaired pulmonary muscles decreases FRC and effectiveness of ventilation
Although there is some increased airway resistance, thus causing depressed ventilations, it is overcome with the potent bronchodilation effects (sevo > others)
Peripheral chemoreceptors: located in the carotid and aortic bodies monitor for hypoxemia, they regulate hypoxic ventilatory response. A PaO2 <60 mmHg is a stimulus to increase minute ventilation to restore arterial oxygenation.
Carotid bodies to relay afferent input to the respiratory center via the glossopharyngeal (CN IX); they are more sensitive to change in arterial gas tensions (PaO2 and PaCO2) and Hydrogen ions.
Aortic bodies relay afferent input to the Vagus nerve (CN X); they are more sensitive to changes in blood pressure.
Volatile gases impair the peripheral chemoreceptors for up to several hours after anesthesia. The impaired response to acute hypoxia (O2) from these chemoreceptors occurs at doses as low as 0.1 MAC, but the response to hypercarbia (CO2) via the medulla/central chemoreceptors is still intact (it needs a much higher MAC to turn off this response)
Proposed MOA: The glomus type I cells in the carotid bodies provide the sensory arm of the hypoxic drive and it is hypothesized that volatile anesthetics create a reactive oxygen species (ROS) that impairs the glomus type I cells.
As anesthetic metabolism is the source of the ROS, the agents that undergo the greatest amount of biotransformation in the body inhibit hypoxic drive the most (from most to least inhibiting: sevo > iso > des)
This is mostly important for patients who rely on a hypoxic drive to breathe (and not the CO2 response), such as emphysema and sleep apnea. Des is the best agent for them.
Summary: Remember that central chemoreceptors are for your CO2 drive, while peripheral chemoreceptors are for your hypoxic drive

Gas Chemistry
There are 3 categories of anesthetic gases:
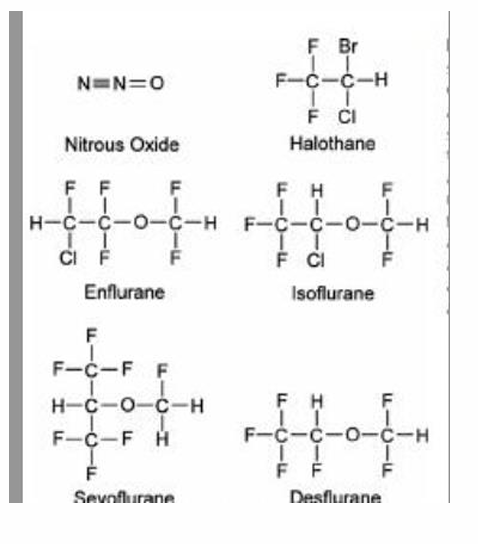
Ethers (R-O-R): des, iso, sevo
Tip: R-O-R is just a skeleton template; you will actually see C-O-C
sevo is a methyl isopropyl ether, while des and sevo are methyl ether ethers.
Alkanes (R-H): halothane, chloroform
Tip: R-H is just a skeleton template; you will actually see C-C
Inorganic gases: N2O
*At room temp and atmospheric pressure, ethers and alkanes exist as liquids.
TIP: local anesthetics are amides or ESTERS
Amides have two i’s in their name: less risk for causing allergies, and have a longer duration of action.
EX: lidocaine, bupivacaine, mepivacaine
ESTERS have one i in their name: higher chance of causing an allergic reaction, and a shorter duration of action.
EX: cocaine, procaine, benzocaine
Halogenation: this is the addition of an inert halogen (e.g., fluorine, chlorine, or bromine) to a chemical structure; halogenation by itself actually increases risk of cardiac dysrhythmias (EX: halothane and iso), although it originally was done to reduce flammability and increase potency.
Modern anesthesia has moved to from general halogenation to fluoridation (fluoride is the smallest of the halogens and keeps tight control of its electrons). This not only reduces flammability, but also minimizes hepatic metabolism and decreases solubility (aka, faster onset/offset).
Chiral carbons: This explains why two drugs with the exact same chemical formula can have completely different effects on different patients. A chiral carbon is a carbon atom that is bonded to four different atoms.
Sevo is the only one that does not contain a chiral carbon
Gas Advantages/Disadvantages
Desirable characteristics: environmentally friendly, no PONV, cheap, not pungent, no airway irritation, stable hemodynamics, fast onset/offset
Iso:
2x as potent as sevo and 5x as potent as des
Chemical structure: 1-chloro 2,2,2,-trifluoroethyl difluoromethyl ether
Vaporizer color: purple
Chemical structure: five fluorides + a chloride
Tip: iso reminds me of a pentagon in math for some reason, so 5 sides
partial pressure: 238 (very similar to halothane)
MAC: 1.15%
Solubility (B:G Partition coefficient): 1.4 (slowest)
Oil:Gas Partition Coefficient: 100 (highest, so the most potent)
Advantages:
Cardioprotective (through Ischemic Preconditioning, “The Protective Effect” by pre-stimulating potassium pumps and mimicking the body's natural defense against ischemia)
small amount of muscle relaxation
muscle relaxation from volatile agents can also be the reason for upper airway obstruction
minimal biotransformation; no significant system toxicity
cheap
Disadvantages:
slow onset/offset
pungent odor (not good for an inhaled induction); airway irritant
potentially coronary steal
Des:
it is nearly identical to isofluorine, except that the chlorine has been replaced with a fluorine atom (it is “fully fluorinated”)
The changes that come with full fluorination: decreased potency, increased vapor pressure (aka, decreased intermolecular attraction so requires a heated vaporizer), and increased resistance to biotransformation (less likely to create TFA), decreased n/v, faster onset/offset
Chemical structure: difluoromethyl 1,2,2,2,-tetralfuoromethyl ether.
Vaporizer color: blue
Chemical structure: six fluorides
Tip: des has 3 letters in it just like six does
partial pressure: 669 (the highest, requiring a special vaporizer due to the lowest boiling point of 22.8 C; the vaporizer is thus 39 C and pressurized to 2 atm)
Remember that boiling point is when vapor pressure = atmospheric pressure)
MAC: 6%
Solubility (B:G Partition coefficient): 0.42 (would be the fastest, but N2O is actually the fastest)
Oil:Gas Partition Coefficient: 20
Advantages:
rapid onset/offset
strongest muscle relaxation effects
minimum metabolism
minimal n/v
Disadvantages:
Causes airway irritation; most pungest of all the gases (do not use for inhalation induction; it should be reserved for maintenance dossing only)
S/s if you WILL use for mask induction: excess salivation, laryngospasm, breath holding (to avoid inhaling this acidic gas)
Can even cause SNS activation d/t amount of irritability
Highest association of creating carbon monoxide with desiccated soda lime compared to sevo or iso
expensive d/t Tec 6 vaporizer (d/t lowest boiling point)
negative climate/environmental impact (compared to sevo and iso)
Sevo:
Even though sevo is heavily fluorinated, it is still 3x as potent as des; likely due to the bulky propyl side chain
Chemical structure: fluoromethyl 2, 2, 2-trifluoro-1(trifluoromethyl) ethyl ether
Vaporizer color: yellow
Chemical structure: seven fluorides
Tip: “sev” for seven
partial pressure: 157 mmHg
MAC: 2%
Solubility (B:G Partition coefficient): 0.6
Oil:Gas Partition Coefficient: 50
Advantages:
fast onset/offset
nonpungent (excellent for inhaled induction; smells sweet like bananas, so preferred in pediatric cases)
possible neuro/cardiac protection
best environmentally protective volatile anesthetic
cheapish
high level of muscle relaxation
minimal n/v
most bronchodilation
Disadvantages:
more expensive than iso
reacts with soda lime to create Compound A (nephrotoxic)
seizures in children (at high doses)
N2O:
Good analgesic, but poor anesthetic by itself (used as an adjunct to volatile gases)
Comes from pipeline supply.
Chemical structure: two nitrogens
partial pressure: 38000
MAC: 104%
Solubility (B:G Partition coefficient): 0.47 (actually has faster onset/offset than des in real life)
Oil:Gas Partition Coefficient: 2 (least potent)
Advantages:
rapid uptake/elimination
virtually no biotransformation/toxicity
little cardiac/respiratory depression; instead increases
Only completely nonpungent/odorless gas, others have at least some sort of smell
reduces MAC requirements
70/30 concentration N2O:O2 is equivalent to 10 mg morphine
no risk of MH
Disadvantages:
expansion of closed air spaces (do not give if the patient has a pneumothorax, dilated bowels, eye surgery, laparoscopic surgery/pneumoperitoneum). This again goes back to how N2O is 34x more soluble than nitrogen (for every 2 molecules of nitrogen that leaves a closed space, 34 molecules of N2O take its place). Normally, a compliant airspace would have increased volume but the pressure would be unchanged (self-limiting). In noncompliant airspace would have no change in volume but increased pressure, risking the following:
1) N2O can increase volume in fixed spaces (e.g., fast equilibrium between space and the blood for pneumothorax, air embolism, pulmonary blebs, and ocular gas bubble placement; slow equilibrium between space and the blood for bowels). EX: During retinal detachment surgery, a gas bubble is placed over the retinal break to act as a splint to hold the retina in place while healing occurs. N2O can expand the SF6 bubble, compromising retinal perfusion and causing permanent blindness. Guidelines:
discontinue N2O 15 minutes before the SF6 bubble is placed; avoid N2O 7-10 days after the SF6 bubble is placed
if medical air is used, avoid N2O for 5 days after
if perfluouropropane is used, avoid N2O for 30 days after
Silicone oil has no interaction with N2O
2) N2O can increase pressure in fixed air spaces (e.g., middle ear and brain during intracranial procedures). EX: avoid for ENT surgeries, as N2O during the procedure can damage a tympanic membrane graft; avoid quickly discontinuing it as it can result in serous otitis. Likewise, N2O can increase the volume and pressure of the ETT, LMA, and balloon tipped PA catheter. This is why they need to be frequently checked on
requires a high concentration; amount of oxygen delivered must be reduced
diffusion hypoxia
negative climate/environmental impact
PONV
nonflammamble, but does support combustion (especially with electrocautery)
immune suppression
potential teratogenicity/spontaneous abortion (many healthcare providers avoid N2O during the first two trimesters of pregnancy
Irreversibly oxidizes/inhibits Cobalt in Vitamin B12, which then inhibits methionine synthase (an enzyme required for folate metabolism and myelin production)
Risk factors: pre-existing Vitamin B12 deficiency, pernicious anemia, alcoholism, strict vegan diet, pregnancy, and recreational use of N2O
S/s: megaloblastic anemia (due to bone marrow suppression), neuropathy, impaired DNA synthesis)