5+6. Biomolecules shapes and sizes
There are 3 key factors that determine molecular structure:
Bond length
Bond angle
Bond rotation
Bond length
Atomic bond length is the the distance between the nuclei of 2 covalently linked atoms. It is determined by:
The atomic radii of the atoms joined by the bond, i.e., the chemical identity of the atoms
The type of covalent bond, i.e., single, double, or triple
The length of a single covalent bond is equivalent to the sum of the atomic radii of the 2 bonded atoms
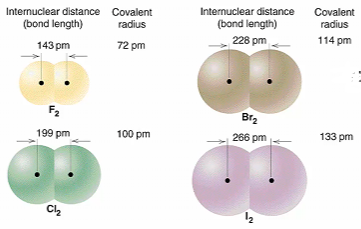
Double and triple bonds have shorter lengths: the increased electron density leads to a greater attractive force on the nuclei of the bonded atoms
Bond rotation
Bonded atoms adopt precise configurations dictated by repulsion of valence electron pairs. Groups of bonded atoms can adopt different orientations relative to each other, as a consequence of bond rotation
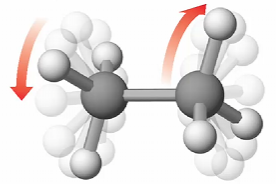
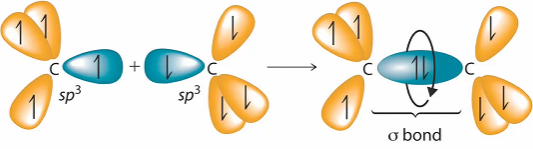
Overlapping sp3 hybrid orbitals on adjacent carbon atoms form a cylindrical symmetrical bond. Because rotation about the bond does not affect the overlap of the bonding orbitals, there is no electronic energy barrier to rotation
Bond rotation cannot occur in double or triple bonds as this would disrupt the p orbital overlap, breaking the bond which is highly unfavourable energetically
Bond angles
Bond angles describes the 3D arrangement of 3 covalently bonded atoms. Atomic bond angles in molecules vary from 60o-1800 depending on the chemical identity of the bonded atoms.
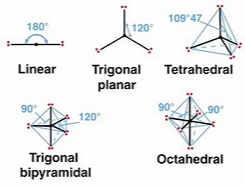
Different groups of atoms arrange themselves in specific orientations, a property that can be described in one of two ways:
Valence shell electron pair repulsion (VSEPR) theory
The hybridisation of atomic orbitals
VSEPR theory
This theory predicts the geometry of molecules based on minimizing the electrostatic repulsion of a molecules’s valence electrons around a central atom
The geometry adopted depends on the number of valence electron pairs
Bonding and non-bonding pairs are involved in repulsion
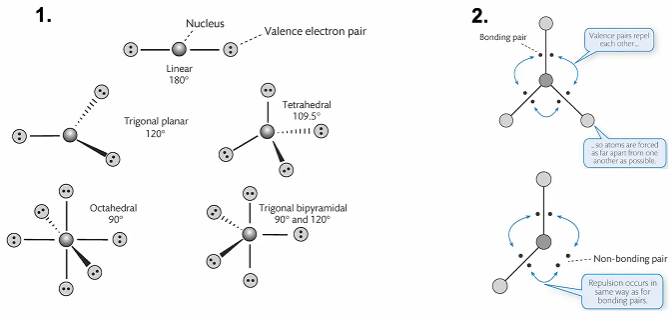
However, VSEPR theory makes the assumption that electrons exist as discrete objects.
Orbital hydridisation
Atomic orbitals do not exhibit the geometries predicted by VSEPR
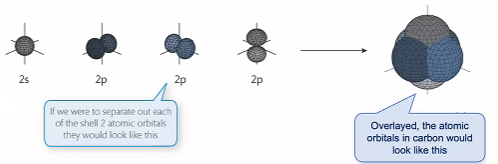
The arrangement of valence electrons as predicted by VSEPR is due to the hybridisation of atomic orbitals which allows the electrons to adopt new geometries
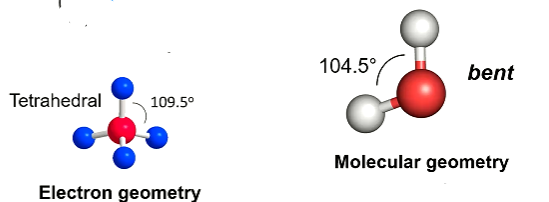
Electron geometry Vs Molecular geometry
When looking at electron geometry, the presence of both bonding pairs and lone pairs are considered, whereas when looking at molecular geometry lone pairs are excluded in deciding the shape of the molecule (however their repulsion is still taken into account for estimating bond angles)
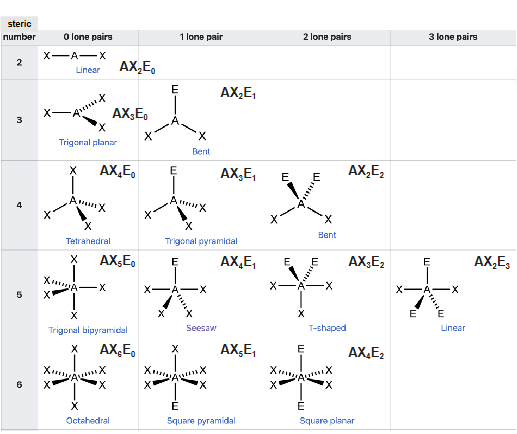
AXE method
The AXE method is used to determine molecular geometry
AXnEm - A: central atom, X: ligand(s) attached to A, E: lone pair(s) on A
Steric number - number of atoms (X) bonded to the central atom (A) and number of lone pairs (E) of valence electrons on A
Isomerism
The existence of different forms of molecules with the same chemical composition but with a different structure or configuration and generally with different physical and chemical properties
Constitutional isomers
Molecules that have the same molecular formula but different connectivities. There are 3 types:
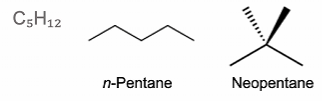
Chain isomerism - arise as a consequence of chain branching
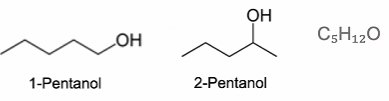
Regioisomerism - arise due to re-positioning of functional groups
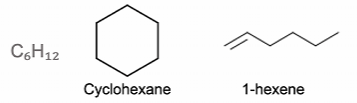
Functional group isomerism - isomers containing different functional groups
Stereoisomers
Molecules that have the same molecular formula and sequence of bonded atoms (constitution), but differ only in the three-dimensional orientations of their atoms in space
Stereocentre: An atom with 3 or more attachments, where swapping 2 of these attachments leads to another stereoisomer
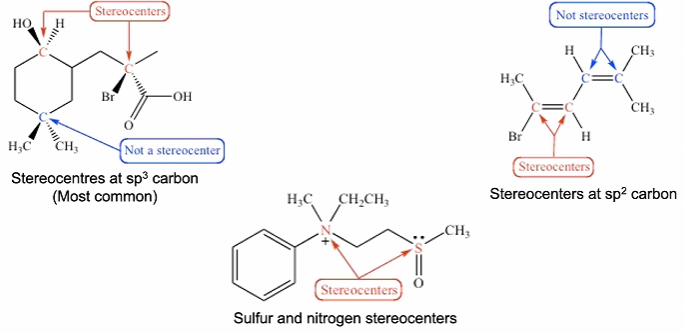
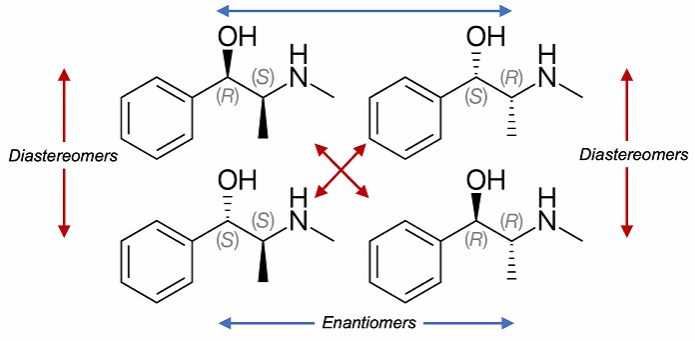
Enantiomers - 2 stereoisomers that are related to each other by a reflection. They are mirror images of each other and are non-superimposable. These have the same physical and chemical properties in non-chiral environments (except in the way they rotate plane-polarised light). However in chiral environments (such as biological systems) they react differently.
Diastereomers - Stereoisomers that have different configurations at one or more (but not all) stereocentres. (Not mirror images like enantiomers). This also includes cis/trans isomers
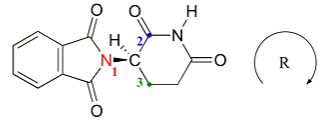
Assigning stereocentres - R/S nomenclature
Assign priority of the 4 groups using the Cahn-Ingold-Prelog convention (highest priority = greatest atomic number)
Rotate the molecule so that the lowest priority group points away from you
If the direction from highest to lowest priority group is clockwise (steer right) then it is the R enantiomer, if the direction from the highest to lowest priority group is anticlockwise (steer left) then it is the S enantiomer
Fisher projection
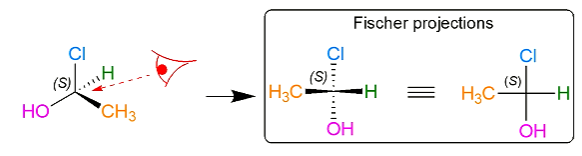
If the lowest priority group sits on the horizontal bond;
and the direction from highest priority to lowest priority group is clockwise then it is the S enantiomer
and the direction from highest priority to lowest priority group is anticlockwise then it is the R enantiomer
If the lowest priority group sits on the vertical bond;
and the direction from highest to lowest priority group is clockwise then it is the R enantiomer
and the direction from highest priority to lowest priority group is anticlockwise then is is the S enantiomer
D/L nomenclature
This nomenclature is mainly used for biological molecules and is based on the smallest 3-carbon monosaccharide, glyceraldehyde
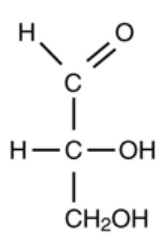
Monosaccharides:
Aldehyde is drawn at the top, then the chiral carbon at the bottom is used to determine which enantiomer it is.
If the -OH at this carbon is on the right, it is the D enantiomer.
If the -OH is on the left, it is the L enantiomer.
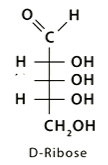
Most natural monosaccharides are D-monosaccharides
Amino acids:
-COOH group is drawn at the top so that the -NH2 group and H atom sit on the horizontal bond
If the -NH2 group is on the left then it is the L enantiomer
If the -NH2 group is on the right then it is the D enantiomer
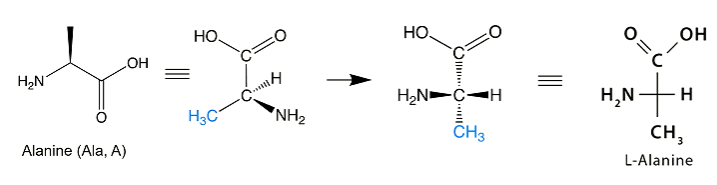
All natural amino acids are L-amino acids
Cis/trans isomers
Cis/trans isomers (or geometric isomers) are stereoisomers describing the relative orientation of functional groups within a molecule. Geometric isomers occur in cases where there is restricted rotation about a bond. They usually have different physical and chemical properties
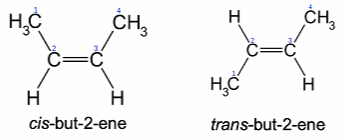
The cis isomer has the identical groups on the same side of the double bond, while the trans isomer has the identical groups on the opposite sides.
E/Z nomenclature can also be used if the carbons involved in the double bond are not attached to identical groups. The Z isomer has the highest priority groups on the same side of the double bond while the E isomer has the highest priority groups on opposite side of the double bond. Priority is assigned using the Cahn-Ingold-Prelog convention.
Conformers and rotamers
Conformational isomerism: a form of stereoisomerism in which isomers can be interconverted by rotations about formally single bonds
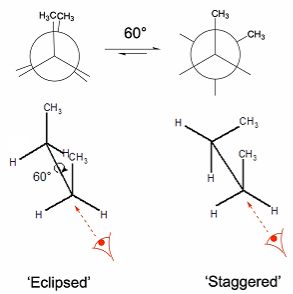
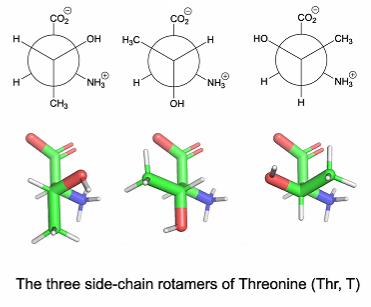
Rotamers: Set of conformers that arise from restricted rotation around a single bond, corresponding to local energy minima
Cycloalkanes: strain and conformational isomers
Cyclopropane - highly strained, sp3 carbons with bond angles of 60o. No conformational isomers
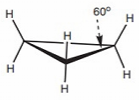
Cyclobutane: Also strained (bond angles 90o). Butterfly conformers preferred over fully eclipsed planar.
Cyclopentane: More stable (bond angles 108o). 2 buckled isomers, planar is too strained due to eclipsed bonds
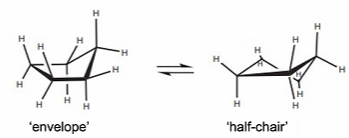
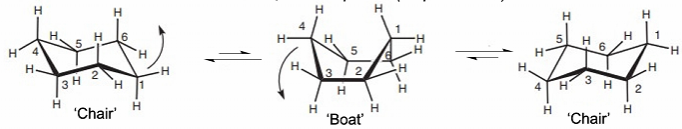
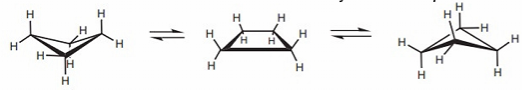
Cyclohexane: all conformers allow ~ 109.5o bond angles. Boat is less stable due to repulsion between eclipsed bonds. The chair conformation can flip, exchanging axial and equatorial positions