Organic Chemistry
A Review of General Chemistry
1.2 Structural Theory of Matter
Organic Compounds (derived from living organisms)
Inorganic Compounds (derived from nonliving sources)
Compounds can have the same chemical formula, yet they differ in their interactions
Tetravalent - four bonds
Trivalent - three bonds
Divalent - two bonds
Monovalent - one bonds
Stronger intermolecular forces means higher boiling point
How to draw Constitutional Isomers
Determine Valency: Atoms with highest Valency are connected first
Carbon : Tetravalent
Hydrogen: Monovalent
Oxygen: Divalent
Connect atoms with highest valency, place monovalent atoms at the periphery (at the end)
1.3 Electrons, Bonds, and Lewis Structures
Covalent bond - two atoms sharing a pair of electrons
the force of repulsion between the negatively charged electrons
the force of repulsion between the positively charged electrons
the force of attraction between two oppositely charged electrons
Electrons are capable of moving in a way that minimize repulsive forces and maximize attractive forces
Hydrogen atoms have an intermolecular distance of 0.74 angstroms
Atomic Structure:
Every atom is composed of protons and neutrons.
For a neutral atom, the number of protons is equal to the number of elections
First shell - 2 electrons, Second shell - 8 electrons
Outermost shell - Valence electrons
Drawing Lewis Structures
Octet Rule: Second-Row elements will form the necessary number of bonds in order to gain 7
4 Rules
Draw all individual atoms
Connect atoms that form more than one bond
Connect the hydrogens
Pair unpaired electrons so atom achieves an octet
1.4 Identifying Formal Charges
Formal Charge - Correct number of valence electrons
Extra electrons (negative formal charge)
Too few electrons (positive formal charge)
If you are having trouble paying attention during a long lecture, your levels of acetylcholine (a neurotransmitter) may be to blame.
1.5 Polar Covalent Bonds
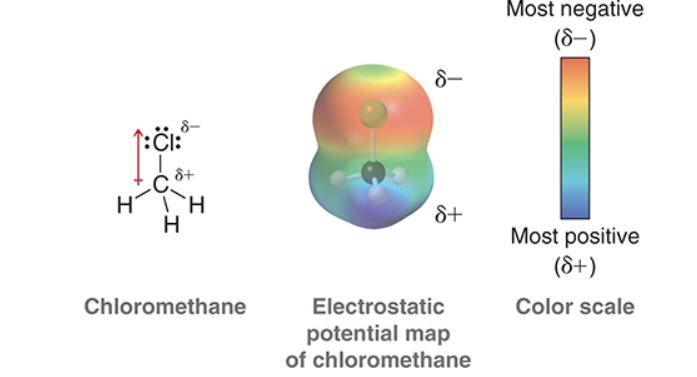
Electronegativity - measure of an atom to attract electrons

If electronegativity is below 0.5, electrons are considered to be equally shared
Examples: C-C and C-H
Covalent
If electronegativity is between 0.5 and 1.7, electrons are considered to be polar.
Examples: O-H and Li-C
If the difference in electronegativity is higher than 1.7, electrons are considered to be ionic pair
Examples: Na-Cl
C-BR and C-I are considered polar covalent
Resulting Partial Charges from Induction
Identify all covalent bonds
Determine direction of each dipole
Indicate the direction of partial charges
Bond Strength increases with more bonds btw
charges are on the element and not on the bond.
the donating atom has a positive charges, and the atom that takes the charge has a negatively charges
Electrostatic Potential Maps
Reading Bond-Line Structures
Bond-line structures - Each corner or endpoint
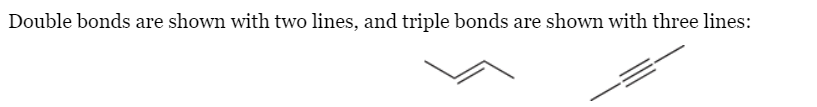
Triple bonds have linear geometry
Hydrogen atoms are not shown because it is assumed that each carbon atom will have enough
1.7 Atomic Orbitals
Electrons were discovered and identified as the source of bonding and Lewis structures were the generally accepted method of mapping electron interactions.
1924: Louis de Broglie - suggested that electrons had the same properties as waves.
Wave Equation - Energy of an atom is “"quantized”, meaning that all electrons are in orbitals (s,p,d,f)
Closest to the nucleus - less energy - less nodes
Farthest from the nucleus - most energy - most nodes
3 rules
Aufau Principle: Lowest Energy Orbital is filled first
Pauli Exclusion Principle: Each orbital can accomodate a maximum of 2 orbitals with opposite spin
Hund’s Rule: One electron is placed BEFORE electrons are paired
Positive charges have empty orbitals, negative charges have positive orbitals
Nodes are the intersections between positively and negatively spun orbital
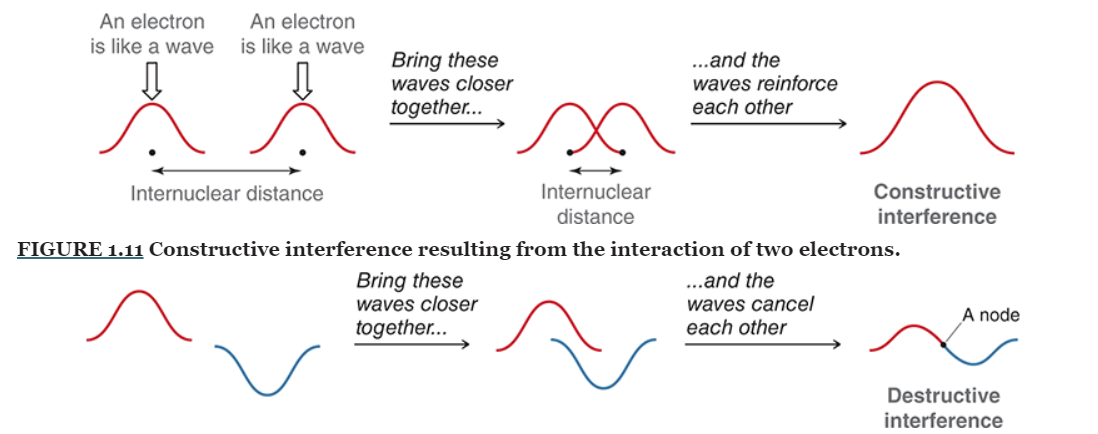
Valence bond theory - sharing electron density
Sigma bonds - Single bonds
Pi bonds - Not sigma bonds
Molecular Orbital theory - implies that orbitals are combined to create new orbitals
An atomic orbital is associated with a singular atom, while a molecular atom is associated with a singular molecule.
Molecule = multiple atoms
Bonding MO: Constructive interference - lower energy
Antibonding MO: destructive interference - higher energy
Smallest Polar molecule - best soluability