28.1 Carbon-carbon bond formation
Carbon-carbon bond formation in synthesis
Reactions that form carbon-carbon bonds have a central importance to organic chemistry as they provide a means of synthesising new compounds containing more carbon atoms.
Formation of nitriles (group -CN)
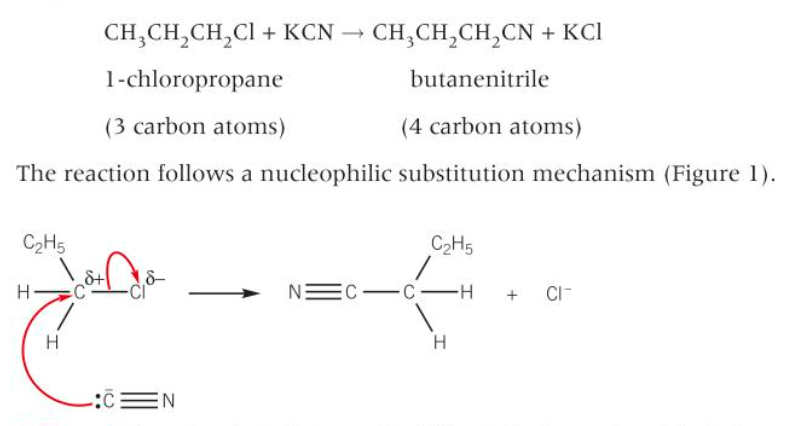
Nitriles from haloalkanes
nitriles can be formed by reacting haloalkanes with sodium cyanide, NaCN or potassium cyanide KCN, in ethanol. In this reaction the length of the carbon chain is increased.
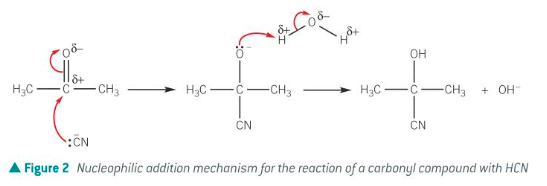
Nitriles from aldehydes and ketones
Aldehydes and ketones will react with hydrogen cyanide in a nucleophilic addition reaction.
CH3COCH3 + HCN → CH3C(OH)(CN)CH3
Product is known as hydroxynitrile or cyanohydrin.
The reaction forms a carbon-carbon bond, thus increasing the number of carbon atoms in the molecule.
Hydrogen cyanide is far too poisonous to use, an increased reaction rate can be obtained in the presence of cyanide ions.
A mixture of sodium cyanide and sulfuric acid is used, which improves safety and increases reaction rate.
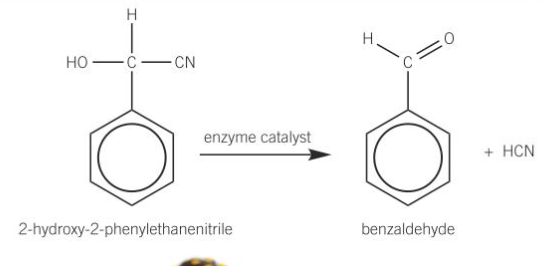
Hydroxynitriles in nature
A species of millipede stores 2-hydroxy-2-phenylethanenitrile in one of its body segments. When it is attacked, it is released from a storage chamber into a reaction chamber where it mixes when an enzyme.
The reaction between the enzyme and hydroxynitrile releases a very toxic hydrogen cyanide together, together with benzaldehyde. The millipede secretes this toxic mixture from the glands located in its body segments. An adult millipede can produce 0.6 mg of hydrogen cyanide which is enough to kill ants and deter most of their other enemies.
Reduction of nitriles
Nitriles can be reduced to amine by reacting with hydrogen in the presence of nickel catalyst.

Hydrolysis of nitriles
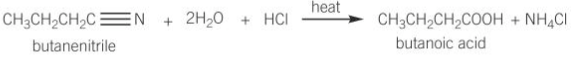
Nitriles undergo hydrolysis to form carboxylic acids by heating with dilute aqueous acid, for example HCL(aq)
Forming carbon-carbon bonds in benzene rings
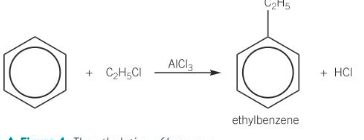
Alkylation
This is a reaction that transfers an alkyl group from a haloalkane to a benzene ring.
The reaction takes place in the presence of a Friedel-Crafts catalyst such as aluminium chloride.
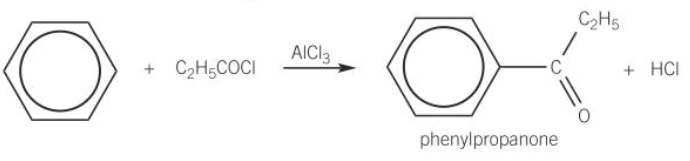
Acylation
When benzene reacts with a acyl chloride in the presence of an aluminium chloride catalyst, a ketone is formed.
This reaction is useful in synthesis as the organic product undergoes the typical reactions of a ketone.
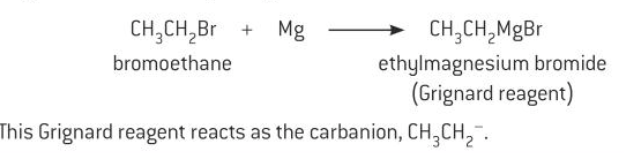
Organometallic compounds in carbon-carbon bond formation
Compounds that contain a carbon-metal bond are known as organometallic compounds. Organometallic compounds are very important for forming carbon-carbon bonds in organic compounds to increase the length of a carbon chain.
Organometallic compounds containing magnesium or lithium are often used for this purpose.
An organometallic compounds provides a source of a nucleophile called a carbanion, which contain a carbon atom with a negative charge.
The carbanion reactions with a positively charged carbon atom to form a new carbon-carbon bond.
Grignard reagents
Organometallic compounds called Grignard reagents are made by reacting magnesium with an alkyl or aryl haloalkanes dissolved in ether solvent.