bonding, structure and the properties of matter
Chemical Bonds
compounds = substance in which two or more elements are chemically bonded
ionic = particles are oppositely charged ions; electrostatic force of attraction between metals and non-metals
metallic = particles are atoms which share delocalised electrons; occurs in metallic elements and alloys
Ionic Bonding
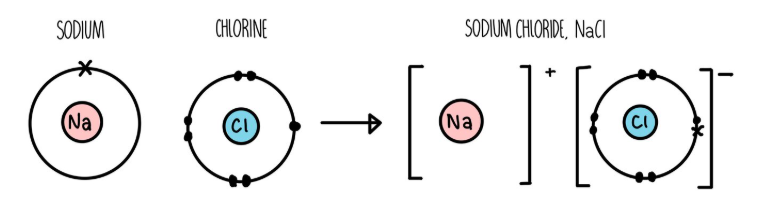
metal + non-metal: electrons in the outer shell of the metal atom are transferred
metal atoms lose electrons to become positively charged ions
non-metal atoms gain electrons to become negatively charged ions
ion = an atom that has lost or gained electrons
ions produced by metals in groups 1 & 2 and ions produced by non-metals in groups 6 & 7 gain full outer shells of electrons so they have the same electronic structure as a group 0 element
Ionic Compounds
a giant structure of ions
held together by strong electrostatic forces of attraction between oppositely charged ions
since the structure is 3D, the forces act in every direction
eg. sodium chloride
Properties
ionic compounds have regular structures (giant ionic lattice) in which there are strong electrostatic forces between oppositely charged ions
they have high MP and BP as a lot of energy is required to break the numerous strong bonds
when melted or dissolved in water, ionic compounds conduct electricity as the ions are charged and free to move so they can carry a current - but they can’t when solid because the ions are fixed
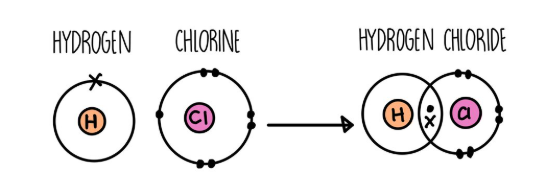
Covalent Bonding
when atoms share one or more pairs of electrons
Covalent Structures
small molecules, such as HCl, H2, O2, Cl2, NH3 have strong covalent bonds within their molecules
Properties: usually gases or liquids with low MP and BP; weak intermolecular forces (increase with size of molecules as they have higher MP and BP); don’t conduct electricity as they don’t have a charge
polymers are large covalently bonded molecules
Properties: have very large molecules; atoms are linked to other by strong covalent bonds; solid at room temp as intermolecular forces are relatively strong
giant covalent structures (macromolecules) consist of many atom covalently bonded in a lattice structure
eg. diamond, graphite and silicon dioxide
Properties: solids with very high MP; all atoms are linked to others by strong covalent bonds which must be overcome to melt or boil the substance
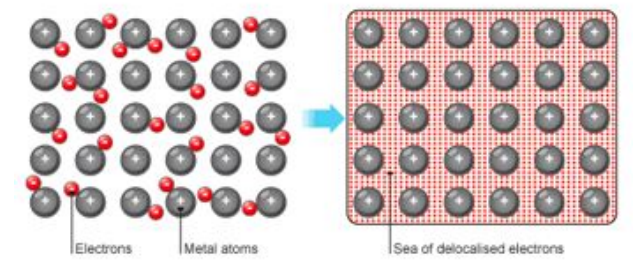
Metallic Bonding
the bonding in a metal consists of positive ions and delocalised electrons arranged in a regular pattern
the delocalised electron system consists of the electrons ‘lost’ from the atoms to form positive ions
delocalised electrons are free to move through the structure
the delocalised electrons are shared through the structure so metallic bonds are strong
Metallic Structures
metals have giant structures of atoms with strong metallic bonding
Properties: most metals have high BP and MP; arranged in layers which can slide over each other so metals are malleable which make them less useful for certain things
alloys are made from 2 or more different types of metals
the different sized atoms distort the layers in the structure so they can’t slide over each other making them harder than pure metal
metal conductors
good conductors as delocalised electrons carry electrical charge through the metal
good conductors of thermal energy because energy is transferred by delocalised electrons
The Three States of Matter
solid, liquid and gas
melting and freezing take place at FP
boiling and condensing take place at the BP
particle theory can help to explain melting, boiling, freezing and condensing
the amount of energy needed to change state from solid to liquid and liquid to gas depends on the strength of the forces between the particles
the nature of the particles involved depends on the type bonding and the structure of the substance
the stronger the forces between particles, the higher the melting and boiling point of the substance
limitations of the particle model include the lack of forces and that all particles are represented as spheres
Structure and Bonding of Carbon
Diamond
in diamond each carbon is joined to 4 other carbons covalently
it’s very hard, has a high MP and does not conduct electricity
Graphite
in graphite each carbon is covalently bonded to 3 other carbons forming layers of hexagonal rings which have no covalent bonds between the layers
the layers can slide over each other due to weak intermolecular forces and no covalent bonds between layers so graphite is soft and slippery
one electron from carbon is delocalised
can conduct electricity because the delocalised electron can move
Graphene and Fullerenes
graphene = single layer of graphite
has properties that make it useful in electronics and composites
very strong because atoms within layers are strongly bonded and elastic because planes of atoms can flex easily without breaking apart
fullerenes = molecules of carbon atoms with hollow shapes (nanoparticles)
based on hexagonal rings of carbon atoms but may also contain ring with five or seven carbon atoms
first fullerene to be discovered was the Buckminsterfullerene - C60 - which is spherical
carbon nanotubes = cylindrical fullerenes
high length to diameter ratio
properties make them useful for nanotechnology, electronics and materials
eg. lubricants, delivering drugs to the body, catalysts, reinforcing materials such as tennis rackets
Nanoscience
Nanoparticles
1 - 100 nanometres across
contain a few hundred atoms
smaller than fine particles which have diameters between 100 and 2500 nm
coarse particles (dust) have diameters between 1×10-5 m and 2.5×10-6 m
as size of cube decreases by a factor of 10, surface area to volume ratio increases by a factor of 10
nanoparticles have different properties to the “bulk” chemical it’s made from because of their high surface area to volume ratio; smaller quantities are needed to be effective than for materials with normal particle sizes
eg. fullerenes have different properties to lumps of carbon
Uses of Nanoparticles
high surface area to volume ratio so good catalysts
highly selective sensors
stronger and lighter building materials
new cosmetics such as suncream and deodorant with no white marks
lubricant coatings for gears or artificial joints as they reduce friction
small electrical circuits for computers
potential disadvantages: concerns about nanoparticles being toxic as they may be able to enter the brain from the bloodstream and cause harm