M6L3 Tumour angiogenesis
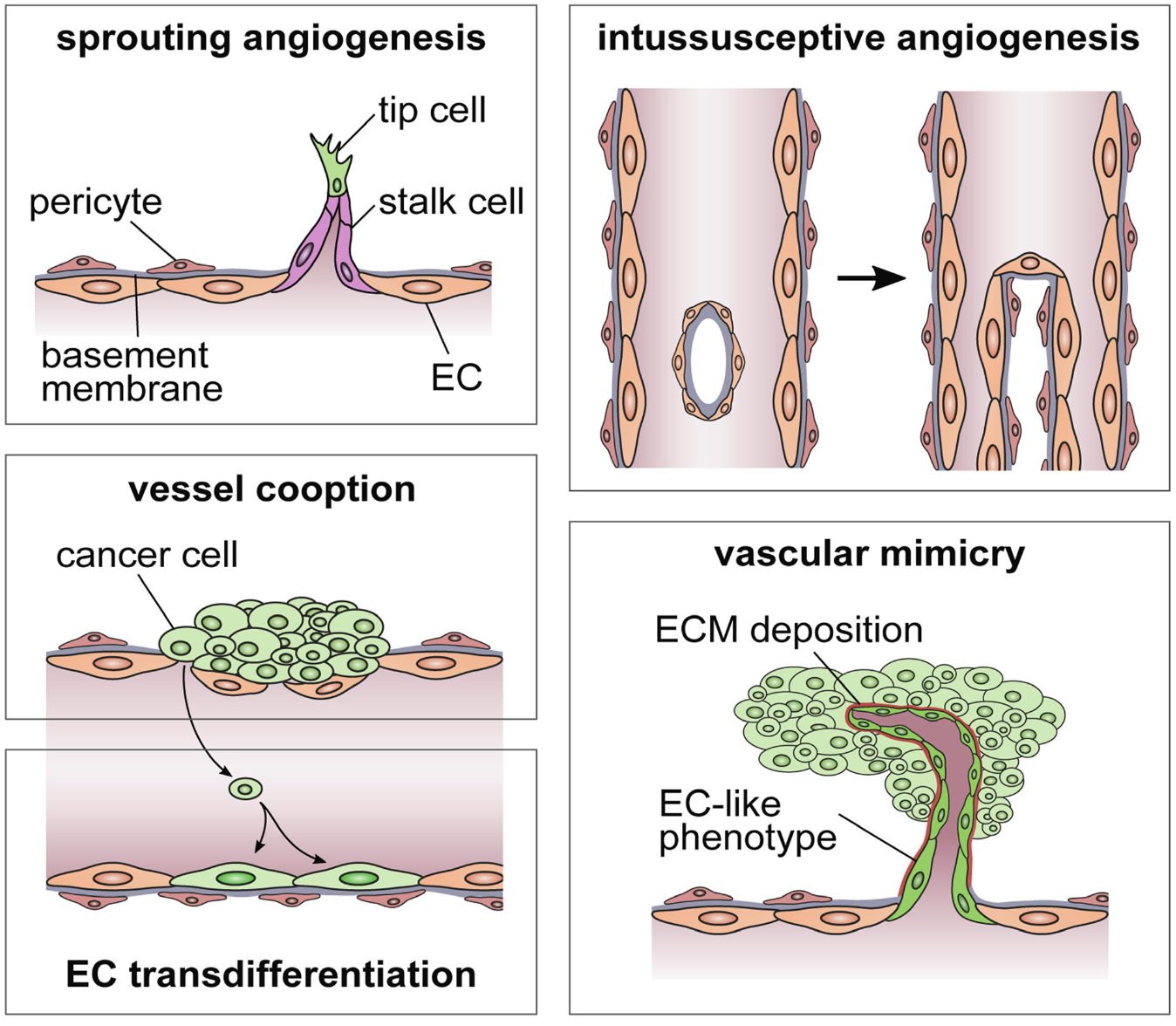
Types of angiogenesis
Sprouting
Vessel cooption
Intussusceptive angiogenesis
Vascular mimicry
Growth of new vessels from pre-existing vessels - used for growth, invasion, metastasis
Angiogenic factors (VEGF) can induce immunosuppression and chemo/radioresistance
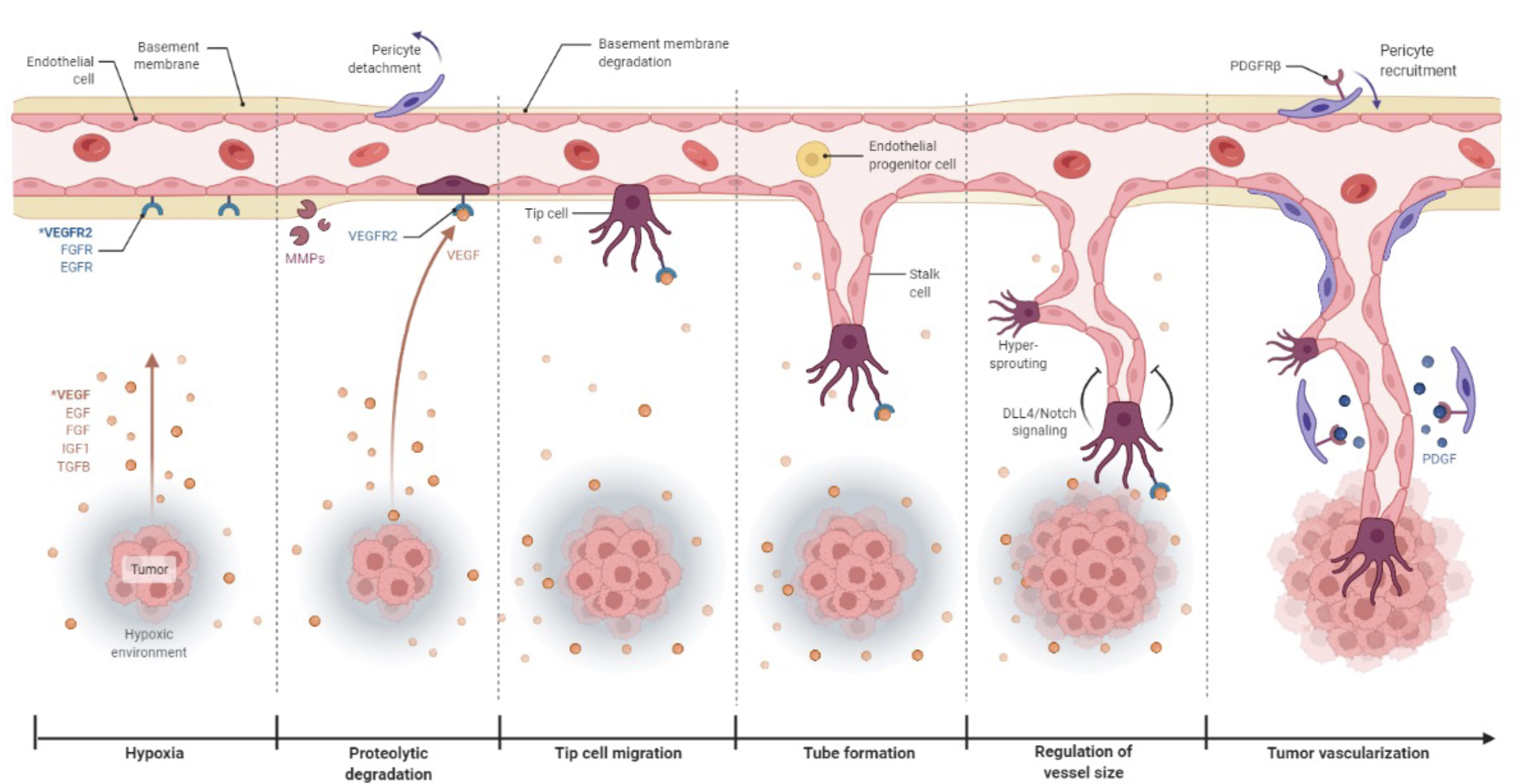
Steps in angiogenesis: hypoxia stimulates pericytes to separate off, BM breaks down and tip cell differentiates and migrates, stalk cells produce sprouting…
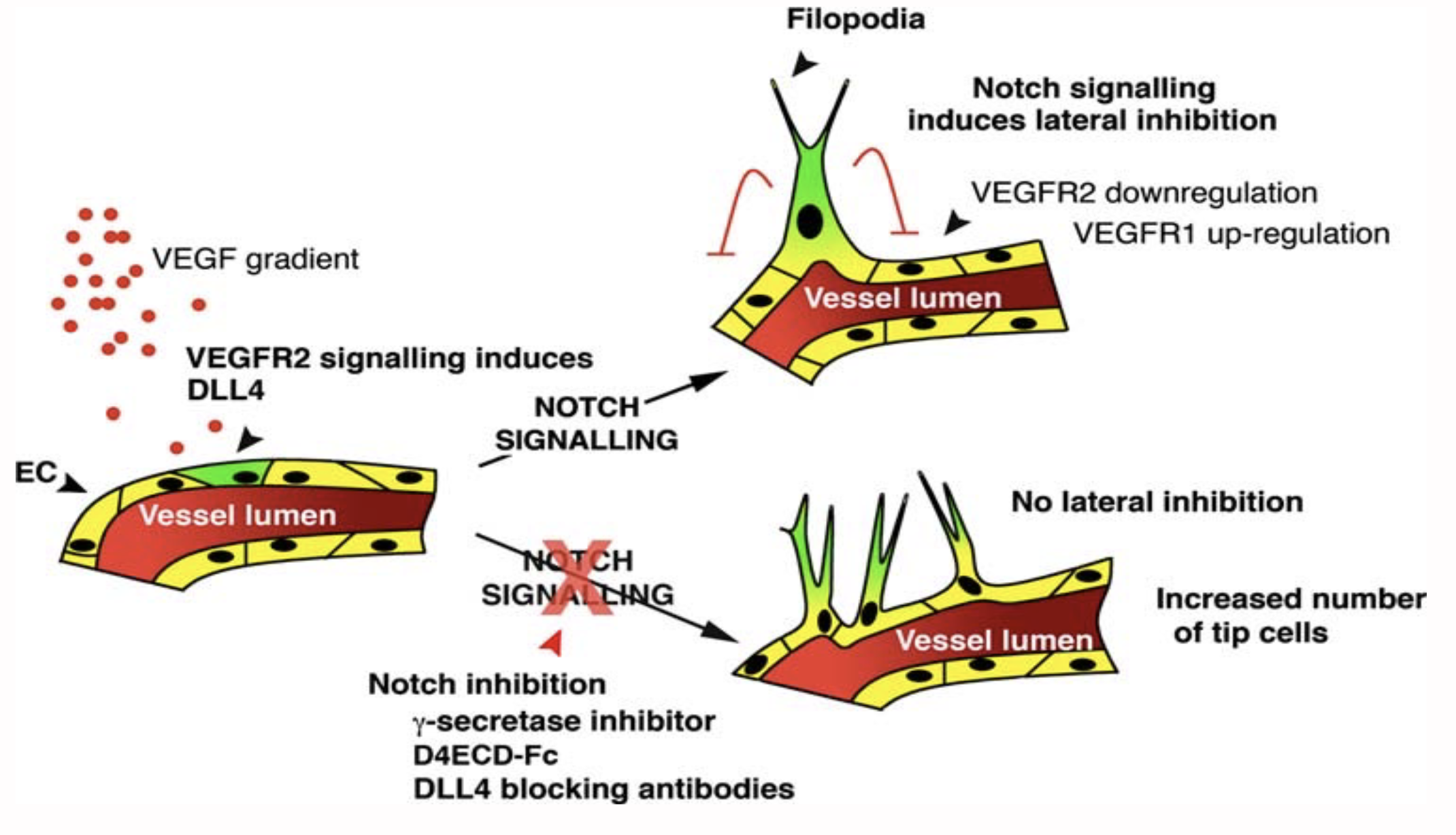
VEGF induces DLL4 —> Notch signalling induces lateral inhibition (closes off the vessels)
High angiogenesis correlated with poor outcomes
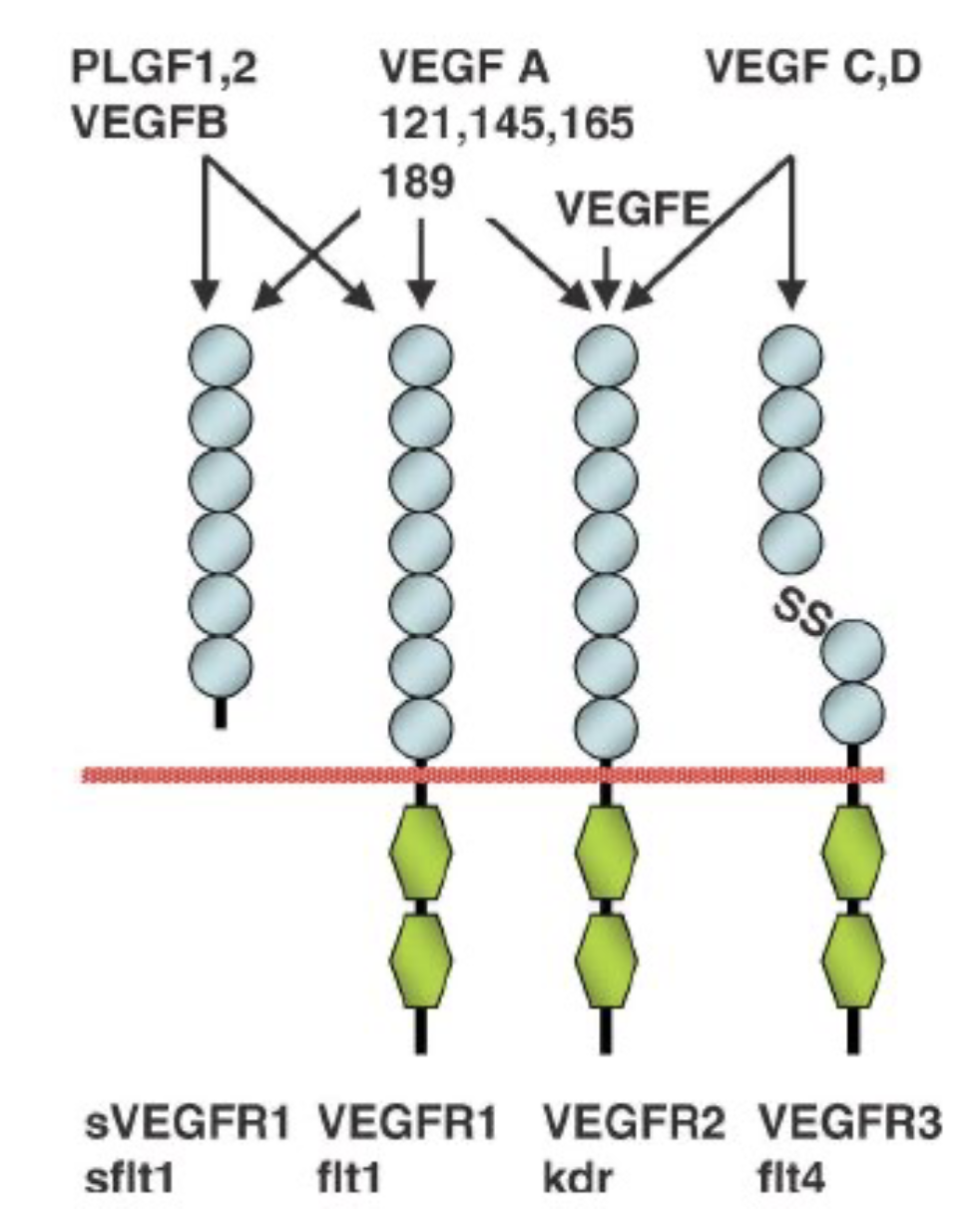
Many isoforms of VEGF receptors
Fruquitinib - potent, highly selective small molecule inhibitor of VEGFR1/2/3 tyrosine kinases for cancer therapy
Tumour abnormal vasculature - blind ends, temporary occlusions, breaks in vessel walls, AV shunt, red blood cell accumulation…
Oxygen diffusion distance is 40-140um
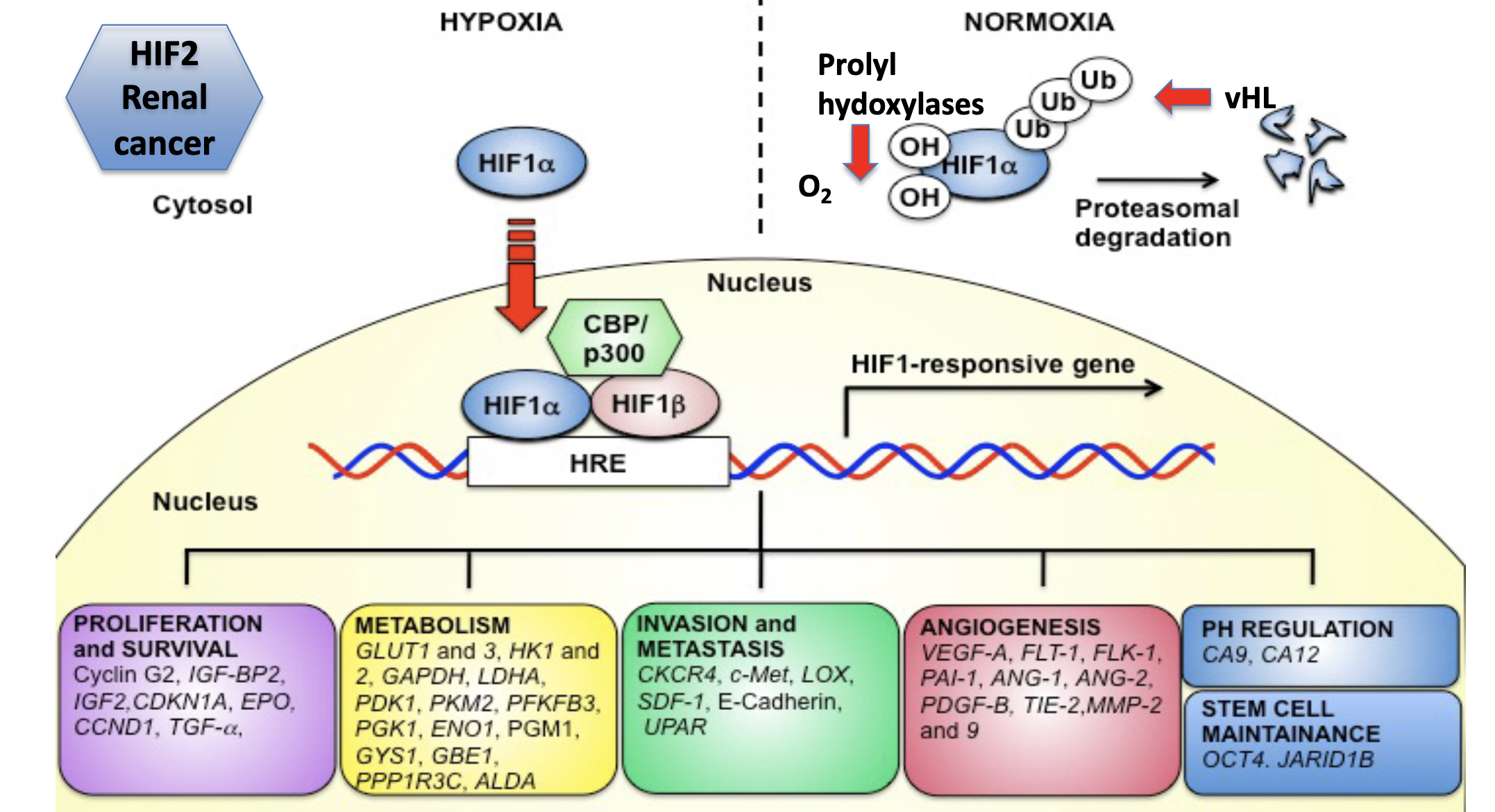
Under normoxia prolyl hydroxylases hydroxylate HIF1a and vHL ubiquitinate it for degradation, but under normaxia HIF1a is stabilised, goes to the nucleus and binds HIF1b and activates HIF1 responsive genes
26 gene hypoxia response profile can be measured using RNA sequencing and related to patient outcomes
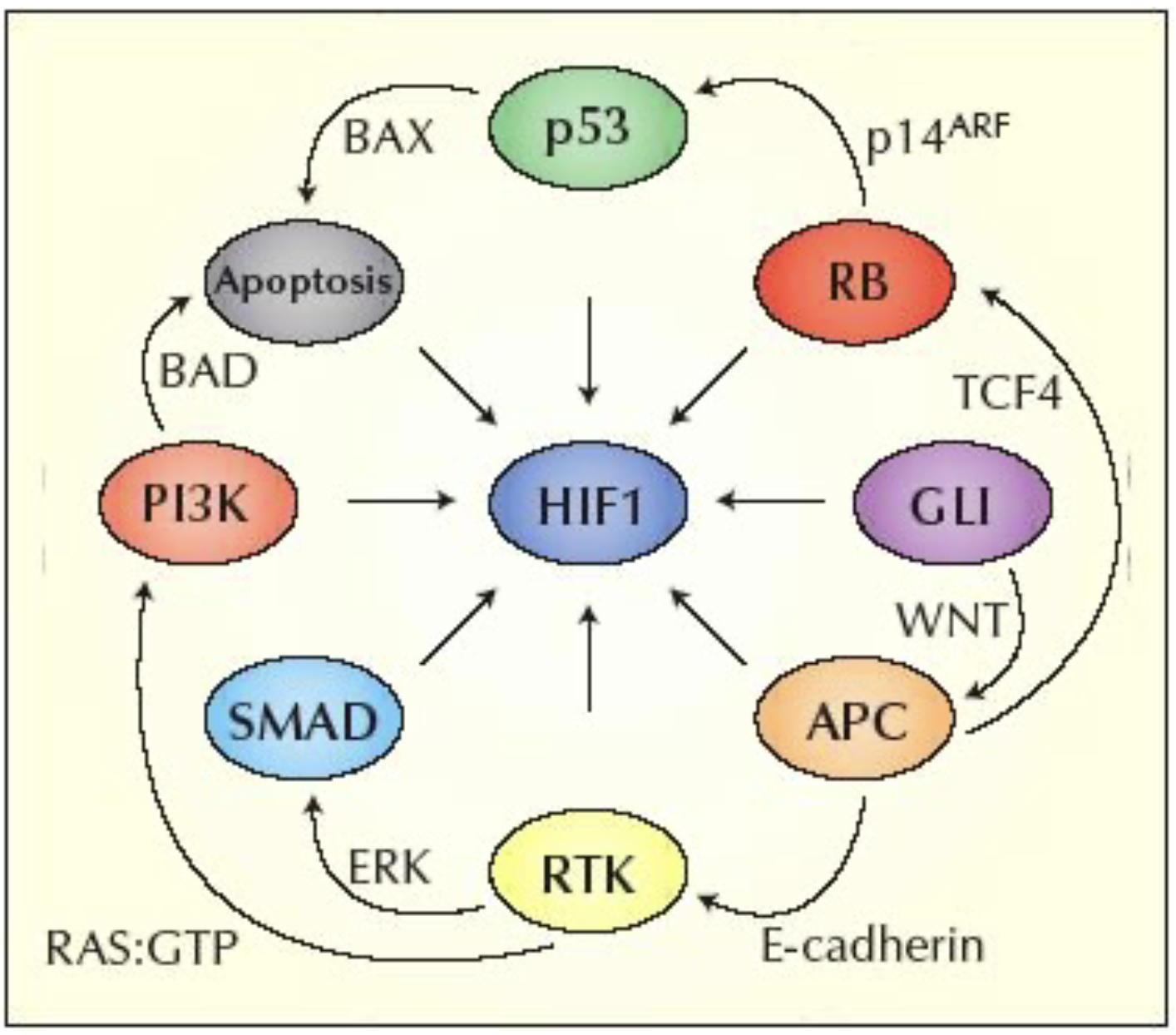
Other cancer pathways can interact with HIF even in the absence of hypoxia
Multiple cell types are involved in angiogenesis - including lyphoangiogenesis (VEGFR3), stroma can also make VEGF, bone marrow cells can differentiate into endothelial cells, myeloid cells (VEGFR1)…
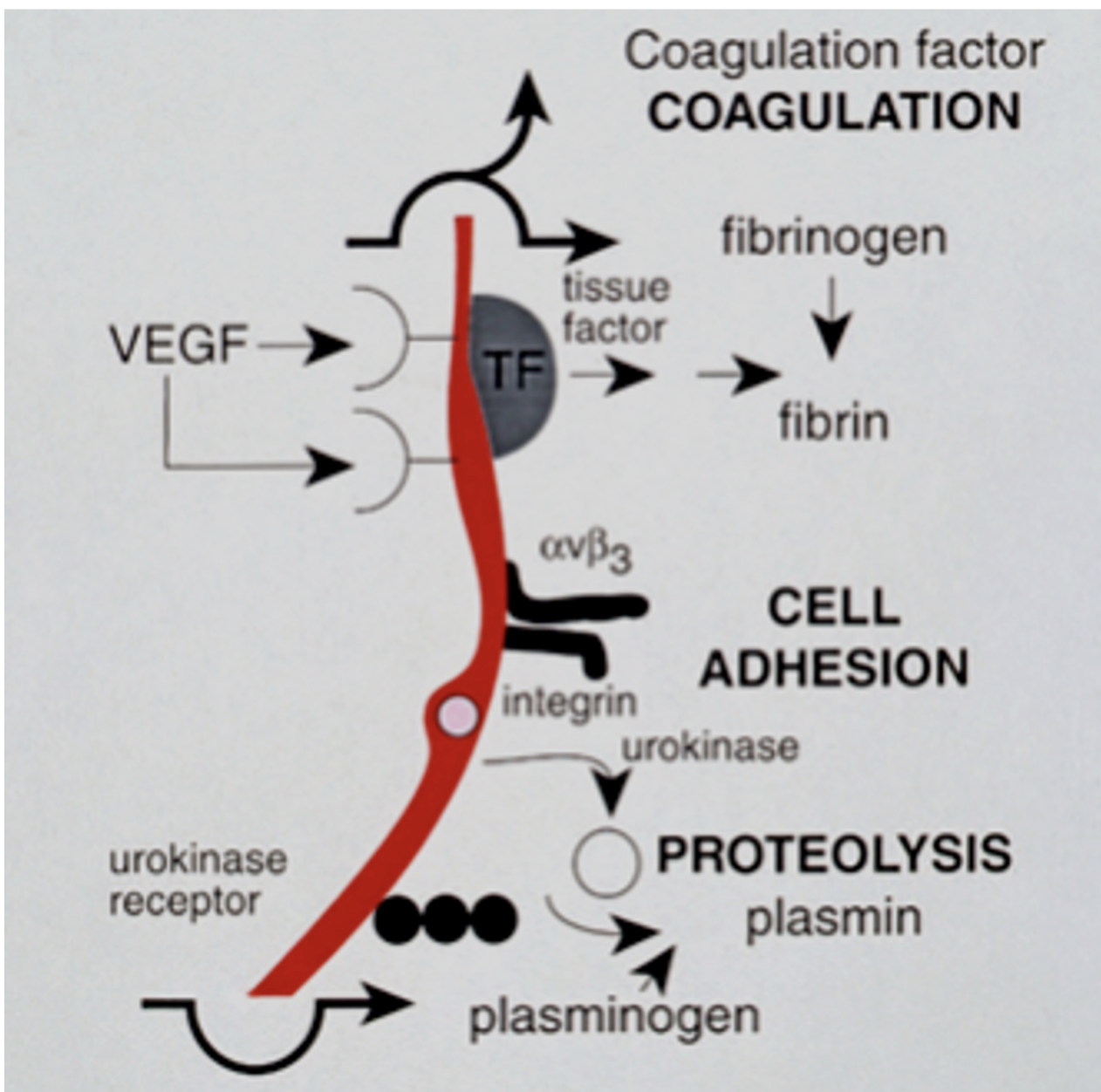
Coagulation —> cell adhesion —> proteolysis
Various other proangiogenic molecules other than VEGF

Various other angiogenic inhibitory proteins
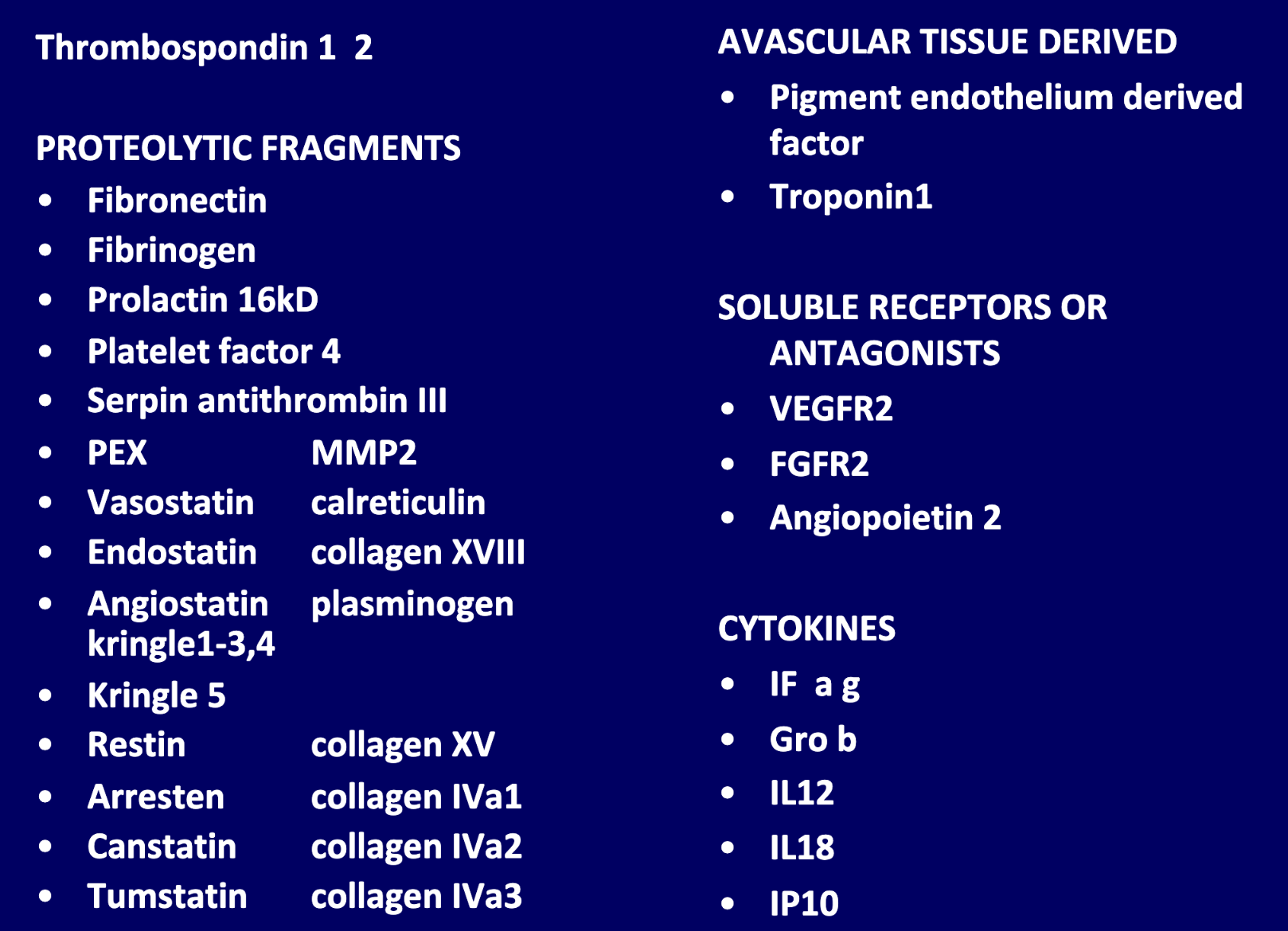
Lens of the eye does not have blood vessels due to anti angiogenic factors
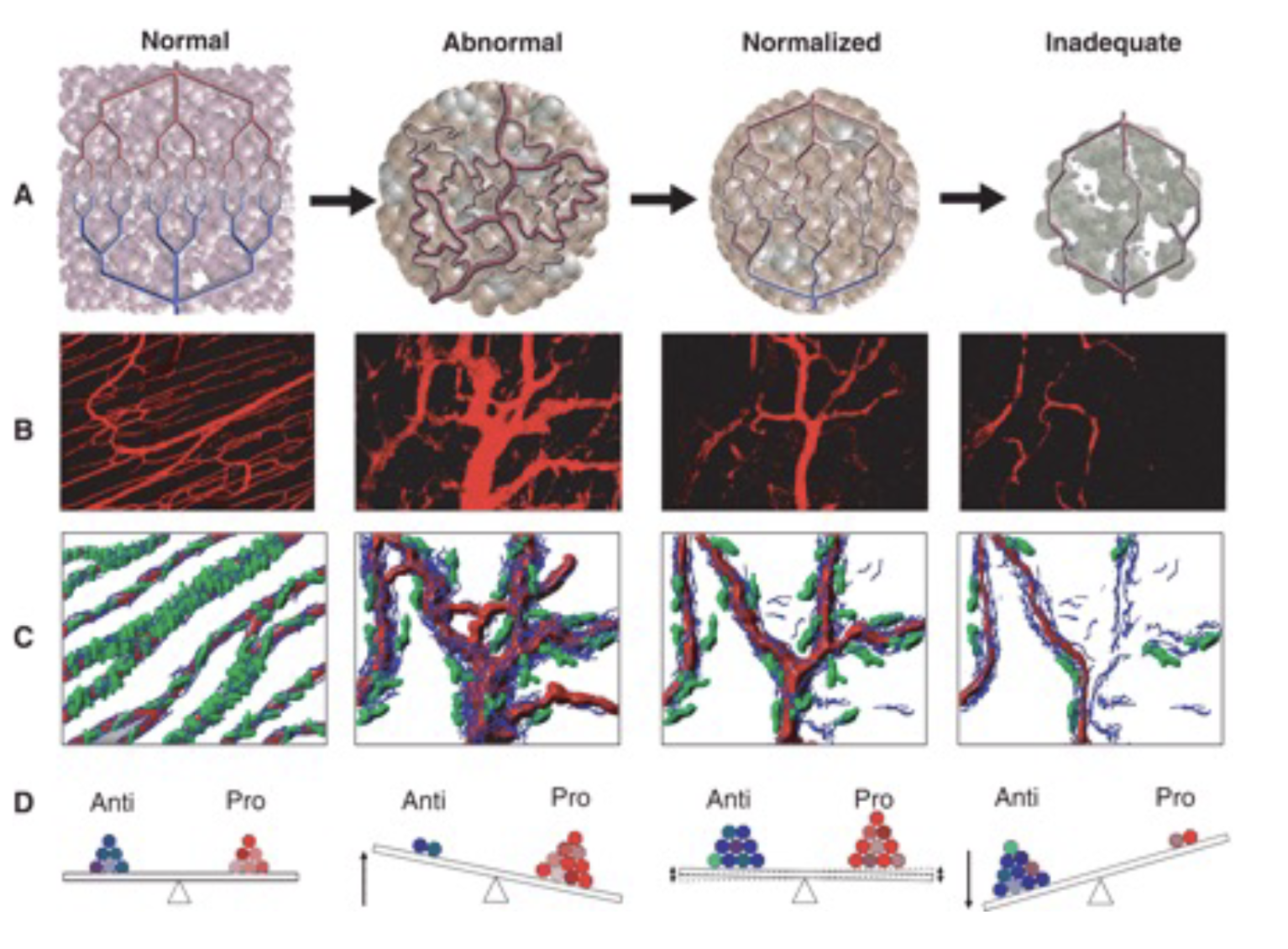
Angiogenic switch
Vascular normalisation occurs as tumour vasculature develops, which is the ideal window for drug delivery
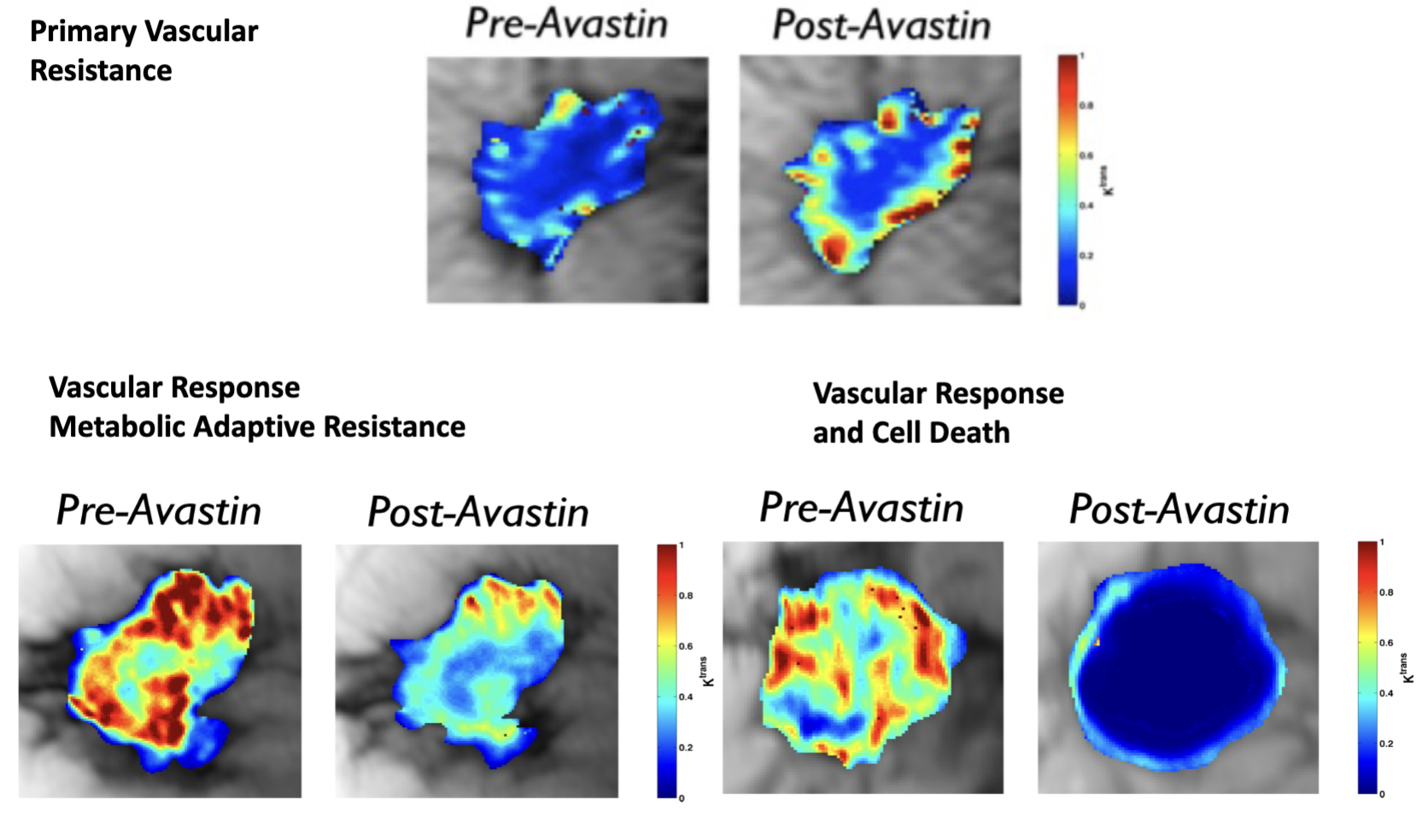
Patterns to bevacizumab resistance - primary vascular resistance, vascular response/metabolic adaptive resistance, vascular response/cell death
Upregulation of genes involved in key cancer pathways (hypoxia and metabolism, immune response and cytokines, angiogenesis…) after bevacizumab suggest possible mechanism of resistance
In vivo CA9 inhibition synergises with bevacizumab
Vessel beds are tissue specific and have different functions, which may underly tumour characteristics depending on the site of occurence
Endothelial metabolism can also be a target of treatment
ECs on tip use glycolysis
Stalk cells use fatty acid oxidation
Acidosis and hypoxia can have direct effects on immune cells as well as indirect physical effects
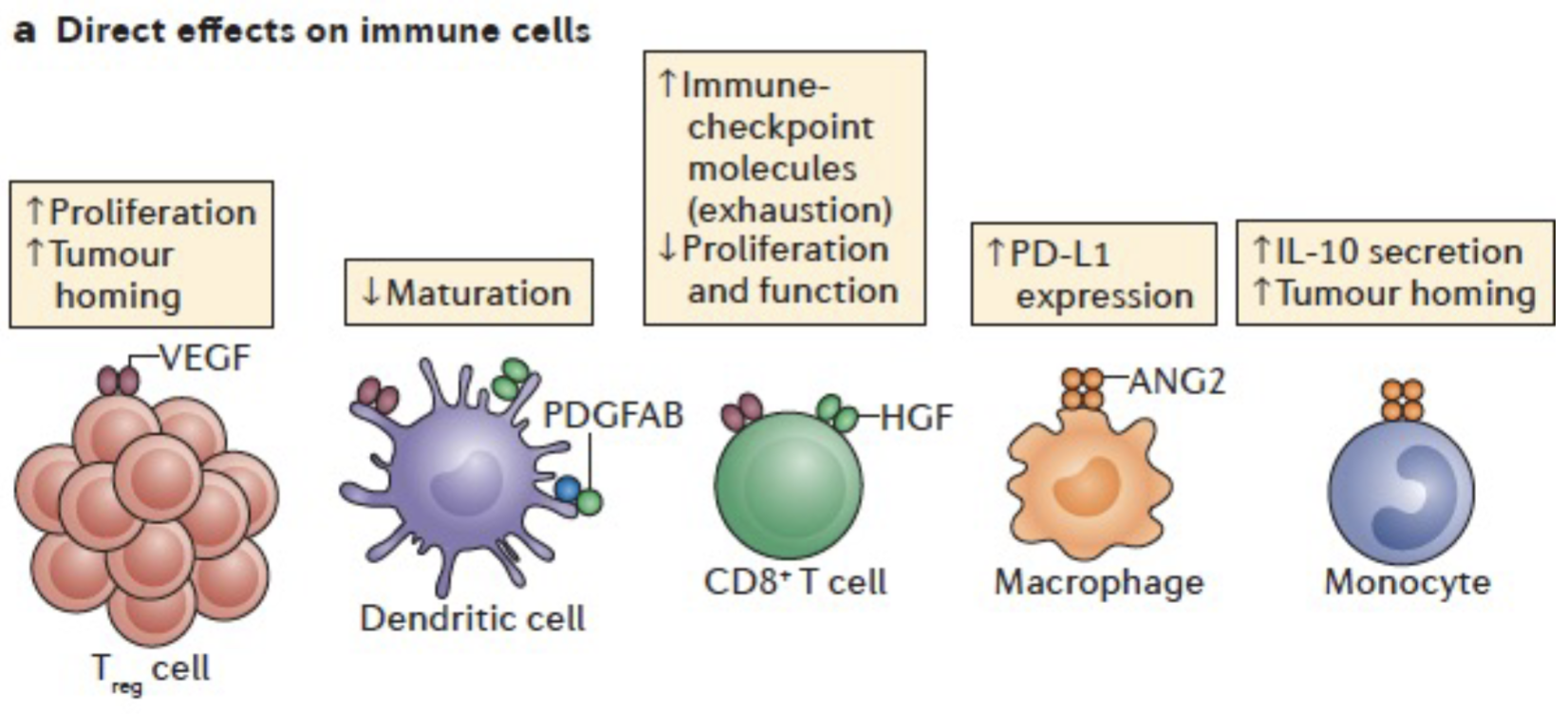
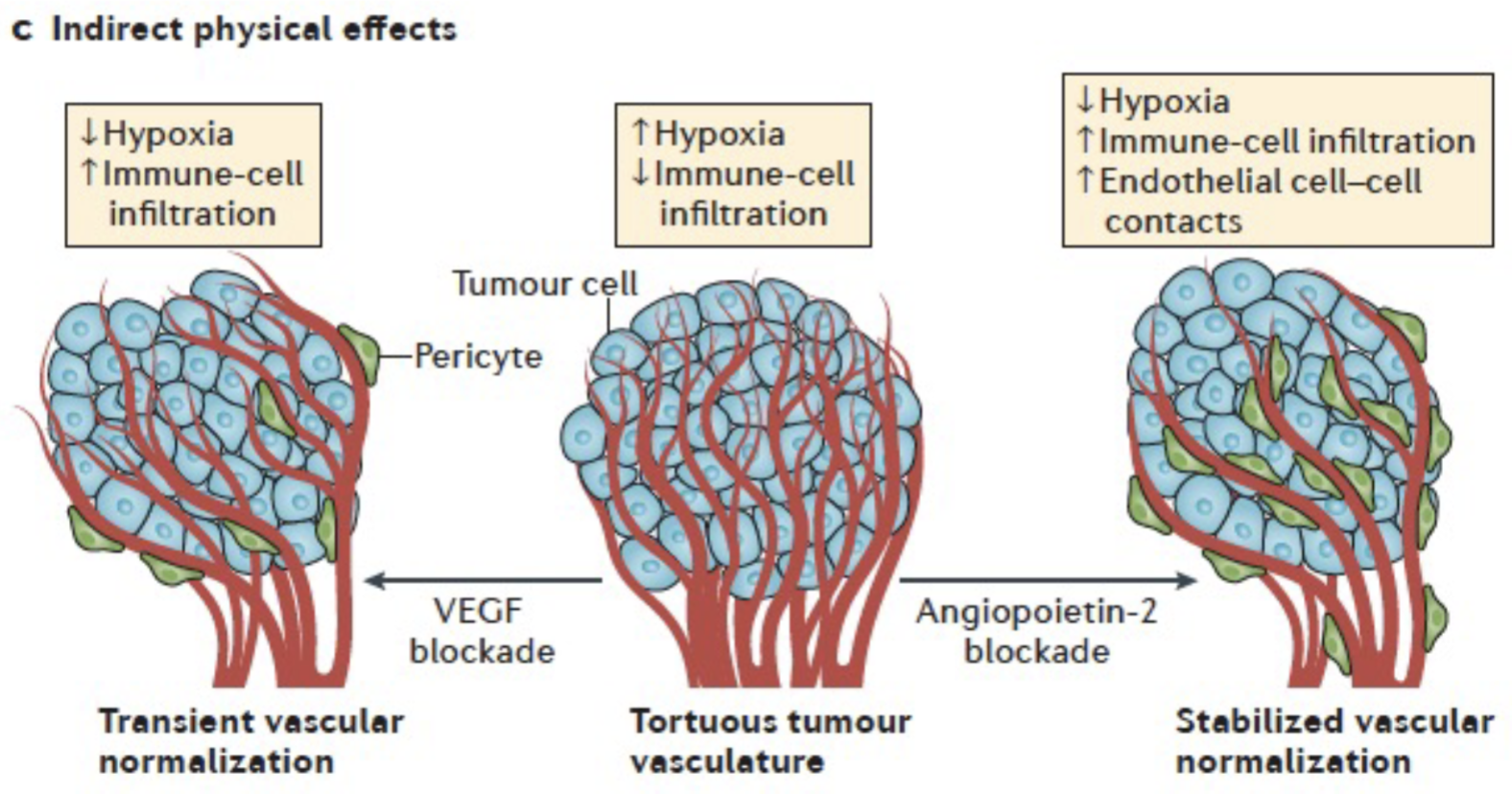
Anti-angiogenic therapy + immunotherapy implications: anti-angiogenics may exacerbate hypoxia and increase immune resistance, or it could cause blood vessel normalisation which is a good window to deliver immunotherapy - hard to predict
PDL1 upregulated in tumours relapsing for anti-angiogenic therapy and IF-g increases PDL1 during antiangiogenic therapy
Anti-PDL1 sustains response to VEGF blockade and enhances vessel normalisation during anti VEGF
Hypoxia activated prodrugs synergise with immunotherapy